 Back
BackChemical Kinetics: Study Guide for General Chemistry
Study Guide - Smart Notes

Chemical Kinetics
Introduction to Chemical Kinetics
Chemical kinetics is the branch of chemistry that studies the rate at which chemical reactions occur, the factors that affect these rates, and the mechanisms by which reactions proceed. Understanding kinetics is essential for controlling reactions in industrial, environmental, and biological contexts.
Reaction Rate: The speed at which reactants are converted to products.
Mechanism: The stepwise sequence of elementary reactions by which overall change occurs.
Applications: Pharmaceuticals, food industry, environmental chemistry, and manufacturing.
Factors Affecting Reaction Rate
Several factors influence how quickly a reaction occurs.
Concentration: Higher concentration increases collision frequency, thus increasing rate.
Surface Area: Greater surface area allows more collisions, especially for solids.
Temperature: Higher temperature increases kinetic energy and collision frequency.
Energy: Only collisions with sufficient energy (activation energy) lead to reaction.

Measuring Reaction Rates
Reaction rate is measured as the change in concentration of reactants or products per unit time.
Average Rate: Calculated over a time interval.
Instantaneous Rate: Rate at a specific moment, given by the slope of the tangent to the concentration vs. time curve.
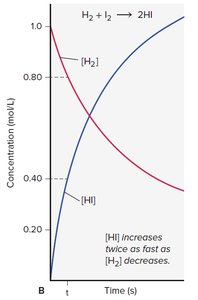
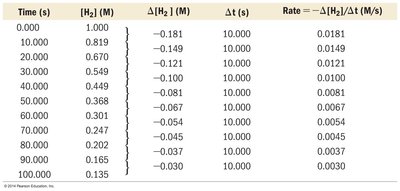
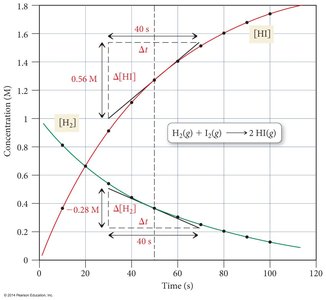
Stoichiometry and Rate Expressions
The rate of disappearance of reactants and appearance of products is related to their stoichiometric coefficients.
General Rate Expression: For a reaction ,
Rate Laws and Reaction Order
Rate laws express the relationship between reaction rate and reactant concentrations.
Rate Law: (for a simple reaction)
Order of Reaction: The exponent indicates how the concentration of A affects the rate. It is determined experimentally.
Overall Order: Sum of exponents in the rate law.
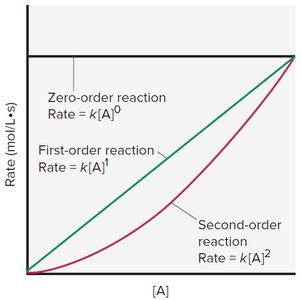
Examples of Reaction Orders
[A] (M) | Initial Rate (M/s) |
|---|---|
0.10 | 0.015 |
0.20 | 0.030 |
0.40 | 0.060 |
First Order: Rate doubles as concentration doubles.
[A] (M) | Initial Rate (M/s) |
|---|---|
0.10 | 0.015 |
0.20 | 0.015 |
0.40 | 0.015 |
Zero Order: Rate is independent of concentration.
[A] (M) | Initial Rate (M/s) |
|---|---|
0.10 | 0.015 |
0.20 | 0.060 |
0.40 | 0.240 |
Second Order: Rate quadruples as concentration doubles.
Units of Rate Constant (k)
Overall Reaction Order | Units of k |
|---|---|
0 | mol/L·s |
1 | 1/s |
2 | L/mol·s |
3 | L2/mol2·s |

Integrated Rate Laws
Integrated rate laws relate reactant concentration to time for different reaction orders.
First Order:
Second Order:
Zero Order:
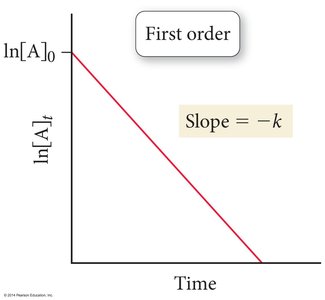
![First order reaction: ln[A] vs. time](https://static.studychannel.pearsonprd.tech/study_guide_files/general-chemistry/sub_images/83035cc9_image_11.png)
![First order reaction: ln[A] vs. time with slope -k](https://static.studychannel.pearsonprd.tech/study_guide_files/general-chemistry/sub_images/83035cc9_image_13.png)
![Second order reaction: 1/[A] vs. time](https://static.studychannel.pearsonprd.tech/study_guide_files/general-chemistry/sub_images/83035cc9_image_14.png)
![Second order reaction: 1/[A] vs. time with slope k](https://static.studychannel.pearsonprd.tech/study_guide_files/general-chemistry/sub_images/83035cc9_image_15.png)
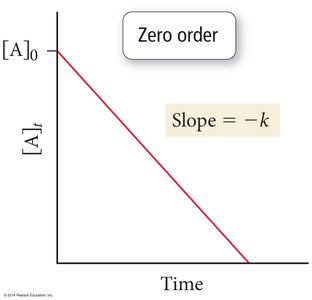
![Zero order reaction: [A] vs. time](https://static.studychannel.pearsonprd.tech/study_guide_files/general-chemistry/sub_images/83035cc9_image_18.png)
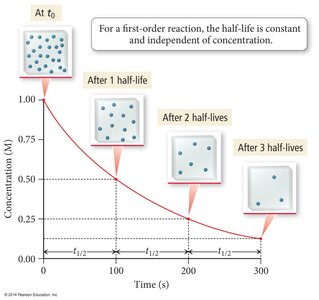
Half-Life of Reactions
The half-life () is the time required for the concentration of a reactant to decrease by half.
First Order: (independent of initial concentration)
Second Order: (depends on initial concentration)
Zero Order: (depends on initial concentration)
Theories of Chemical Kinetics
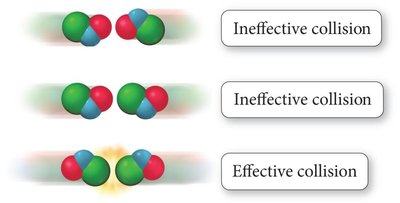
Collision Theory
Collision theory states that reactant particles must collide with sufficient energy and proper orientation to react.
Effective Collision: Leads to product formation.
Ineffective Collision: Does not result in reaction.
Activation Energy (): Minimum energy required for reaction.
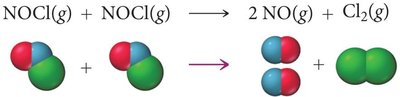
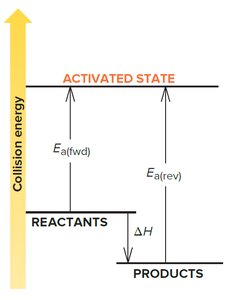
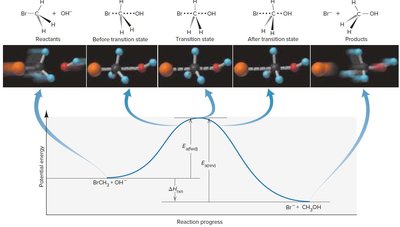
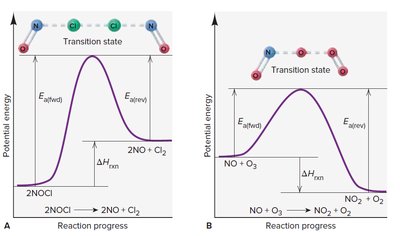
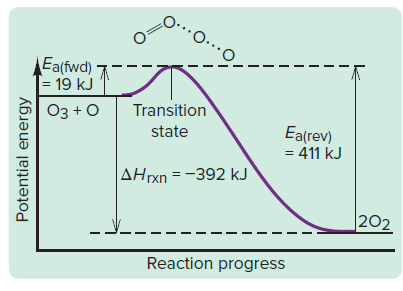
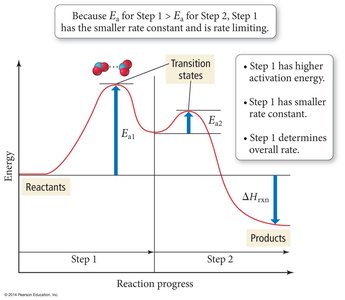
Energy Profile and Transition State Theory
The transition state theory describes the highest energy state (activated complex) during a reaction.
Energy Profile: Shows energy changes as reactants convert to products.
Transition State: Unstable, high-energy state between reactants and products.
Activation Energy: Difference in energy between reactants and transition state.
Enthalpy Change (): Difference in energy between reactants and products.
Temperature and Reaction Rate
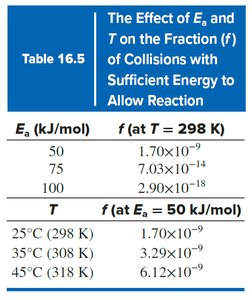
Increasing temperature increases the rate constant and reaction rate by raising the fraction of collisions with sufficient energy.
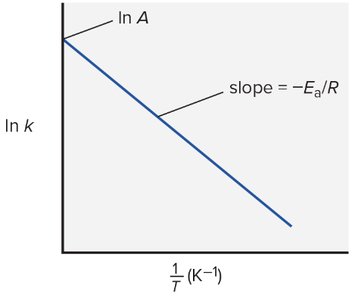
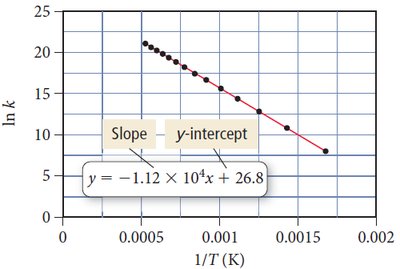
Arrhenius Equation:
Effect of Temperature: Higher T increases k and rate.
Effect of Activation Energy: Lower increases rate.
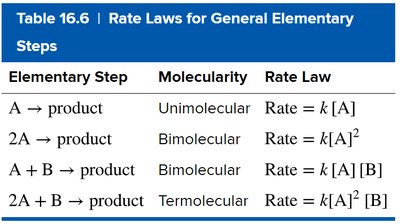
Reaction Mechanisms and Molecularity
Mechanism: Sequence of elementary steps.
Molecularity: Number of reactant particles in an elementary step (unimolecular, bimolecular, termolecular).
Rate-Determining Step: Slowest step, limits overall rate.
Catalysts
Catalysts increase reaction rate by providing an alternative pathway with lower activation energy.
Homogeneous Catalysis: Catalyst in same phase as reactants.
Heterogeneous Catalysis: Catalyst in different phase.
Enzymes: Biological catalysts, highly specific.
Summary Table: Key Equations and Concepts
Concept | Equation |
|---|---|
General Rate Law | |
First Order Integrated | |
Second Order Integrated | |
Zero Order Integrated | |
First Order Half-Life | |
Second Order Half-Life | |
Zero Order Half-Life | |
Arrhenius Equation |
Practice and Application
Use experimental data to determine reaction order and rate constant.
Apply integrated rate laws to calculate concentrations at given times.
Interpret energy diagrams to identify activation energy and enthalpy change.
Recognize the effect of temperature and catalysts on reaction rate.
Additional info: This guide expands on lecture notes and textbook images, providing definitions, equations, and visual aids for key concepts in chemical kinetics.
