Back
BackClassification and Balancing of Chemical Reactions: Study Notes
Study Guide - Smart Notes

Classification and Balancing of Chemical Reactions
Chemical Equations
Chemical equations are symbolic representations of chemical reactions, showing the substances involved and their relative amounts. They are fundamental to understanding chemical processes and must obey the law of conservation of mass.
Reactants: Substances that undergo change, written on the left side of the arrow.
Products: Substances formed in the reaction, written on the right side of the arrow.
Conditions: Special conditions (e.g., heat, catalysts) are written above the arrow.
Physical States: Indicated by (s) for solid, (l) for liquid, (g) for gas, and (aq) for aqueous solution.
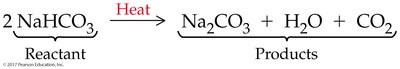
Balancing Chemical Equations
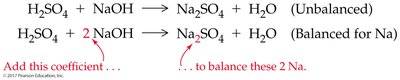
Balancing ensures the same number and type of atoms on both sides of the equation, reflecting the conservation of mass. Coefficients are used to balance equations without altering chemical identities.
Step 1: Write the unbalanced equation with correct formulas.
Step 2: Add coefficients to balance atoms, starting with elements that appear in only one reactant and one product.
Step 3: Check that all atoms are balanced.
Step 4: Ensure coefficients are in the lowest whole-number ratio.
Example: The Haber process for ammonia synthesis:
Example: Combustion of methane:
Example: Decomposition of sodium chlorate:
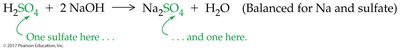
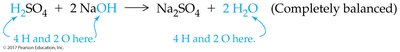
Precipitation Reactions and Solubility Guidelines
Precipitation reactions occur when two aqueous ionic solutions combine to form an insoluble solid (precipitate). The likelihood of precipitation depends on the solubility of the products.
Solubility: The amount of a compound that dissolves in a solvent at a given temperature.
Low solubility leads to precipitation; high solubility means no precipitate forms.

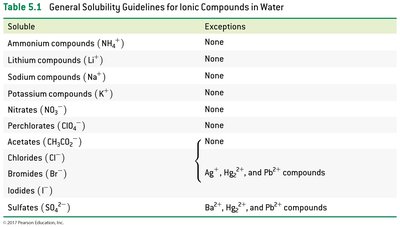
General Solubility Guidelines:
Soluble | Exceptions |
|---|---|
Ammonium (NH4+), Lithium (Li+), Sodium (Na+), Potassium (K+) | None |
Nitrates (NO3-), Perchlorates (ClO4-), Acetates (CH3CO2-) | None |
Chlorides (Cl-), Bromides (Br-), Iodides (I-) | Ag+, Hg22+, Pb2+ compounds |
Sulfates (SO42-) | Ba2+, Hg22+, Pb2+ compounds |
Acids, Bases, and Neutralization Reactions
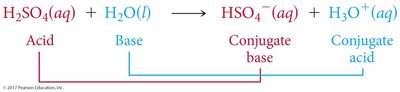
Acid-base neutralization reactions involve an acid reacting with a base to produce water and a salt. These reactions remove H+ and OH- ions from solution.
Acid (HA): Donates H+
Base (MOH): Donates OH-
Salt: Ionic compound formed from the acid and base ions
Example:
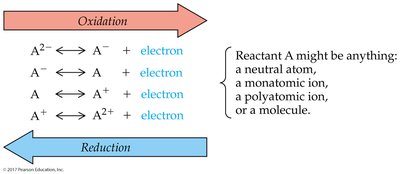
Redox (Oxidation-Reduction) Reactions
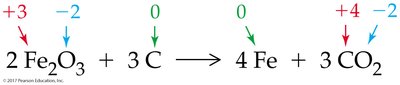
Redox reactions involve the transfer of electrons between atoms, resulting in changes in oxidation numbers. Oxidation is the loss of electrons, while reduction is the gain of electrons.
Oxidizing agent: Gains electrons (is reduced)
Reducing agent: Loses electrons (is oxidized)
Redox reactions are common in corrosion, combustion, respiration, bleaching, and metallurgy.
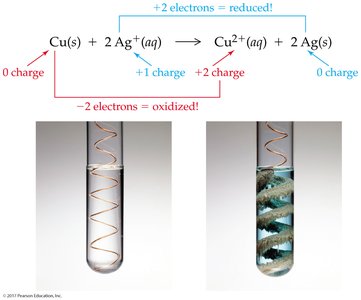
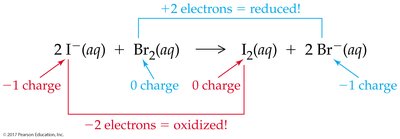
Recognizing Redox Reactions and Assigning Oxidation Numbers
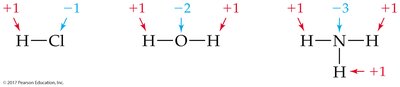
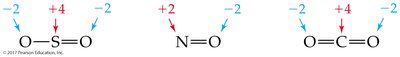
Oxidation numbers help track electron transfer in reactions. Several rules are used to assign oxidation numbers:
Elemental state: 0
Monatomic ion: Equal to its charge
Hydrogen: Usually +1; Oxygen: Usually –2; Halogens: Usually –1
Sum in a neutral compound: 0
Sum in a polyatomic ion: Equal to the ion's charge
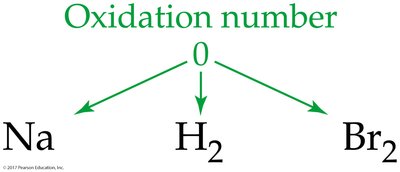
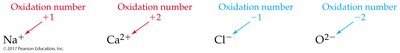
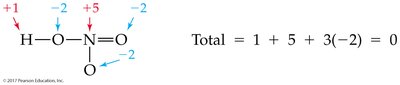
Net Ionic Equations
Net ionic equations show only the ions and molecules directly involved in a chemical reaction, omitting spectator ions. This provides a clearer picture of the actual chemical change.
Ionic equation: All ions are shown explicitly.
Spectator ions: Ions that do not participate in the reaction.
Net ionic equation: Only includes species that undergo change.
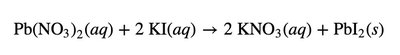
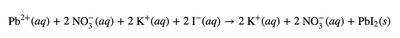
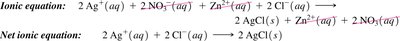
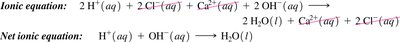
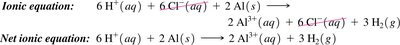
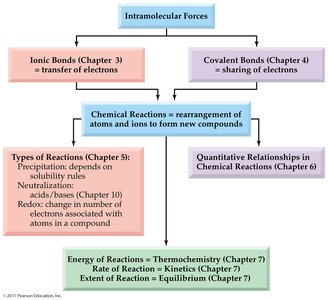
Concept Map: Types of Chemical Reactions
This concept map summarizes the relationships between intramolecular forces, types of chemical bonds, chemical reactions, and the classification of reactions (precipitation, neutralization, redox), as well as their connections to other topics such as thermochemistry, kinetics, and equilibrium.