 Back
BackComposition of Substances and Solutions: Formula Mass, Empirical Formulas, and Solution Concentrations
Study Guide - Smart Notes

Formula Mass and the Mole Concept
Definition and Calculation of Formula Mass
The formula mass of a substance is the sum of the average atomic masses of all the atoms in the chemical formula. For covalent substances, this is also called the molecular mass. The formula mass is calculated as follows:
Formula mass = [(number of atoms of element 1) × (atomic mass of element 1)] + [(number of atoms of element 2) × (atomic mass of element 2)] + ...
The unit for atomic mass is amu (atomic mass unit).
The molar mass (g/mol) is numerically equal to the formula mass (amu), but expressed in grams per mole.
Example: For CO2 (1 C, 2 O):
Formula mass = (1 × 12.01 amu) + (2 × 16.00 amu) = 44.01 amu
Molar mass = 44.01 g/mol
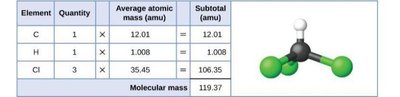
Note: For ionic compounds, the term "formula mass" is used instead of "molecular mass" because they do not exist as discrete molecules.
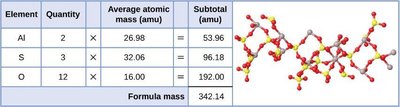
Determining Empirical and Molecular Formulas
Percent Composition
Percent composition (or mass percent) is the percentage by mass of each element in a compound. It is calculated as:
Mass percent of element x = (mass of element x in 1 mol of compound / mass of 1 mol of compound) × 100%
The sum of all elements' percentages in a compound should be 100%.
Example: In a 10.0 g sample containing 2.5 g H and 7.5 g C:
% H = (2.5 g / 10.0 g) × 100 = 25%
% C = (7.5 g / 10.0 g) × 100 = 75%
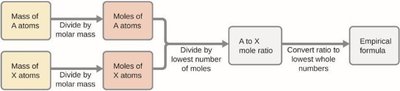
Empirical Formula from Experimental Data
The empirical formula represents the simplest whole-number ratio of atoms in a compound. It can be determined from mass or percent composition data using the following steps:
Convert mass (or percent) of each element to moles using molar mass.
Divide each mole value by the smallest number of moles calculated.
If necessary, multiply ratios by an integer to obtain whole numbers.
Example: A compound contains 1.71 g C and 0.287 g H:
Moles C = 1.71 g / 12.01 g/mol = 0.142 mol
Moles H = 0.287 g / 1.008 g/mol = 0.285 mol
Ratio: C:H = 0.142:0.285 ≈ 1:2 → Empirical formula = CH2
Empirical Formula from Percent Composition
To determine the empirical formula from percent composition:
Assume a 100 g sample (percentages become grams).
Convert grams to moles for each element.
Divide by the smallest number of moles.
Multiply to get whole-number ratios if needed.
Derivation of Molecular Formulas
The molecular formula is a whole-number multiple of the empirical formula. It is determined by:
n = (molecular or molar mass) / (empirical formula mass)
Molecular formula = (empirical formula) × n
Example: Empirical formula CH2O (mass = 30 amu), molecular mass = 180 amu:
n = 180 / 30 = 6
Molecular formula = C6H12O6
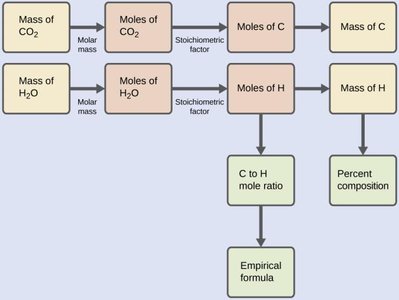
Combustion Analysis
Combustion analysis is a gravimetric method used to determine the elemental composition of hydrocarbons and related compounds. The sample is combusted in oxygen, producing CO2 and H2O, which are absorbed and weighed to determine the amount of C and H in the original sample.
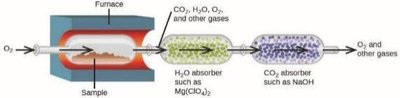
The process involves:
Weighing the sample and combusting it in excess O2
Absorbing H2O and CO2 in separate traps
Calculating the moles of C and H from the masses of CO2 and H2O produced
Solutions and Their Composition
Definition of Solutions
A solution is a homogeneous mixture of two or more substances. The solvent is the component present in the greatest amount, and the solute is present in a lesser amount. If water is the solvent, the solution is called aqueous.

Concentration of Solutions
Concentration describes the amount of solute per unit volume of solution. Solutions can be described as dilute (small amount of solute) or concentrated (large amount of solute).
Molarity (M)
Molarity (M) is the number of moles of solute per liter of solution:
Used as a conversion factor between moles and volume.
Example: A 2.0 M sugar solution contains 2.0 moles of sugar per 1 L of solution.
Dilution of Solutions
Dilution is the process of decreasing the concentration of a solution by adding more solvent. The amount of solute remains constant:
Where and are the initial molarity and volume, and and are the final molarity and volume after dilution.
Other Units for Solution Concentrations
Mass Percentage
Mass percentage is the ratio of the mass of a component to the total mass of the solution, multiplied by 100:
Mass percentage =

Example: Liquid bleach contains 7.4% NaOCl by mass.
Volume Percentage
Volume percentage is used when both solute and solvent are liquids:
Volume percentage =
Example: Rubbing alcohol is often sold as a 70% vol aqueous solution.
Mass-Volume Percentage
Mass-volume percent is the mass of solute per volume of solution, expressed as a percentage:
For example, physiological saline is 0.9% m/v, meaning 0.9 g solute per 100 mL solution.
Parts per Million (ppm) and Parts per Billion (ppb)
Very low concentrations are often expressed as ppm or ppb:
ppm =
ppb =
Example: The EPA requires action if lead in tap water reaches 15 ppb.
