 Back
BackCovalent Bonding, Bond Energies, and Bond Polarity: Study Notes
Study Guide - Smart Notes

Chapter 7: Covalent Bonding and Electron-Dot Structures
Introduction to Covalent Bonding
Covalent bonding occurs when two nonmetal atoms share electrons to achieve stable electron configurations. This type of bonding is characterized by high effective nuclear charge (Zeff), high ionization energy (IE), and high (negative) electron affinity (EA), resulting in tightly held outer electrons. Each covalent bond consists of a localized electron pair shared between two atoms, with attractive forces between nuclei and electrons and repulsive forces between electron pairs and between nuclei.
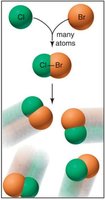
Bond Order and Electron Pairs
The number of valence electron pairs shared by bonded atoms determines the bond order:
Single bond: 1 shared electron pair (bond order = 1)
Double bond: 2 shared electron pairs (bond order = 2)
Triple bond: 3 shared electron pairs (bond order = 3)
Lone pair: A nonbonding or unshared electron pair
Bond order affects both bond length and bond energy.
Bond Length and Bond Energy
Bond length is the most stable internuclear distance, representing the balance between attractive and repulsive forces. The lowest point in the potential energy curve corresponds to the bond length. Bond energy is the energy required to break a covalent bond and is a measure of bond strength. It is always a positive value.
Smaller atoms: Shorter bond length, higher bond energy
Larger atoms: Longer bond length, lower bond energy
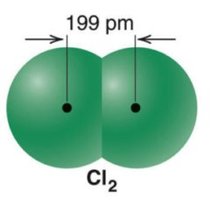
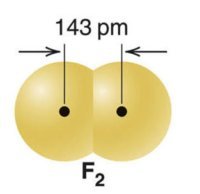
Bond order also influences bond length and energy: higher bond order results in shorter bond length and higher bond energy.
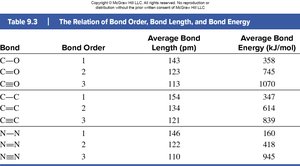
Bond | Bond Order | Average Bond Length (pm) | Average Bond Energy (kJ/mol) |
|---|---|---|---|
C=O | 1 | 143 | 358 |
C=O | 2 | 123 | 745 |
C=O | 3 | 113 | 1070 |
C–C | 1 | 154 | 347 |
C=C | 2 | 134 | 614 |
C≡C | 3 | 121 | 839 |
N–N | 1 | 146 | 160 |
N=N | 2 | 122 | 418 |
N≡N | 3 | 110 | 945 |
Bond Energy and Chemical Reactions
During a chemical reaction, bonds in reactants are broken (endothermic process) and new bonds are formed in products (exothermic process). The overall enthalpy change of reaction () can be calculated using bond energies:
If more energy is released in forming bonds than is required to break them, the reaction is exothermic (negative ). If more energy is required to break bonds than is released, the reaction is endothermic (positive $\Delta H_{\text{rxn}}$).
Infrared (IR) Spectroscopy and Molecular Vibrations
IR spectroscopy is a tool used to study covalently bonded substances. Molecular vibrations absorb energy in the IR region, and the energy absorbed depends on bond strength, type of vibrational motion, and atomic masses. Triatomic and diatomic molecules exhibit different types of vibrational motions, such as stretching (symmetric and asymmetric) and bending.
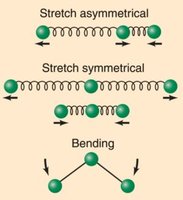
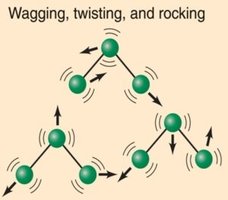
IR spectra display peaks corresponding to different bond vibrations, which can be used to identify functional groups in molecules.

Physical Properties of Covalent Compounds
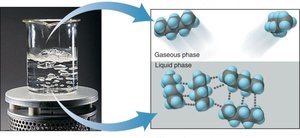
Intermolecular Forces and States of Matter
Covalent compounds typically have weak intermolecular forces, resulting in low boiling and melting points. They are often liquids or gases at room temperature, and only a small amount of energy is required to overcome these forces during phase changes.
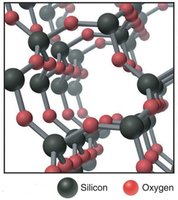
Network Covalent Structures
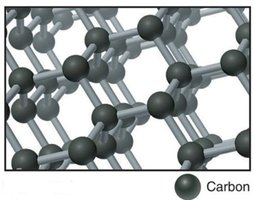
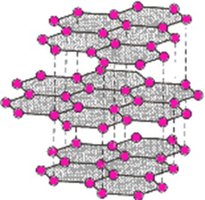
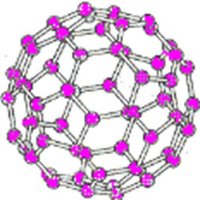
Some covalent compounds form strong three-dimensional lattices, known as network covalent structures. These have very high melting points and distinct physical properties compared to typical covalent compounds. Examples include diamond (carbon), quartz (silicon dioxide), graphite, and fullerenes (buckyballs and nanotubes).
Diamond: Strong 3D structure, high melting point, electrical insulator
Quartz: Strong 3D structure, high melting point, electrical insulator
Graphite: Strong 2D layers, high melting point, electrically conductive
Fullerenes: Weaker covalent structure, lower melting point
Electrical Conductivity
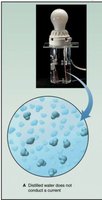
Covalent compounds generally do not conduct electricity because they lack free ions or electrons. In contrast, ionic compounds conduct electricity when dissolved in water or melted, due to the presence of mobile ions.
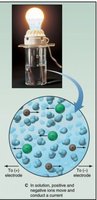
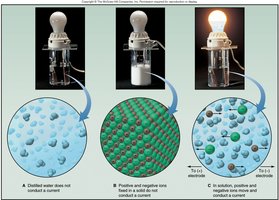
Bond Polarity and Electronegativity
Electronegativity (EN)
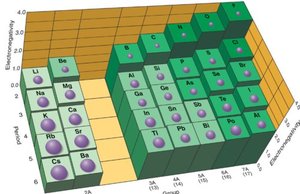
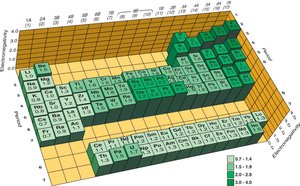
Electronegativity is the ability of an atom to attract electrons to itself when sharing them in a covalent bond. It is quantified by the Pauling scale, with fluorine assigned the highest value (4.0). Electronegativity is not the same as electron affinity, which is the energy change when an electron is added to a gaseous atom.
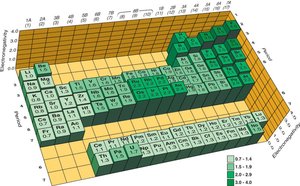
Electronegativity increases across a period and decreases down a group. It is directly related to effective nuclear charge (Zeff) and inversely related to atomic size.
Bond Polarity and Dipole Moments
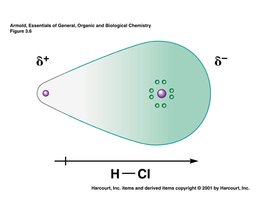
Bond polarity arises when two atoms with different electronegativities share electrons unequally. The greater the difference in electronegativity (ΔEN), the more polar the bond. A dipole arrow points toward the more electronegative atom, indicating the direction of electron density.
Nonpolar covalent bond: Equal sharing of electrons (ΔEN = 0)
Polar covalent bond: Unequal sharing of electrons (0 < ΔEN < 1.7)
Ionic bond: Electron transfer (ΔEN > 1.7)
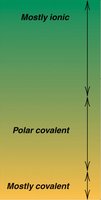
ΔEN | Ionic Character |
|---|---|
> 1.7 | Mostly ionic |
0.4 – 1.7 | Polar covalent |
< 0.4 | Mostly covalent |
0 | Nonpolar covalent |
Examples and Applications
To determine bond polarity, calculate the difference in electronegativity between the two atoms. For example, the bond between phosphorus and oxygen (ΔEN = 1.4) is more polar than the bond between carbon and hydrogen (ΔEN = 0.4).
Classify the bond character of compounds based on ΔEN:
Compound | ΔEN | Bond Character |
|---|---|---|
KCl | 2.2 | Ionic |
P4 | 0 | Nonpolar covalent |
SO2 | 1.0 | Polar covalent |
Br2 | 0 | Nonpolar covalent |
NO2 | 0.5 | Polar covalent |

Properties of Period 3 Chlorides
The physical properties of chlorides in Period 3 vary with their bond character. Ionic chlorides (e.g., NaCl, MgCl2) have high melting points and conduct electricity when molten. Covalent chlorides (e.g., SiCl4, PCl3, S2Cl2, Cl2) have lower melting points and do not conduct electricity.
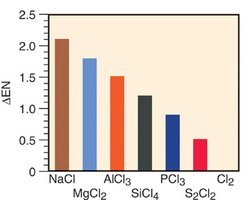

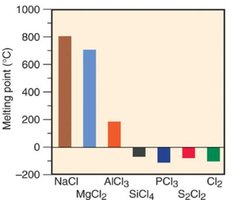
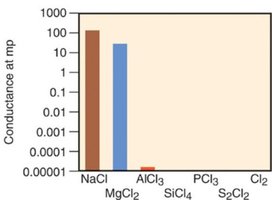
Additional info: The classification of bonds as ionic, polar covalent, or nonpolar covalent is a continuum, and the properties of compounds reflect the nature of their bonding. Understanding bond polarity is essential for predicting molecular properties such as solubility, melting point, and conductivity.
