 Back
BackEstimating Reaction Enthalpy Using Bond Energies
Study Guide - Smart Notes

Ch. 5 – Thermochemistry
Enthalpies from Bond Energies
Understanding the energetics of chemical reactions is essential in general chemistry. One useful technique for estimating the enthalpy change of a reaction is to use average bond enthalpies, which quantify the energy required to break or form chemical bonds. This approach allows us to relate the breaking and forming of specific bonds to the overall enthalpy of a reaction.
Bond Breaking and Bond Formation
Bond breaking is an endothermic process—energy must be supplied to break chemical bonds.
Bond formation is an exothermic process—energy is released when new bonds are formed.
By identifying which bonds are broken and which are formed in a reaction, we can estimate the reaction’s enthalpy change quantitatively.
Calculating Reaction Enthalpy from Bond Energies
To estimate the enthalpy change of a reaction using bond energies, follow these steps:
List all bonds broken in the reactants and all bonds formed in the products.
Sum the average bond enthalpies for all bonds broken (energy input).
Sum the average bond enthalpies for all bonds formed (energy released).
Apply the following equation:
A positive value for indicates an endothermic reaction, while a negative value indicates an exothermic reaction.
Using Tabulated Bond Energies
Average bond enthalpies for many common bonds are tabulated and can be used for these calculations. These values are typically given in units of kJ/mol and represent averages over many compounds.
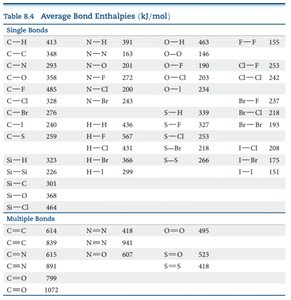
Example Calculation
Suppose you are asked to estimate the enthalpy change for a reaction. The steps are:
Write the balanced chemical equation.
Draw Lewis structures for all reactants and products to identify all bonds present.
Count the number of each type of bond broken and formed (be careful and systematic!).
Look up the average bond enthalpies for each bond type in the provided table.
Apply the equation above to calculate .
Example: If a reaction breaks two C–H bonds and forms one C=O bond, use the table to find the bond enthalpies and substitute into the equation.
Important Notes
Bond enthalpy values are averages and may not be exact for every molecule.
This method provides an estimate, not an exact value, for reaction enthalpy.
Careful accounting of all bonds broken and formed is crucial for accuracy.
Additional info: In practice, this method is especially useful for reactions where standard enthalpies of formation are not available, or for gaining insight into the energetic favorability of proposed reaction mechanisms.
