 Back
BackElectronic Configurations and Periodic Trends: Study Notes for General Chemistry
Study Guide - Smart Notes

The Periodic Table and Electronic Configurations
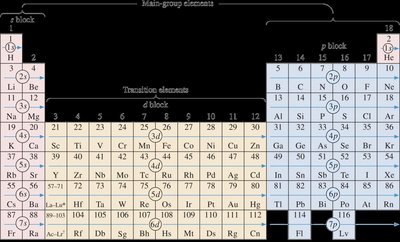
Overview of the Periodic Table
The periodic table is a systematic arrangement of elements based on their atomic number, electron configurations, and recurring chemical properties. It is divided into blocks (s, p, d, f) corresponding to the subshells being filled by electrons.
Main-group elements are found in the s and p blocks.
Transition elements occupy the d block.
Lanthanides and actinides are in the f block.

Rules for Assigning Electrons to Orbitals
Principles Governing Electron Configuration
Electron configuration describes the distribution of electrons in atomic orbitals. Three fundamental rules guide this process:
Aufbau Principle: Electrons fill orbitals in order of increasing energy, starting from the lowest energy level.
Pauli Exclusion Principle: No two electrons in an atom can have the same set of four quantum numbers. Each orbital can hold a maximum of two electrons with opposite spins.
Hund’s Rule: Electrons occupy degenerate (equal energy) orbitals singly before pairing, maximizing the number of unpaired electrons with parallel spins.
Order of Filling Electronic Subshells
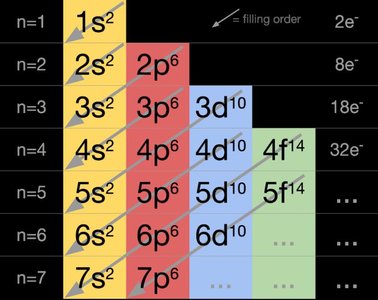
The order in which subshells are filled is determined by their energy levels. The filling sequence can be visualized using the diagonal rule.
Filling order: 1s → 2s → 2p → 3s → 3p → 4s → 3d → 4p → 5s → 4d → 5p → 6s → 4f → 5d → 6p → 7s → 5f → 6d → 7p
Maximum electrons per subshell: s (2), p (6), d (10), f (14)
Electronic Configurations: Notation and Diagrams
spdf Notation and Electron-in-Box Diagrams
Electronic configurations can be represented in different ways:
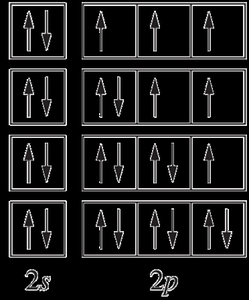
spdf notation: Shows the number of electrons in each subshell, e.g., 1s22s22p2.
Electron-in-box diagrams: Visualize electron spins and orbital occupancy.
For example, the electron configuration of carbon (Z=6) is:
spdf notation: 1s22s22p2
Electron-in-box diagram:
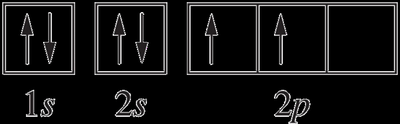
Electrons in singly occupied orbitals of the same subshell have parallel spins, which is the lowest energy (ground-state) configuration.
Expanded Electron Configurations
Expanded notation specifies the distribution of electrons among individual orbitals, e.g., 1s22s22px12py1.
Electron Configurations for Elements
Examples for elements N, O, F, Ne (Z = 7–10):
N: [He]2s22p3
O: [He]2s22p4
F: [He]2s22p5
Ne: [He]2s22p6
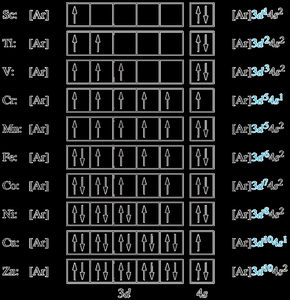
For transition elements (Z = 21–30), electrons fill the 3d orbitals. Exceptions include Cr ([Ar]3d54s1) and Cu ([Ar]3d104s1).
Periodic Trends
Introduction to Periodic Trends
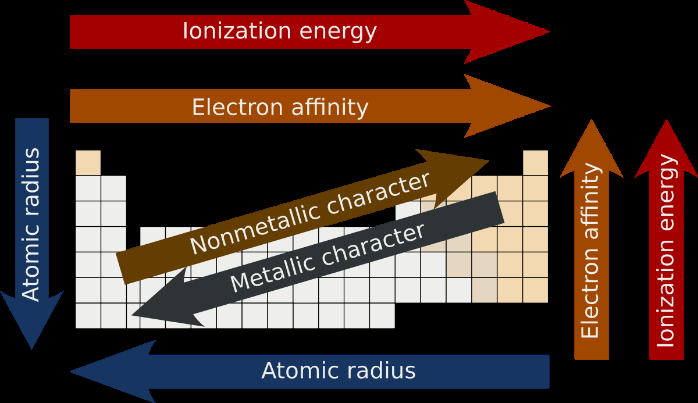
Periodic trends are patterns observed in the properties of elements across periods and groups in the periodic table. These include:
Electronegativity
Ionisation energy
Electron affinity
Atomic radius
Ionic radius
Melting point
Metallic character
Ionisation Energy
Ionisation energy (I.E.) is the energy required to remove an electron from a gaseous atom. Successive ionisation energies increase as electrons are removed, especially after removing all valence electrons.
Sharp rises in I.E. occur when removing electrons from a new shell closer to the nucleus.
Example: Sodium shows sharp increases after the first and tenth electrons are removed.
General equation:
Electron Affinity
Electron affinity (E.A.) is the energy change when an electron is added to a gaseous atom. A more negative E.A. indicates a greater tendency to gain electrons.
First E.A. is usually negative (energy released).
Second E.A. is positive due to repulsion (energy required).
Example equations:
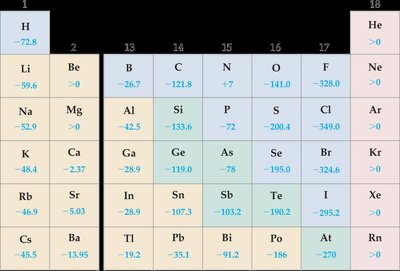
E.A. increases across a period and decreases down a group. Smaller atoms to the right of the periodic table have more negative E.A.
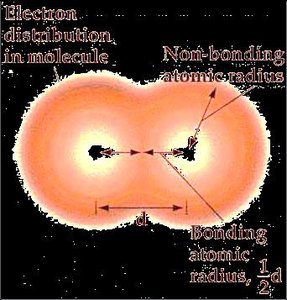
Atomic Radius
The atomic radius is a measure of the size of an atom. It can be defined as:
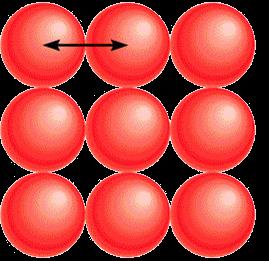
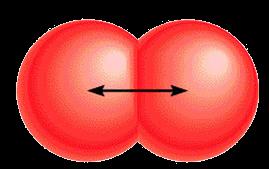
Metallic radius: Half the distance between nuclei of adjacent atoms in a metallic lattice.
Covalent radius: Half the distance between nuclei of identical atoms bonded together.
Ionic Radius
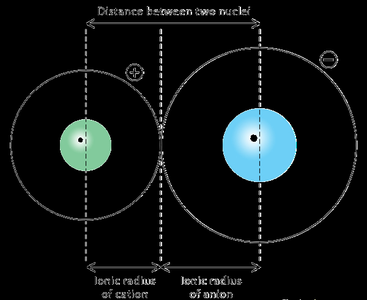
Ionic radius refers to the size of an ion (cation or anion). It is related to the distance between neighboring cations and anions in a crystal lattice.
Cations are smaller than their parent atoms due to loss of electrons.
Anions are larger than their parent atoms due to gain of electrons.
Example:
Mg: 1s22s22p63s2 → Mg2+: 1s22s22p6
F: 1s22s22p5 → F-: 1s22s22p6
Summary Table: Periodic Trends
The following table summarizes the main periodic trends:
Trend | Across Period | Down Group |
|---|---|---|
Atomic Radius | Decreases | Increases |
Ionisation Energy | Increases | Decreases |
Electron Affinity | Becomes more negative | Becomes less negative |
Metallic Character | Decreases | Increases |
Nonmetallic Character | Increases | Decreases |
Practice Problems
Electronic Configurations
Write the electronic configurations for P, Fe, Ag, Ho, Os.
Represent configurations in spdf notation and electron-in-box diagrams.
Periodic Trends
Arrange As, Sn, Br, Sr in order of increasing first ionisation energy.
Arrange K+, Cl-, S2-, Ca2+ in order of increasing ionic size.
Additional info: These notes expand on brief points from the original materials, providing definitions, examples, and visual aids for clarity and completeness.
