 Back
BackChemical Kinetics: Reaction Rates, Rate Laws, and Catalysis
Study Guide - Smart Notes

Chemical Kinetics
Introduction to Kinetics
Chemical kinetics is the study of the rates at which chemical reactions occur and the factors that affect these rates. Understanding kinetics allows chemists to control reaction speed and optimize conditions for desired outcomes.
Reaction Rate: The change in concentration of a reactant or product per unit time, typically expressed in molarity (M).
Kinetics: The branch of chemistry concerned with reaction rates and mechanisms.
Reaction Rate Expressions
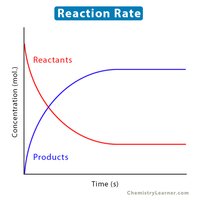
Reaction rates are always positive and can be written in terms of reactant or product concentrations. The rate expression is a mathematical representation of the reaction rate.
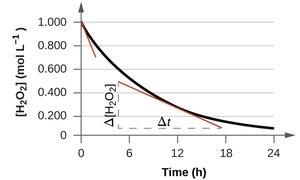
Average Reaction Rate: Calculated over a time interval using initial and final concentrations.
Instantaneous Reaction Rate: The rate at a specific moment, determined by the slope of a tangent to a concentration vs. time curve.
Initial Reaction Rate: The instantaneous rate at time zero.
Example: For the decomposition of hydrogen peroxide: 2H2O2(aq) → 2H2O(l) + O2(g)
Relative Rates and Stoichiometry
The rate of a reaction can be expressed in terms of any reactant or product. Stoichiometric coefficients from the balanced equation relate the rates of change for each species.
Factors Affecting Reaction Rates
Chemical Nature of Reactants
The inherent properties of reactants, such as their position in the periodic table, influence reaction speed. For example, alkali metals react faster with water as you move down the group.
State of Subdivision
Increasing the surface area of reactants increases the reaction rate by enhancing contact between reactant particles.

Temperature
Raising the temperature increases the kinetic energy of molecules, leading to more frequent and energetic collisions, thus increasing the reaction rate.
Concentration
Higher concentrations of reactants result in more collisions and a faster reaction rate.
Catalysts
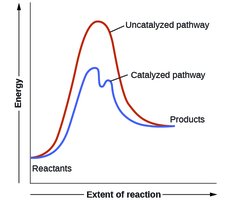
Catalysts increase reaction rates by providing an alternative pathway with lower activation energy, without being consumed in the reaction.
Rate Laws
Definition and Formulation
Rate laws express the relationship between reaction rate and reactant concentrations. Only reactants appear in the rate law.
General form:
k: Rate constant, specific to the reaction and temperature.
m, n: Reaction orders, determined experimentally.
Reaction Order
The order with respect to each reactant indicates how the rate depends on its concentration.
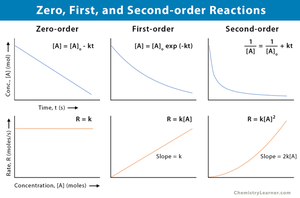
Zero Order: Rate is independent of concentration.
First Order: Rate is directly proportional to concentration.
Second Order: Rate is proportional to the square of concentration.
Overall Order: Sum of individual orders.
Determining Rate Laws
Rate laws are determined experimentally by measuring initial rates with varying concentrations of reactants.
Vary one reactant's concentration while keeping others constant.
Measure the change in rate to determine the order with respect to that reactant.
Integrated Rate Laws
Concentration-Time Relationships
Integrated rate laws relate reactant concentration to time, depending on reaction order.
First Order:
Second Order:
Zero Order:
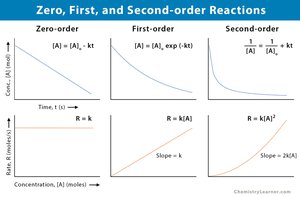
Half-Life
The half-life is the time required for half of the reactant to be consumed.
First Order: Half-life is independent of initial concentration.
Second Order and Zero Order: Half-life depends on initial concentration.
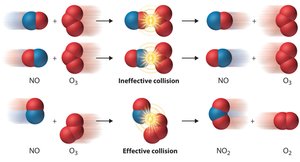
Collision Theory
Principles of Collision Theory
Collision theory states that reactants must collide to react. The rate of reaction is proportional to the frequency of collisions.
Effective Collisions: Require proper orientation and sufficient kinetic energy.
Activation Energy (Ea): Minimum energy required for a reaction to occur.
Transition State and Reaction Energy Diagram
Reactants form an activated complex at the transition state, which is the highest energy point along the reaction path.
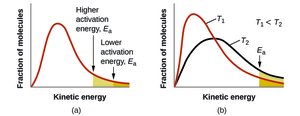
Temperature and Reaction Rate
Kinetic Theory and Arrhenius Equation
Temperature increases the fraction of molecules with energy equal to or greater than Ea, thus increasing reaction rate. The Arrhenius equation relates rate constant to temperature and activation energy:
Taking the natural logarithm:
Catalysis
Role of Catalysts
Catalysts lower the activation energy, providing an alternative pathway for the reaction. They are regenerated and not consumed in the process.
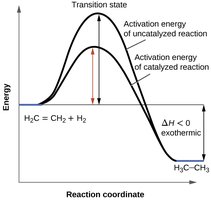
Types of Catalysts
Homogeneous Catalysts: Same phase as reactants; form reactive intermediates.
Heterogeneous Catalysts: Different phase; often solid catalysts with gas or liquid reactants (e.g., catalytic converters).
Enzymes: Biological protein catalysts.
----------------------------------------
