 Back
BackChemical Reactions and Chemical Quantities: Combustion, Stoichiometry, and Reactivity
Study Guide - Smart Notes

Chemical Reactions and Chemical Quantities
Combustion of Fossil Fuels and the Greenhouse Effect
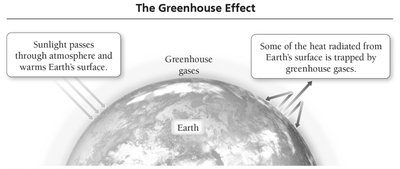
The combustion of fossil fuels is a major chemical process that releases energy and greenhouse gases into the atmosphere. Greenhouse gases, such as carbon dioxide (CO2), play a crucial role in regulating Earth's temperature by trapping heat.
Greenhouse gases allow sunlight to enter the atmosphere and warm Earth's surface.
They prevent some of the heat generated by sunlight from escaping back into space, maintaining Earth's energy balance.
The balance between incoming solar energy and outgoing heat determines Earth's average temperature.
Global Warming and Atmospheric Changes
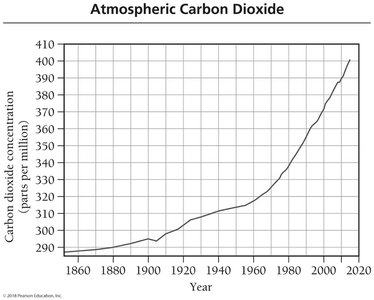
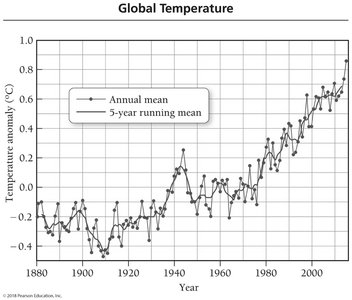
Scientific measurements show a correlation between rising atmospheric CO2 levels and increasing global temperatures since the Industrial Revolution.
Since 1860, atmospheric CO2 levels have increased by 37%.
During the same period, the average global temperature has risen by approximately 0.8 °C (1.4 °F).
Ongoing research investigates whether these trends are causally linked.
Chemical Changes and Chemical Equations
Physical vs. Chemical Changes
Understanding the difference between physical and chemical changes is fundamental in chemistry.
Physical changes alter only the state or appearance of a substance, not its composition. The identity of the atoms or molecules remains unchanged.
Chemical changes (chemical reactions) alter the composition of matter. Atoms rearrange to form new substances with different properties.
Balancing Chemical Equations
Chemical equations represent the transformation of reactants into products. They must be balanced to obey the law of conservation of mass and charge.
Equations state the molecular or ionic formulas of reactants and products.
They indicate the physical states (solid, liquid, gas, aqueous) of each substance.
Coefficients express the relative number of moles of each substance.
Example: Combustion of methane:
Example: Combustion of hexane:
Example: Reaction between lead(II) nitrate and potassium phosphate:
Stoichiometry: Quantitative Aspects of Chemical Reactions
Stoichiometric Relationships
Stoichiometry involves using balanced chemical equations to relate the amounts of reactants and products in a reaction.
The coefficients in a balanced equation specify the relative amounts (in moles) of each substance.
Stoichiometric ratios are used as conversion factors between reactants and products.
Example: Octane combustion:
Possible mole ratios: , , etc.
Mass-to-Mass Conversions
To determine the mass of a product formed from a given mass of reactant:
Convert mass of reactant (A) to moles using molar mass.
Use the stoichiometric ratio from the balanced equation to find moles of product (B).
Convert moles of product to mass using molar mass.
Example: Neutralization of stomach acid by aluminum hydroxide:
Given 3.26 g Mg(OH)2, calculate the mass of HCl neutralized.
Limiting Reactant, Theoretical Yield, and Percent Yield
In reactions with multiple reactants, the limiting reactant is the one that is completely consumed first, thus limiting the amount of product formed.
Theoretical yield: Maximum amount of product possible, based on the limiting reactant.
Actual yield: Amount of product actually obtained from the reaction (usually less than theoretical yield).
Percent yield: Ratio of actual yield to theoretical yield, expressed as a percentage.
Percent yield formula:
Example: Synthesis of ammonia:
Calculate the theoretical yield of ammonia from 2.34 kg H2 and 25.6 kg N2.
Example: Extraction of iron from iron ore:
Given 245 g Fe2O3 and 131.2 g CO, producing 82.3 g Fe, determine the limiting reactant, theoretical yield, percent yield, and mass of excess reactant remaining.
Types of Chemical Reactions
Combustion Reactions
A combustion reaction involves the reaction of a substance with O2 to form one or more oxygen-containing compounds, often releasing heat and light.
Products typically include water (H2O), carbon oxides (CO, CO2), nitrogen oxides (NO, NO2), and sulfur oxides (SO2, SO3).
Complete combustion of a carbon-based compound produces CO2 and H2O.
Reactivity of Alkali Metals
The alkali metals (Group 1A) are highly reactive due to their single valence electron (ns1 configuration). They readily form 1+ cations and react vigorously with nonmetals and water.
Reaction with halogens (X):
Reaction with water:
The reaction with water is highly exothermic and can be explosive due to ignition of hydrogen gas.

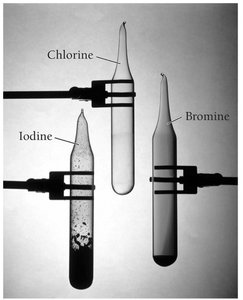
Reactivity of Halogens
The halogens (Group 7A) have ns2np5 outer electron configurations and are the most reactive nonmetals. They tend to gain one electron to form 1- anions.
React with metals to form ionic compounds (metal halides):
React with hydrogen to form hydrogen halides:
Examples of Balanced Chemical Equations
Aluminum metal and fluorine gas:
Lithium metal and water:
Hydrogen gas and bromine:
