Back
BackChemical Reactions: Mole and Mass Relationships
Study Guide - Smart Notes

Chapter 6: Chemical Reactions – Mole and Mass Relationships
6.1 The Mole and Avogadro’s Number
The concept of the mole allows chemists to count atoms, molecules, or formula units by weighing them. Avogadro’s number provides the link between the atomic scale and the macroscopic scale.
Atomic Weight: The average mass of an element’s atoms, expressed in atomic mass units (amu).
Molecular Weight (MW): The sum of the atomic weights of all atoms in a molecule (for covalent compounds) or formula unit (for ionic compounds).
Mole (mol): The amount of a substance that contains exactly 6.022 × 1023 elementary entities (Avogadro’s number, NA).
Molar Mass: The mass in grams of 1 mole of a substance, numerically equal to its molecular or formula weight in amu.

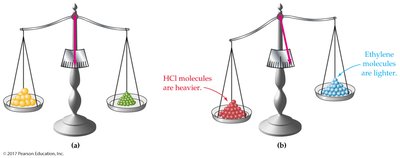
Key Principle: Samples of different substances contain the same number of molecules or formula units when their mass ratio matches their molecular or formula weight ratio.
Avogadro’s Number (NA): 6.022 × 1023 entities per mole.

Visual Example: One mole of different substances (e.g., sulfur, copper, mercury, helium) contains the same number of particles but different masses.

Worked Example: Calculating Molar Mass and Number of Molecules
To find the molar mass, sum the atomic weights of all atoms in the molecular formula.

To convert mass to number of molecules, use the molar mass and Avogadro’s number as conversion factors.
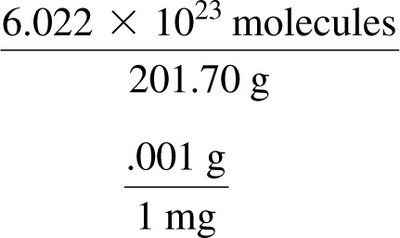

Worked Example: Atoms to Mass Conversion
To convert number of atoms to mass, use the atomic weight and Avogadro’s number.
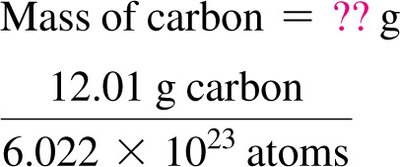
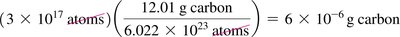
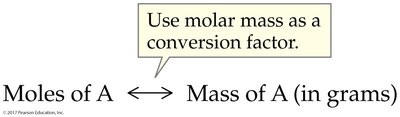
6.2 Gram-Mole Conversions
Molar mass serves as a bridge between the number of moles and the mass of a substance. It is essential for converting between these quantities in chemical calculations.
Key Equation:
Key Equation:
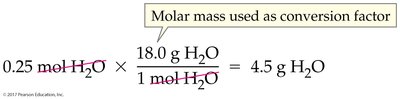
Worked Example: Moles to Mass
Given moles of a substance, multiply by its molar mass to find the mass in grams.

Worked Example: Mass to Moles and Ions
Given mass, convert to moles using molar mass, then use the chemical formula to find moles of specific ions.
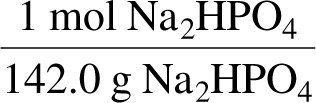

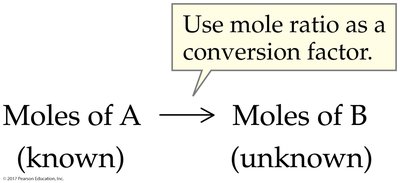
6.3 Mole Relationships and Chemical Equations
Balanced chemical equations provide the mole ratios needed to relate quantities of reactants and products. These ratios are essential for stoichiometric calculations.
Stoichiometry: The calculation of reactants and products in chemical reactions using balanced equations.
Mole Ratio: The ratio of coefficients from a balanced equation, used as a conversion factor.


Worked Example: Using Mole Ratios
Use coefficients from the balanced equation to set up conversion factors between reactants and products.

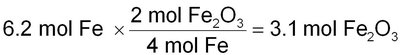
6.4 Mass Relationships and Chemical Equations
Mass relationships in chemical reactions are determined by combining mole ratios from balanced equations with molar masses. Mass-to-mass conversions require several steps.
Types of Conversions:
Mole-to-mole: Use mole ratios from the balanced equation.
Mole-to-mass or mass-to-mole: Use molar mass as a conversion factor.
Mass-to-mass: Convert mass to moles, use mole ratio, then convert back to mass.
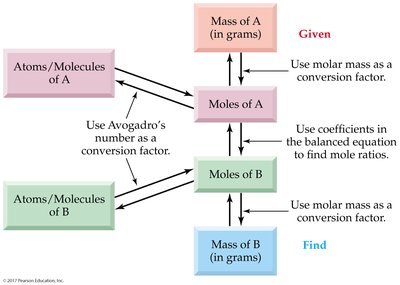
General Steps for Mass Relationships:
Write the balanced chemical equation.
Choose appropriate molar masses and mole ratios.
Set up the factor-label (dimensional analysis) expression.
Calculate and check your answer.
Worked Example: Mass of Product from Moles of Reactant
Convert moles of reactant to moles of product using the mole ratio, then to grams using molar mass.
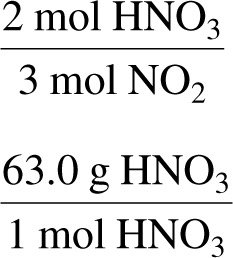
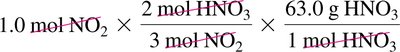
Worked Example: Mass-to-Mass Conversion
Convert mass of product to moles, use mole ratio to find moles of reactant, then convert to mass.
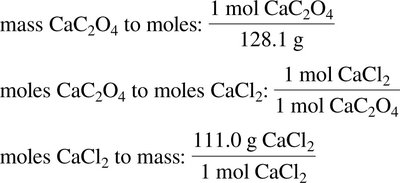
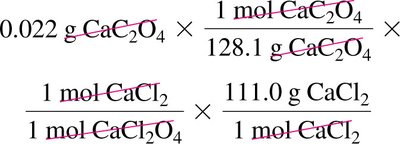
6.5 Limiting Reagent and Percent Yield
In most chemical reactions, one reactant is used up before the others, limiting the amount of product formed. The efficiency of a reaction is measured by percent yield.
Limiting Reagent: The reactant that is completely consumed first, thus limiting the amount of product formed.
Theoretical Yield: The maximum amount of product that can be formed from the limiting reagent.
Actual Yield: The amount of product actually obtained from a reaction.
Percent Yield: The ratio of actual yield to theoretical yield, expressed as a percentage.
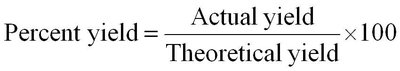
Worked Example: Calculating Percent Yield
Percent yield is calculated as:

Worked Example: Identifying Limiting Reagent and Theoretical Yield
Convert masses of reactants to moles, use mole ratios to determine which reactant produces less product (limiting reagent), then calculate theoretical yield.

Worked Example: Actual Yield from Percent Yield
Calculate theoretical yield, then multiply by percent yield (as a decimal) to find actual yield.

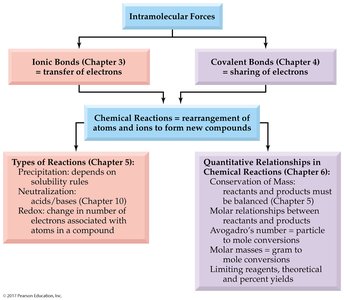
Concept Map: Quantitative Relationships in Chemical Reactions
This concept map summarizes the relationships between atoms, moles, mass, and chemical equations, and the steps for solving stoichiometry problems.