 Back
BackExam 2 Review: Compounds, Lewis Structures, VSEPR, and Bonding (CHEM 1210)
Study Guide - Smart Notes

Compounds and Chemical Formulas
Ionic vs. Covalent Compounds
Compounds are classified as either ionic or covalent based on the nature of the bonding between their constituent atoms.
Ionic Compounds: Formed between a metal and a nonmetal. Electrons are transferred from the metal to the nonmetal, resulting in the formation of ions that attract each other via electrostatic forces.
Covalent (Molecular) Compounds: Formed between two nonmetals. Electrons are shared between atoms, creating a bond.
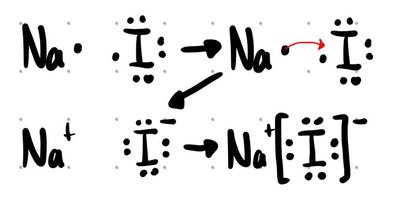
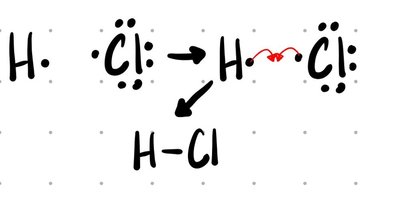
Formulas: Empirical vs. Molecular
Chemical formulas represent the composition of compounds.
Empirical Formula: The simplest whole-number ratio of atoms in a compound (e.g., NO2).
Molecular Formula: The actual number of each atom in a molecule (e.g., N2O4).
Polyatomic Ions
Polyatomic ions are charged species composed of multiple atoms covalently bonded. It is important to distinguish between ammonia (NH3) and ammonium (NH4+).
Naming Compounds
Salts (Ionic Compounds): Name the positive ion first, then the anion (add "-ide" suffix to the anion name). Example: NaCl is Sodium Chloride.
Covalent Compounds: Use prefixes (mono-, di-, tri-, tetra-) to indicate the number of each element. Example: H2O is Dihydrogen monoxide.
Transition Metals: Include the charge in parentheses after the metal's name. Example: AgHCO3 is Silver (I) Bicarbonate.
Lewis Structures and VSEPR
Formal Charge
Formal charge helps determine the most stable Lewis structure for a molecule.
Formula:
Zero formal charge is most favorable; negative formal charge should be on the most electronegative atom.
Steps to Draw a Lewis Structure
Calculate the total number of valence electrons (add for anions, subtract for cations).
Draw connectivity (first atom listed is usually central).
Distribute electrons to the most electronegative atoms first.
Fill octets (add double/triple bonds as needed; note exceptions for expanded or less-than octets).
Minimize formal charges (add bonds to reduce charges).
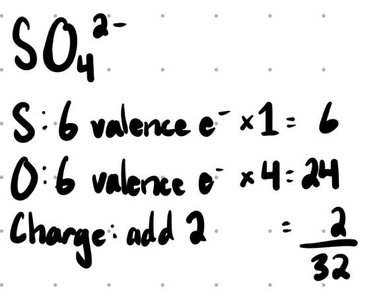
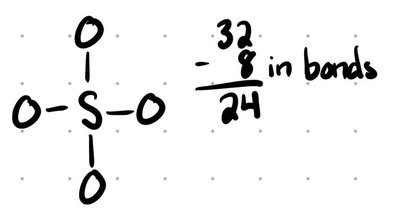
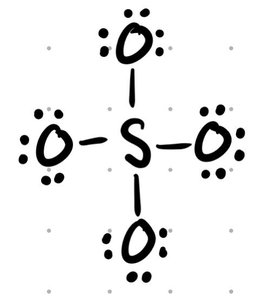
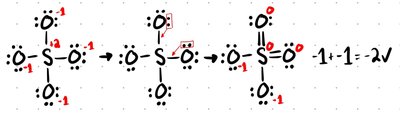
Resonance Structures
Resonance structures are multiple valid Lewis structures for a molecule, differing only in electron arrangement. The actual molecule is a hybrid of all resonance forms.
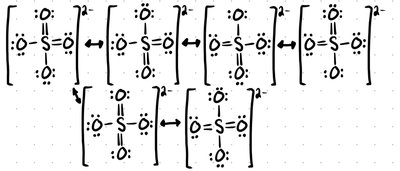
Bond Order
Bond order indicates the number of bonds between two atoms. For resonance structures, add the bond orders and divide by the number of A-B bonds.
Formula:
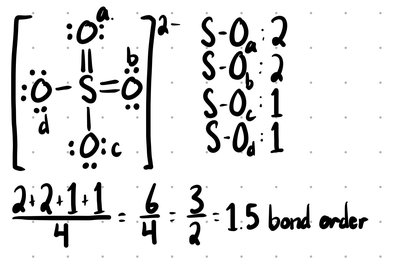
Lewis Structure Practice
Examples of Lewis structures, resonance, formal charges, and bond order:
SeO2: Bond order = 2
ClO-: Bond order = 1
NO2-: Bond order = 1.5
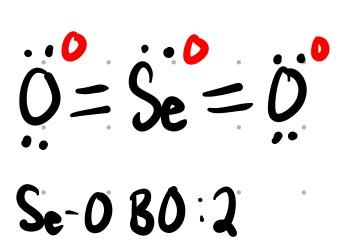
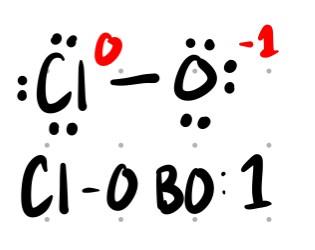

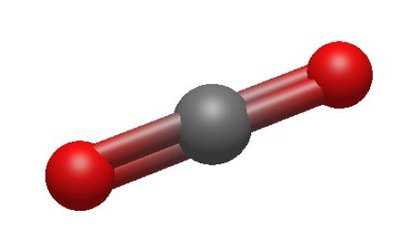
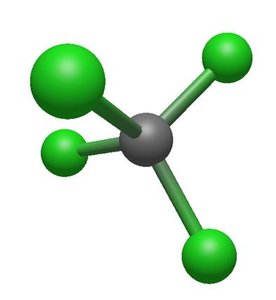
VSEPR Theory
Valence Shell Electron Pair Repulsion (VSEPR) Theory predicts the 3D arrangement of atoms in a molecule based on repulsion between electron groups.
Common geometries: Linear, Trigonal Planar, Tetrahedral, Trigonal Pyramidal, Bent, Octahedral
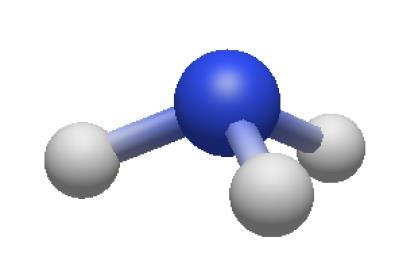
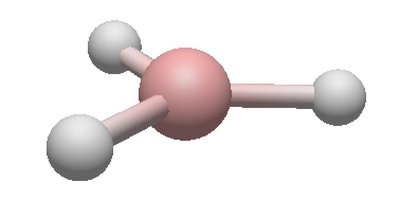
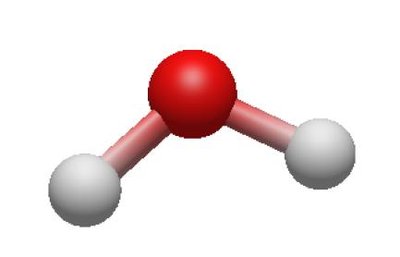
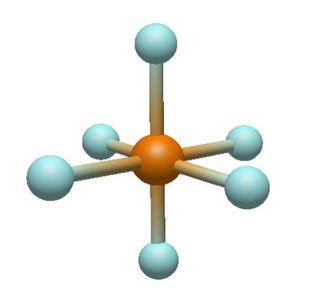
Electron and Molecular Geometries Table
The following table summarizes electron groups, bonding groups, lone pairs, geometries, bond angles, and examples:
Electron Groups | Bonding Groups | Lone Pairs | Electron Geometry | Molecular Geometry | Approximate Bond Angles | Example |
|---|---|---|---|---|---|---|
2 | 2 | 0 | Linear | Linear | 180° | CO2 |
3 | 3 | 0 | Trigonal planar | Trigonal planar | 120° | BF3 |
3 | 2 | 1 | Trigonal planar | Bent | <120° | SO2 |
4 | 4 | 0 | Tetrahedral | Tetrahedral | 109.5° | CH4 |
4 | 3 | 1 | Tetrahedral | Trigonal pyramidal | <109.5° | NH3 |
4 | 2 | 2 | Tetrahedral | Bent | <109.5° | H2O |
Bond Polarity and Electronegativity
Bond Polarity
Bond polarity arises from differences in electronegativity between atoms.
Non-Polar: Electrons are shared equally (e.g., H-H).
Polar: Electrons are shared unequally, shifted toward the more electronegative atom (e.g., H-Cl).
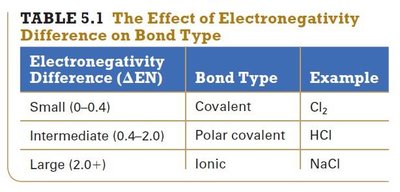
Electronegativity Difference and Bond Type
The type of bond is determined by the difference in electronegativity (ΔEN) between atoms.
Electronegativity Difference (ΔEN) | Bond Type | Example |
|---|---|---|
Small (0–0.4) | Covalent | Cl2 |
Intermediate (0.4–2.0) | Polar covalent | HCl |
Large (2.0+) | Ionic | NaCl |

Determining Overall Polarity
To determine if a molecule is polar or nonpolar:
Symmetry Method: If the molecule is completely symmetric, it is nonpolar.
Polar Bonds Method: Draw arrows for bond polarity and see if they cancel out.
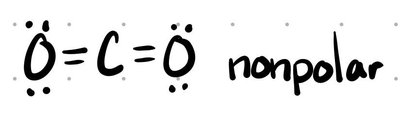

Valence Bond and Molecular Orbital Theory
Valence Bond Theory
Atomic orbitals (s, p, d, f) overlap to form hybrid orbitals, which then overlap to create bonds. The number of hybrid orbitals formed equals the number of atomic orbitals combined.
Example: One s and two p orbitals combine to form three sp2 orbitals.
Hybridization can be predicted using Lewis structures and VSEPR theory.
Bond Types: Sigma and Pi Bonds
Sigma (σ) Bonds: Formed by direct overlap of atomic orbitals; first bond in double/triple bonds.
Pi (π) Bonds: Formed by side-by-side overlap of p orbitals; second bond in double bonds, second and third in triple bonds.
Molecular Orbital (MO) Theory
Atomic orbitals combine to form molecular orbitals. Bonding orbitals strengthen bonds, while antibonding orbitals weaken them.
Paramagnetic: Molecules with unpaired electrons.
Diamagnetic: Molecules with all electrons paired.
Bond Order Formula:
Practice: Lewis Symbols for Ionic Compounds
Lewis symbols represent ions in ionic compounds, showing the transfer of electrons and resulting charges.
Compound | Lewis Symbol |
|---|---|
SrO | Sr2+ [O]2− |
Li2S | Li+ [S]2− Li+ |
CaI2 | [I]− Ca2+ [I]− |
RbF | Rb+ [F]− |
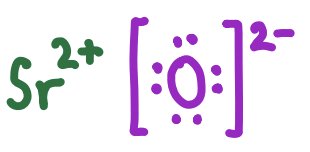
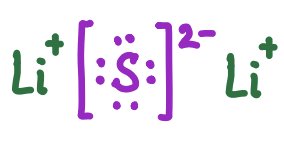
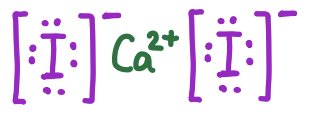
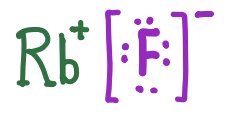
Summary and Study Tips
Review all concepts, especially Lewis structures, resonance, formal charge, VSEPR, and bond polarity.
Practice naming compounds and drawing molecular geometries.
Use provided equation sheets and periodic tables during exams.
Get adequate rest and bring necessary materials to the exam.
