Back
BackChemical Reactions: Energy, Rates, and Equilibrium – Study Guide
Study Guide - Smart Notes

Chemical Reactions: Energy, Rates, and Equilibrium
7.1 Energy and Chemical Bonds
Chemical bonds are associated with energy, which can be classified as potential or kinetic. Understanding these forms of energy is fundamental to predicting the behavior of chemical reactions.
Potential Energy: Stored energy due to position or arrangement. Examples include water behind a dam or a coiled spring.
Kinetic Energy: Energy of motion, such as water flowing over a dam or a moving car.
Bond Formation: When atoms form ionic or covalent bonds, potential energy is often converted to heat.
Heat: A measure of the kinetic energy of particles in a substance.
Stability: Products with less potential energy than reactants are more stable and less likely to undergo further change.
7.2 Heat Changes during Chemical Reactions
Bond dissociation energy is the energy required to break a bond in a molecule. The heat changes during reactions are governed by the law of conservation of energy.
Bond Dissociation Energy: The energy needed to break a specific bond in a molecule.
Endothermic Reaction: Absorbs heat; heat is a reactant. ΔH is positive.
Exothermic Reaction: Releases heat; heat is a product. ΔH is negative.
Law of Conservation of Energy: Energy cannot be created or destroyed, only transformed.
Heat of Reaction (ΔH): The difference between heat absorbed in breaking bonds and heat released in forming bonds.
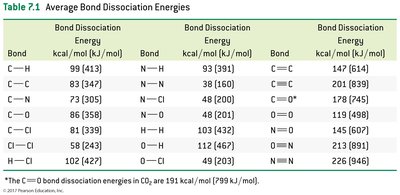
Bond Dissociation Energies Table
This table provides average bond dissociation energies for common bonds, useful for calculating heats of reaction.
7.3 Exothermic and Endothermic Reactions
Reactions can be classified based on whether they release or absorb heat. The sign and magnitude of ΔH indicate the direction and amount of heat transfer.
Exothermic: Products have stronger bonds than reactants; energy is released.
Endothermic: More energy is required to break bonds than is released; energy is absorbed.
ΔH Calculation:


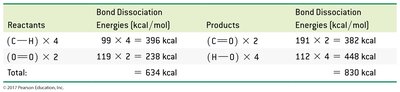

Worked Examples
Combustion of Methane: ,
Calculating Heat Released: Use mole and mass conversions to determine heat released for a given amount of reactant.
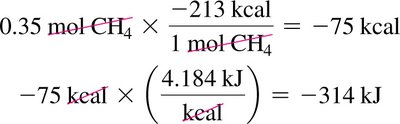
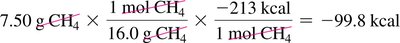
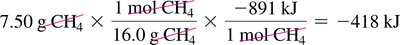
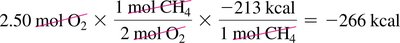
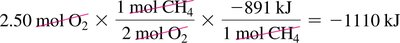
7.4 Why Do Reactions Occur? Free Energy
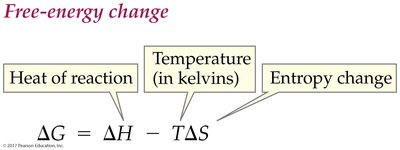
The spontaneity of a reaction depends on enthalpy (ΔH), entropy (ΔS), and free energy (ΔG). Spontaneous processes proceed without external influence.
Spontaneous Process: Proceeds on its own once started.
Nonspontaneous Process: Requires continuous external influence.
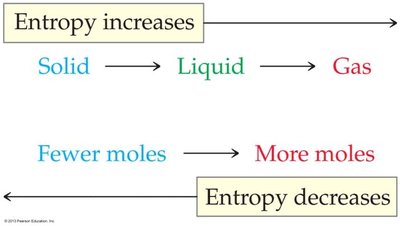
Entropy (S): Measure of molecular disorder; increases with more disorder.
Free Energy Change (ΔG):
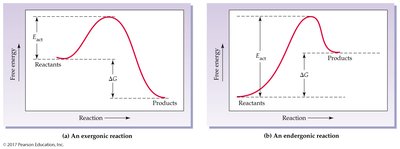
Exergonic: Spontaneous, ΔG negative.
Endergonic: Nonspontaneous, ΔG positive.

7.5 How Do Chemical Reactions Occur? Reaction Rates
Reaction rates depend on the frequency and energy of collisions between reactant molecules. Activation energy is the minimum energy required for a reaction to occur.
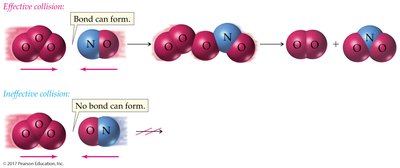
Collision Theory: Reactants must collide with proper orientation and sufficient energy.
Activation Energy (Eact): Energy barrier that must be overcome for a reaction.
Reaction Rate: Speed at which reactants are converted to products.
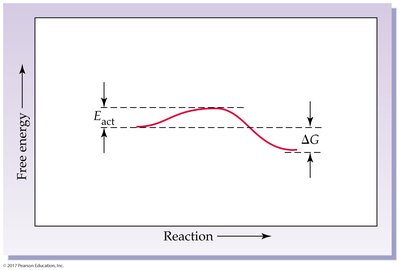
7.6 Effects of Temperature, Concentration, and Catalysts on Reaction Rates
Reaction rates can be increased by raising temperature, increasing concentration, or adding a catalyst.
Temperature: Higher temperature increases collision frequency and energy, speeding up reactions.
Concentration: Higher concentration increases collision frequency, often increasing reaction rate.
Catalyst: Lowers activation energy, providing an alternative pathway for the reaction.


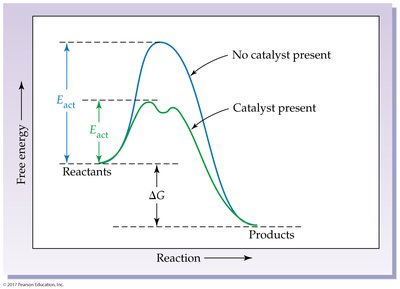

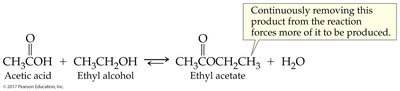
7.7 Reversible Reactions and Chemical Equilibrium
Reversible reactions can proceed in both directions. Chemical equilibrium is reached when the rates of forward and reverse reactions are equal.
Forward Reaction: Reactants to products.
Reverse Reaction: Products to reactants.
Chemical Equilibrium: State where concentrations of reactants and products remain constant.

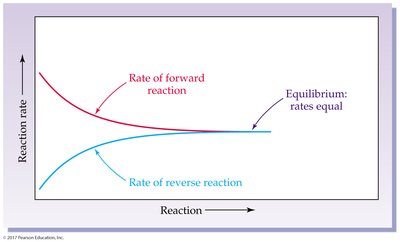
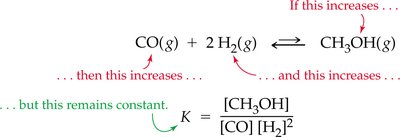
7.8 Equilibrium Equations and Equilibrium Constants
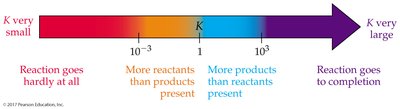
The equilibrium constant (K) quantifies the ratio of product to reactant concentrations at equilibrium. The value of K predicts the extent of a reaction.
Equilibrium Constant (K):
Interpretation: Large K means reaction favors products; small K favors reactants.
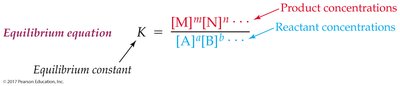
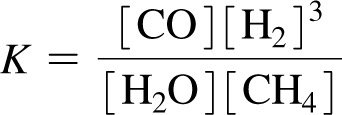
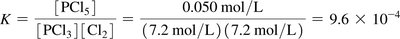
7.9 Le Châtelier’s Principle: The Effect of Changing Conditions on Equilibria
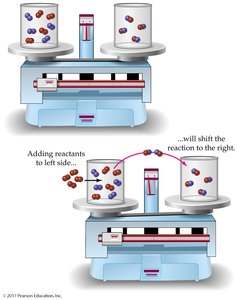
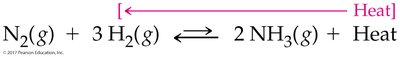
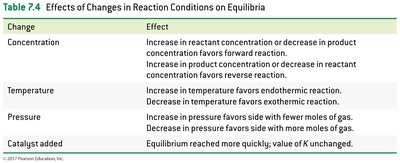
Le Châtelier’s principle states that a system at equilibrium will adjust to relieve any applied stress, such as changes in concentration, temperature, or pressure.
Concentration: Increasing reactant concentration shifts equilibrium to products; increasing product concentration shifts equilibrium to reactants.
Temperature: Increasing temperature favors endothermic direction; decreasing temperature favors exothermic direction.
Pressure: Increasing pressure favors the side with fewer gas molecules.
Catalyst: Speeds up reaching equilibrium but does not affect equilibrium concentrations.
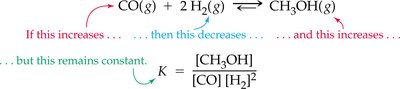

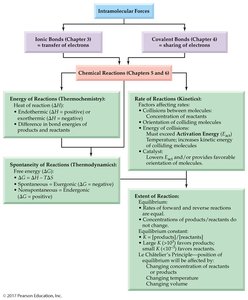
Concept Map
This concept map summarizes the relationships between energy, spontaneity, reaction rates, and equilibrium in chemical reactions.