 Back
BackElectronic Structure of Atoms: The Bohr Model and Quantum Mechanics
Study Guide - Smart Notes

Electronic Structure of Atoms
The Bohr Atom
The Bohr model, developed by Niels Bohr in 1912-1913, describes electrons as existing in specific, quantized orbits around the nucleus. These orbits correspond to discrete energy levels, and electrons cannot exist between these levels. The energy of an electron in an atom is therefore quantized, meaning it can only have certain allowed values.
Quantized Energy Levels: Electrons revolve in fixed orbits at set distances from the nucleus, similar to planets around the sun.
Ground State and Excited State: The lowest energy state is called the ground state. Any higher energy state is an excited state.
Energy Transitions: Electrons can move between energy levels by absorbing or emitting energy in the form of photons.
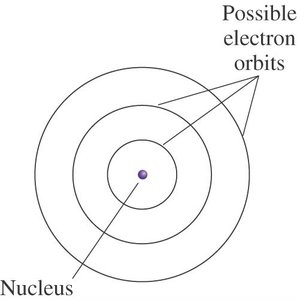
Bohr’s Model of the Atom (1913)
Bohr’s model explains the emission and absorption of light by hydrogen atoms. When an electron falls from a higher energy orbit (e.g., n = 3) to a lower one (e.g., n = 2), a photon is emitted. The energy of the photon corresponds to the difference in energy between the two orbits.
Photon Emission: The energy of the emitted photon is given by , where is Planck’s constant and is the frequency of the photon.
Energy Levels: The energy of an electron in the nth orbit is , where is the Rydberg constant ( J).
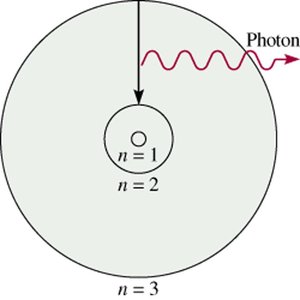
Quantization of Energy and Electron Transitions
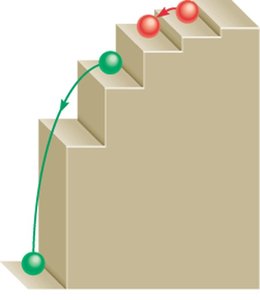
Electrons can only occupy certain energy levels, and transitions between these levels involve discrete energy changes. The quantized nature of these transitions can be visualized as steps, not a continuous ramp.
Energy Absorption and Emission: A positive means energy is absorbed (electron moves to a higher level), while a negative means energy is released (electron falls to a lower level).
Photon Wavelength: The wavelength of the emitted or absorbed photon can be calculated using .
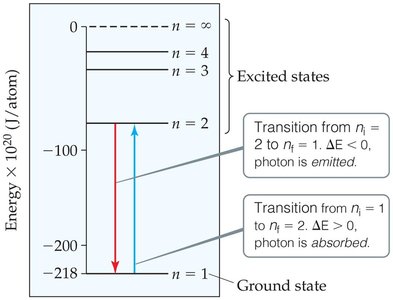
Energy Level Diagrams and Calculations
Energy level diagrams help visualize the transitions between different energy states. The energy difference between levels determines whether a photon is emitted or absorbed.
Ground State: The lowest energy level ().
Excited States: Higher energy levels ().
Transition Direction: Downward transitions emit photons; upward transitions absorb photons.
Mathematical Description of Energy Transitions
The energy change associated with an electron transition between two energy levels in a hydrogen atom is given by:
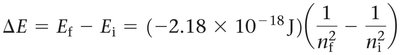
Where and are the initial and final principal quantum numbers, respectively.
Limitations of the Bohr Model
Bohr’s model accurately predicts the spectral lines of hydrogen but fails for atoms with more than one electron.
It does not explain why electron energies are quantized or why electrons are restricted to certain orbits.
The Dual Nature of the Electron
Louis de Broglie proposed that particles such as electrons have wave-like properties. The wavelength () of a particle is given by:
Wave-Particle Duality: Light can behave as both a wave and a particle; electrons exhibit similar duality.
Significance: Wave properties are significant only for submicroscopic particles due to the small value of Planck’s constant ().
The Heisenberg Uncertainty Principle
Werner Heisenberg formulated the uncertainty principle, which states that it is impossible to know both the position and momentum of a particle with absolute precision:
Implication: The more precisely we know a particle’s momentum, the less precisely we can know its position, and vice versa.
Quantum Mechanics and the Schrödinger Equation
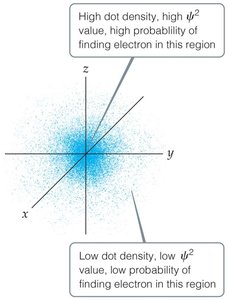
Erwin Schrödinger developed quantum mechanics, a mathematical framework that incorporates both the wave and particle nature of matter. The solutions to the Schrödinger equation are called wave functions (), and the square of the wave function () gives the probability density of finding an electron in a particular region of space.
Electron Density: Regions with high values have a high probability of containing an electron.
Quantum Numbers and Atomic Orbitals
Quantum Numbers
Quantum numbers describe the properties of atomic orbitals and the electrons within them. Each orbital is defined by a set of three quantum numbers:
Principal Quantum Number (): Indicates the energy level and size of the orbital ().
Angular Momentum Quantum Number (): Defines the shape of the orbital ( to ).
Magnetic Quantum Number (): Describes the orientation of the orbital in space ( to ).
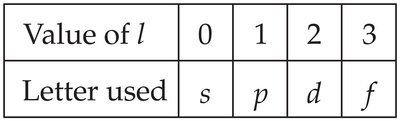
The values of are designated by letters:
Value of l | 0 | 1 | 2 | 3 |
|---|---|---|---|---|
Letter used | s | p | d | f |
Relationship Among Quantum Numbers
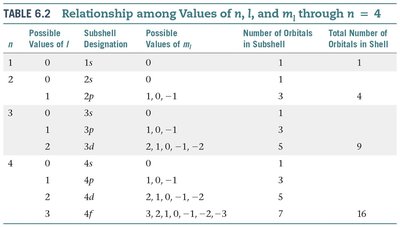
The possible combinations of , , and determine the number and types of orbitals in each energy level. The table below summarizes these relationships for up to 4:
n | Possible Values of l | Subshell Designation | Possible Values of ml | Number of Orbitals in Subshell | Total Number of Orbitals in Shell |
|---|---|---|---|---|---|
1 | 0 | 1s | 0 | 1 | 1 |
2 | 0 | 2s | 0 | 1 | 4 |
2 | 1 | 2p | -1, 0, 1 | 3 | |
3 | 0 | 3s | 0 | 1 | 9 |
3 | 1 | 3p | -1, 0, 1 | 3 | |
3 | 2 | 3d | -2, -1, 0, 1, 2 | 5 | |
4 | 0 | 4s | 0 | 1 | 16 |
4 | 1 | 4p | -1, 0, 1 | 3 | |
4 | 2 | 4d | -2, -1, 0, 1, 2 | 5 | |
4 | 3 | 4f | -3, -2, -1, 0, 1, 2, 3 | 7 |
Atomic Orbitals: Shapes and Properties
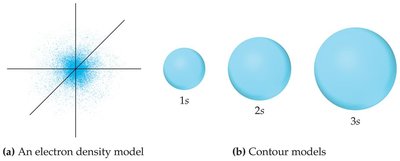
s Orbitals
s orbitals () are spherical in shape. The size of the sphere increases with the principal quantum number .
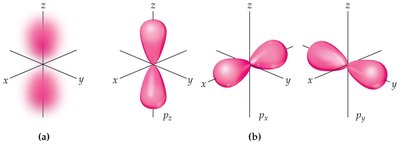
p Orbitals
p orbitals () have a dumbbell shape with two lobes and a node at the nucleus. There are three p orbitals (px, py, pz) in each energy level where .
d and f Orbitals
d orbitals () have more complex shapes, typically with four lobes. There are five d orbitals in each energy level where . f orbitals () are even more complex, with seven equivalent orbitals in each sublevel for .
Additional info: The shapes of f orbitals are not shown here, but they are important in the chemistry of transition and inner transition elements.
