 Back
BackGeneral Chemistry Exam 1 Review: Matter, Atoms, Molecules, and Reaction Stoichiometry
Study Guide - Smart Notes

Introduction: Matter, Energy, and Measurement
Definitions and Properties of Matter
Matter is defined as anything that occupies space and has mass. Understanding the basic properties and classifications of matter is fundamental to chemistry.
Atom: The smallest unit into which an element can be divided without losing its identity.
Element: A substance that cannot be broken down into simpler components.
Compound: A substance composed of two or more elements chemically combined.
Physical Change: Alters the state or appearance of matter without changing its composition.
Chemical Change: Alters the composition of matter, resulting in the rearrangement of atoms.
Example: Flammability is a chemical change.
Measurement and Significant Figures
Accurate measurement and proper use of significant figures are essential in chemical calculations.
Significant Figures: Digits in a measurement that are known with certainty plus one estimated digit.
Example: The number 1.050 × 109 has 4 significant figures.
Example: 0.00253 has 3 significant figures.
Unit Conversion: 6.00 inches = 15.2 cm.
Example Calculation: (1.3 × 103)(5.724 × 104) = 7.4 × 107 (rounded to correct sig figs).
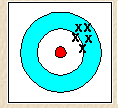
Accuracy vs. Precision
Accuracy refers to how close a measurement is to the true value, while precision refers to how close repeated measurements are to each other.
Example: The image below shows a set of measurements that are precise but not necessarily accurate.
Atoms, Molecules, and Ions
Atomic Structure and Mass
Atoms consist of protons, neutrons, and electrons. The atomic mass is calculated as the sum of protons and neutrons.
Atomic Mass Equation:
Example: An oxide ion (O2–) has 8 protons and 10 electrons.
Neutrons: Have no charge.
Example: One atom of 79Br has 44 neutrons.
Diatomic Elements and Molecular Compounds
Certain elements exist naturally as diatomic molecules, and molecular compounds are typically composed of nonmetals.
Diatomic Elements: Br2, I2, N2, Cl2, H2, O2, F2
Molecular Compounds: Usually comprised of nonmetals.
States of Nonmetals: Solid, liquid, or gas.
Structural and Empirical Formulas
Formulas represent the composition and structure of compounds.
Molecular Formula: Shows the exact number of atoms of each element (e.g., dinitrogen tetroxide is N2O4).
Empirical Formula: Shows the simplest whole-number ratio (e.g., C6H12O6 has empirical formula CH2O).
Structural Formula: Shows the bonding arrangement of atoms.
Common Ions and Nomenclature
Chemical nomenclature is used to name ions and compounds.
Example: Calcium Nitrate is Ca(NO3)2.
Example: Acetic acid is CH3COOH.
Example: Iron(III) Chlorate is Fe(ClO3)3.
Oxoanion of Sulfur with -2 charge: Sulfite ion.
Chemical Reactions and Reaction Stoichiometry
Types of Chemical Reactions
Chemical reactions are classified based on the changes occurring in reactants and products.
Combination Reaction: Two or more substances combine to form one product.
Decomposition Reaction: A single compound breaks down into two or more products.
Combustion Reaction: A substance reacts with oxygen, releasing energy and forming products like CO2 and H2O.
Balancing Chemical Equations
Balancing equations ensures the conservation of mass and atoms.
Example: 6 Li(s) + 1 N2(g) → 2 Li3N(s)
Example: 2 N2H4 + N2O4 → 3 N2 + 4 H2O
Example: 2 C3H10(g) + 11 O2(g) → 6 CO2(g) + 10 H2O(g)
Stoichiometry: Mole Calculations
Stoichiometry involves calculating the quantities of reactants and products in chemical reactions using balanced equations.
Subscripts: Indicate the number of atoms of each element.
Superscripts: Indicate the charge of ions.
Example: KClO3 Decomposition
1.65 mol KClO3 produces 2.48 mol O2
3.50 mol KCl needed requires 3.50 mol KClO3
2.73 mol KClO3 produces 2.73 mol KCl

Example: Molecule and Atom Calculations
Calculate molecules in 1.058 g H2O: molecules
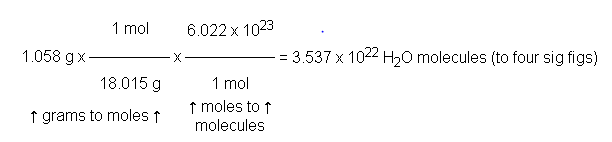
Example: Comparing Molecules in Samples
10.0 g O2 contains more molecules than 50.0 g I2
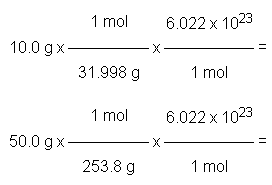
Molar Mass Calculations
Molar mass is the mass of one mole of a substance, used for converting between grams and moles.
Example: Molar mass of PbSO4 is 303.3 g/mol

Stoichiometry: Mass Calculations in Reactions
Mass calculations use balanced equations and molar masses to determine the amount of product or reactant.
Example: 4 Fe + 3 O2 → 2 Fe2O3
61.0 g Fe2O3 produced from 42.7 g Fe
56.6 g Fe2O3 produced from 17.0 g O2
53.7 g O2 needed to react with 125 g Fe

Example: Combustion of Butane
303 g CO2 produced from 100 g butane
358 g O2 needed for 100 g butane
2.33 g H2O produced from 5.38 g O2

Limiting Reactant and Yield
The limiting reactant determines the maximum amount of product that can be formed in a reaction.
Example: In the reaction 2 Al(s) + Fe2O3(s) → 2 Fe(l) + Al2O3(s), aluminum is the limiting reactant and 5.4 moles of iron can be formed.

Additional info:
Chalcogens are found in Group 6 (also known as Group 16).
Noble gases are inert due to their full valence electron shells.
