 Back
BackExam 2 Study Guide: Reactions in Aqueous Solution & Electronic Structure of Atoms
Study Guide - Smart Notes

Chapter 4 – Reactions in Aqueous Solution
4.1 – General Properties of Aqueous Solutions
An aqueous solution is a solution in which water is the solvent. Understanding the behavior of substances in water is fundamental to chemistry.
Electrolyte: A substance that dissolves in water to produce ions, thus conducting electricity. Strong electrolytes dissociate completely (e.g., NaCl), while weak electrolytes dissociate partially (e.g., acetic acid).
Nonelectrolyte: A substance that dissolves in water but does not produce ions (e.g., sugar).
Examples: NaCl (strong electrolyte), CH3COOH (weak electrolyte), glucose (nonelectrolyte).
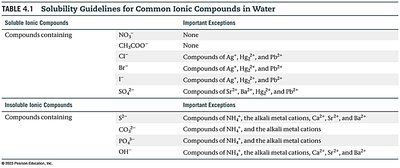
4.2 – Precipitation Reactions
Precipitation reactions occur when two solutions of ionic compounds are mixed and an insoluble product (precipitate) forms.
Solubility chart: Used to determine if a compound is soluble (aq) or insoluble (s) in water.
Precipitate: An insoluble solid formed in a reaction, usually denoted as (s).
Ionic equation: Shows all ions present in a reaction. Net ionic equation: Shows only the ions and molecules directly involved in the reaction.
Spectator ions: Ions that do not participate in the reaction and remain unchanged.
Example: Mixing AgNO3 and NaCl forms AgCl (s) as a precipitate.
4.3 – Acids, Bases, and Neutralization Reactions
Acids and bases are fundamental chemical species in aqueous reactions. Their strength and behavior are important for predicting reaction outcomes.
Acid: Produces H+ ions in solution. Base: Produces OH- ions.
Strong acids/bases: Completely dissociate in water. Weak acids/bases: Partially dissociate.
Neutralization reaction: Acid reacts with base to form water and a salt.
Common products: Water (H2O) and an ionic compound (salt).
Net ionic equation for strong acid/base:
4.4 – Oxidation-Reduction Reactions
Redox reactions involve the transfer of electrons between species. Understanding oxidation numbers is key to identifying these reactions.
Oxidation: Loss of electrons. Reduction: Gain of electrons. (OIL RIG: Oxidation Is Loss, Reduction Is Gain)
Oxidation number: Assigned to elements to track electron transfer. Rules are based on element type and compound structure.
Example: In NaCl, Na has +1, Cl has -1 oxidation number.
4.5 – Concentrations of Solutions
Concentration describes the amount of solute in a given volume of solution. Molarity is the most common unit.
Molarity (M):
Conversion: 1 L = 1000 mL
Dilution equation:
Ion concentration: For Na2SO4, [Na+] = 2 × [Na2SO4], [SO42-] = [Na2SO4]
4.6 – Solution Stoichiometry and Chemical Analysis
Stoichiometry in solutions involves using concentrations and volumes to calculate reactant and product quantities.
Titration: A method to determine concentration by reacting a known volume with a solution of known concentration.
Dimensional analysis: Used to convert units and solve stoichiometry problems.
Chapter 6 – Electronic Structure of Atoms
6.1-6.4 – Nature/Behavior of Light, Quantized Energy, and the Bohr Model
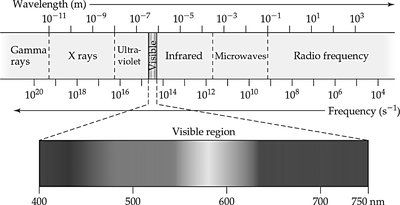
Light and energy are quantized in atomic systems. The Bohr model explains electron transitions and energy levels.
Frequency (ν): Number of cycles per second (Hz).
Wavelength (λ): Distance between peaks (meters).
Relationship: (c = speed of light, m/s)
Photon energy: (h = Planck's constant, J·s)
Bohr model: Electrons occupy quantized energy levels; transitions emit/absorb photons.
Energy change:
Emission: Electron drops to lower energy level, emitting photon. Excitation: Electron absorbs energy and moves to higher level.
6.5/6.6 – Quantum Mechanics, Atomic Orbitals/Representations of Orbitals
Quantum numbers describe the properties and locations of electrons in atoms.
Principal quantum number (n): Indicates energy level; n = 1, 2, 3, ...
Angular momentum quantum number (l): Defines orbital shape; l = 0 (s), 1 (p), 2 (d), 3 (f)
Magnetic quantum number (ml): Specifies orientation; ml = -l to +l
Shapes: s (sphere), p (dumbbell), d (cloverleaf)
6.7-6.9 – Many-Electron Atoms, Orbital Diagrams, and Electron Configurations
Electron configurations and orbital diagrams show how electrons fill atomic orbitals according to specific rules.
Spin quantum number (ms): +1/2 or -1/2; describes electron spin.
Pauli Exclusion Principle: No two electrons in an atom can have the same set of quantum numbers.
Hund’s Rule: Electrons fill degenerate orbitals singly before pairing.
Electron configuration: Order of filling: 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, ...
Condensed configuration: Uses noble gas core to simplify notation (e.g., [Ne]3s23p1).
Orbital capacity: s (2 electrons), p (6), d (10)
Example: Electron configuration for sodium (Na): 1s22s22p63s1; condensed: [Ne]3s1.
