 Back
BackGeneral Chemistry Exam 3 Study Guidance
Study Guide - Smart Notes

Q1. What is the coefficient on ammonia in the balanced equation for the reaction of gaseous ammonia (NH3) with gaseous oxygen to form gaseous nitrogen monoxide and gaseous water?
Background
Topic: Balancing Chemical Equations
This question tests your ability to balance a chemical equation involving ammonia, oxygen, nitrogen monoxide, and water.
Key Terms and Concepts:
Balancing equations: Ensuring the same number of each atom on both sides of the equation.
Chemical equation: NH3 + O2 → NO + H2O
Step-by-Step Guidance
Write the unbalanced equation:
Count the number of each atom on both sides.
Start by balancing the nitrogen atoms, then hydrogen, and finally oxygen.
Adjust coefficients as needed to ensure all atoms are balanced.
Try solving on your own before revealing the answer!
Q2. Give the complete ionic equation for when aqueous solutions of Na2SO3 and HCl are mixed.
Background
Topic: Ionic Equations and Gas-Evolving Reactions
This question tests your understanding of how to write complete ionic equations and recognize gas-evolving reactions.
Key Terms and Concepts:
Complete ionic equation: Shows all soluble ionic substances dissociated into ions.
Gas evolution: Some reactions produce a gas, such as SO2.
Step-by-Step Guidance
Write the molecular equation for the reaction.
Identify all strong electrolytes and write them as ions.
Recognize if a gas forms (e.g., SO2).
Combine ions to show the complete ionic equation, including any gases or precipitates formed.
Try solving on your own before revealing the answer!
Q3. What kind of reaction occurs when aqueous bromic acid is mixed with calcium hydroxide?
Background
Topic: Types of Chemical Reactions
This question tests your ability to classify reactions, especially acid-base neutralization and precipitation reactions.
Key Terms:
Acid-base neutralization: Acid reacts with base to form water and a salt.
Precipitation: Formation of an insoluble solid.
Step-by-Step Guidance
Write the formulas for bromic acid and calcium hydroxide.
Predict the products of the reaction.
Determine if water is formed (neutralization) or a precipitate forms.
Try solving on your own before revealing the answer!
Q4. Indicate the balanced net ionic equation for: NH4Br (aq) + KOH (aq) → H2O (l) + NH3 (g) + KBr (aq)
Background
Topic: Net Ionic Equations and Gas Evolution
This question tests your ability to write net ionic equations, especially for reactions that produce a gas.
Key Terms and Concepts:
Net ionic equation: Shows only the species that actually change during the reaction.
Gas evolution: NH3 gas is produced.
Step-by-Step Guidance
Write the full ionic equation, dissociating all strong electrolytes.
Identify and remove spectator ions.
Write the net ionic equation with only the reacting species.
Try solving on your own before revealing the answer!
Q5. What kind of reaction occurs when aqueous hydrochloric acid is mixed with sodium carbonate?
Background
Topic: Types of Chemical Reactions (Gas Evolution)
This question tests your ability to recognize gas-evolving reactions, especially when acids react with carbonates.
Key Terms:
Gas evolution: Reaction produces a gas (CO2).
Acid-base reaction: Acid reacts with a base or carbonate.
Step-by-Step Guidance
Write the chemical equation for HCl and Na2CO3.
Predict the products, including any gases formed.
Classify the reaction based on the products.
Try solving on your own before revealing the answer!
Q6. A 25.00 mL sample of H2SO4(aq) requires 30.55 mL of 0.5510 M NaOH for its titration. What was the concentration of the H2SO4?
Background
Topic: Acid-Base Titration Calculations
This question tests your ability to use titration data to calculate the concentration of an acid.
Key Formula:
and are the stoichiometric coefficients from the balanced equation.
Step-by-Step Guidance
Write the balanced equation for the reaction between H2SO4 and NaOH.
Identify the stoichiometry (mole ratio) between acid and base.
Convert all volumes to liters.
Set up the titration equation and solve for the unknown concentration.
Try solving on your own before revealing the answer!
Q7. Determine the number of liters of CO2 formed at STP when 6.93 grams of C2H6 is burned in excess oxygen gas.
Background
Topic: Stoichiometry and Gas Laws
This question tests your ability to use stoichiometry and the ideal gas law to relate mass of a reactant to volume of a gas produced at STP.
Key Terms and Formulas:
STP: Standard Temperature and Pressure (0°C, 1 atm, 22.4 L/mol for gases).
Stoichiometry: Use the balanced equation to relate moles of reactant to moles of product.
Step-by-Step Guidance
Write the balanced equation for the combustion of ethane.
Convert grams of C2H6 to moles using its molar mass.
Use the mole ratio to find moles of CO2 produced.
Convert moles of CO2 to liters at STP using 22.4 L/mol.
Try solving on your own before revealing the answer!
Q8. What volume of a 0.5500 M solution of AgNO3 is needed to completely react with 100.0 mL of a 0.2500 M solution of CaCl2?
Background
Topic: Solution Stoichiometry
This question tests your ability to use molarity and stoichiometry to determine the volume of one solution needed to react with another.
Key Formula:
Where is molarity, is volume, and is the stoichiometric coefficient.
Step-by-Step Guidance
Write the balanced equation for the reaction.
Calculate moles of CaCl2 present.
Use the stoichiometric ratio to find moles of AgNO3 needed.
Calculate the required volume of AgNO3 solution using its molarity.
Try solving on your own before revealing the answer!
Q9. What mass of CO2 can be formed from 183 mg of O2? Assume excess C3H7SH.
Background
Topic: Limiting Reactant and Stoichiometry
This question tests your ability to identify the limiting reactant and use stoichiometry to calculate the mass of product formed.
Key Terms and Formulas:
Limiting reactant: The reactant that is completely consumed first.
Stoichiometry: Use the balanced equation to relate moles of O2 to moles of CO2.
Step-by-Step Guidance
Convert mass of O2 to moles using its molar mass.
Use the balanced equation to find the mole ratio between O2 and CO2.
Calculate moles of CO2 produced.
Convert moles of CO2 to grams using its molar mass.
Try solving on your own before revealing the answer!
Q10. Which substance is the limiting reactant when 4.0 g of sulfur reacts with 3.0 g of oxygen and 8.0 g of potassium hydroxide?
Background
Topic: Limiting Reactant
This question tests your ability to determine which reactant will be consumed first in a chemical reaction.
Key Terms and Formulas:
Limiting reactant: The reactant that determines the maximum amount of product formed.
Step-by-Step Guidance
Convert the mass of each reactant to moles using their molar masses.
Divide the moles of each reactant by its coefficient in the balanced equation.
The smallest value indicates the limiting reactant.
Try solving on your own before revealing the answer!
Q11. What is the percent yield if 0.30 mol Ba(NO3)2 and 0.25 mol Na3PO4 react to produce 0.066 mol Ba3(PO4)2?
Background
Topic: Percent Yield
This question tests your ability to calculate percent yield from actual and theoretical yields.
Key Formula:
Step-by-Step Guidance
Determine the limiting reactant using the mole ratios from the balanced equation.
Calculate the theoretical yield of Ba3(PO4)2 in moles.
Use the actual yield given to calculate percent yield.
Try solving on your own before revealing the answer!
Q12. If 4.0 mol of propane are burned, how many moles of carbon dioxide will be produced?
Background
Topic: Stoichiometry
This question tests your ability to use mole ratios from a balanced equation to determine the amount of product formed.
Key Terms and Formulas:
Stoichiometry: Use the balanced equation to relate moles of reactant to moles of product.
Step-by-Step Guidance
Write the balanced equation for the combustion of propane.
Identify the mole ratio between propane and carbon dioxide.
Multiply the moles of propane by the ratio to find moles of CO2 produced.
Try solving on your own before revealing the answer!
Q13. A system releases 587 kJ of heat and does 115 kJ of work on the surroundings. What is the change in internal energy of the system?
Background
Topic: First Law of Thermodynamics
This question tests your understanding of the relationship between heat, work, and internal energy.
Key Formula:
= heat (negative if released)
= work (negative if done by the system)
Step-by-Step Guidance
Assign the correct signs to and based on the information given.
Plug the values into the formula .
Calculate the sum to find the change in internal energy.
Try solving on your own before revealing the answer!
Q14. A 11.6-g sample of silicon at 86.0°C is immersed into 26.0 g of water at 24.0°C. What is the final temperature when they reach thermal equilibrium?
Background
Topic: Calorimetry and Heat Transfer
This question tests your ability to use specific heat and the principle of conservation of energy to find the final temperature.
Key Formula:
= mass, = specific heat, = change in temperature
Step-by-Step Guidance
Set up the heat lost by silicon equal to the heat gained by water.
Write the equations for for both substances.
Set up the equation and solve for the final temperature .
Try solving on your own before revealing the answer!
Q15. A cylinder with a moving piston expands from an initial volume of 0.150 L against an external pressure of 1.70 atm. The expansion does 296 J of work on the surroundings. What is the final volume of the cylinder?
Background
Topic: Work and Gas Expansion
This question tests your ability to relate work, pressure, and volume change in a gas system.
Key Formula:
= work (in J), = external pressure (in atm), = change in volume (in L)
Step-by-Step Guidance
Convert work to L·atm if necessary (1 L·atm = 101.3 J).
Rearrange the formula to solve for .
Add to the initial volume to find the final volume.
Try solving on your own before revealing the answer!
Q16. Propane has a normal boiling point of 231.2 K and a heat of vaporization of 19.04 kJ/mol. What is the vapor pressure of propane at 298.2 K?
Background
Topic: Clausius-Clapeyron Equation
This question tests your ability to use the Clausius-Clapeyron equation to relate vapor pressure and temperature.
Key Formula:
= vapor pressure at (boiling point, so atm)
in J/mol, J/(mol·K)
Step-by-Step Guidance
Convert to J/mol if needed.
Plug in the values for , , , and into the equation.
Solve for (vapor pressure at 298.2 K).
Try solving on your own before revealing the answer!
Q17. Calculate the amount of heat required to condense 2.36 kg of water at its boiling point. (ΔHvap = 40.7 kJ/mol at 100°C)
Background
Topic: Phase Changes and Enthalpy
This question tests your ability to calculate the heat involved in a phase change using enthalpy of vaporization.
Key Formula:
= moles of water, = enthalpy of vaporization
Step-by-Step Guidance
Convert mass of water to moles using its molar mass.
Multiply moles by to find the heat required.
Try solving on your own before revealing the answer!
Q18. Methane is used in a laboratory burner. When 1 mole of methane burns at constant pressure, it produces 804 kJ of heat and does 3 kJ of work. What is the value of ΔH for the combustion of one mole of methane?
Background
Topic: Enthalpy and Internal Energy
This question tests your understanding of the relationship between heat, work, and enthalpy at constant pressure.
Key Formula:
At constant pressure, enthalpy change equals heat exchanged.
Step-by-Step Guidance
Identify the sign of heat (exothermic or endothermic).
Relate the given values to the definition of enthalpy change.
Try solving on your own before revealing the answer!
Q19. Titanium reacts with iodine to form titanium(III) iodide, emitting heat. Determine the mass of titanium that reacts if 2.38×103 kJ of heat is emitted by the reaction.
Background
Topic: Stoichiometry and Enthalpy
This question tests your ability to relate the enthalpy change of a reaction to the amount of reactant consumed.
Key Formula:
per mole of reaction from the balanced equation.
Use the ratio of heat emitted to to find moles of Ti reacted.
Step-by-Step Guidance
Calculate the number of moles of reaction using the heat emitted and .
Use the stoichiometry of the reaction to find moles of Ti.
Convert moles of Ti to mass using its molar mass.
Try solving on your own before revealing the answer!
Q20. Solid CO2 "disappears" after sitting at room temperature for a while. There is no puddle of liquid. What happened?
Background
Topic: Phase Changes (Sublimation)
This question tests your understanding of phase changes, specifically sublimation.
Key Terms:
Sublimation: Direct transition from solid to gas without passing through the liquid phase.
Step-by-Step Guidance
Recall the properties of CO2 at room temperature and atmospheric pressure.
Identify the phase change that occurs without a liquid phase.
Try solving on your own before revealing the answer!
Q21. Use the bond energies provided to estimate ΔH°rxn for the reaction: 2 CH3OH (l) + 3 O2 (g) → 2 CO2 (g) + 4 H2O (g)
Background
Topic: Bond Energies and Enthalpy Calculations
This question tests your ability to estimate the enthalpy change of a reaction using bond energies.
Key Formula:
Step-by-Step Guidance
List all bonds broken in the reactants and sum their energies.
List all bonds formed in the products and sum their energies.
Subtract the total energy of bonds formed from the total energy of bonds broken.
Try solving on your own before revealing the answer!
Q22. The combustion of ethene occurs via the reaction: C2H4 (g) + 3 O2 (g) → 2 CO2 (g) + 2 H2O (g). Calculate the enthalpy for the combustion of 1 mole of ethene using the provided heat of formation values.
Background
Topic: Enthalpy of Reaction from Heats of Formation
This question tests your ability to use standard enthalpies of formation to calculate the enthalpy change for a reaction.
Key Formula:
Use the table of values provided.
Step-by-Step Guidance
Write the balanced equation and identify the number of moles of each substance.
Multiply each value by the number of moles for each product and reactant.
Sum the products and reactants separately, then subtract reactants from products.
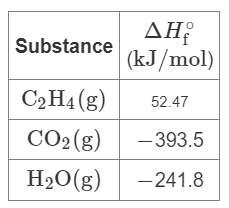
Try solving on your own before revealing the answer!
Q23. Calculate ΔHrxn for: 3 C (s) + 4 H2 (g) → C3H8 (l) using the given reactions and ΔH values.
Background
Topic: Hess's Law
This question tests your ability to use Hess's Law to combine reactions and calculate the enthalpy change for a target reaction.
Key Terms and Concepts:
Hess's Law: The enthalpy change for a reaction is the sum of the enthalpy changes for the steps into which the reaction can be divided.
Step-by-Step Guidance
Write the target reaction and the given reactions.
Manipulate the given reactions (reverse or multiply as needed) to sum to the target reaction.
Add the corresponding ΔH values, accounting for any changes made to the equations.
Try solving on your own before revealing the answer!
Q24. Under what conditions is the change in enthalpy, ΔHrxn, equal to the heat evolved in a chemical reaction?
Background
Topic: Enthalpy and Heat
This question tests your understanding of the relationship between enthalpy change and heat under different conditions.
Key Terms:
Enthalpy (ΔH): At constant pressure, the heat evolved or absorbed equals the change in enthalpy.
Step-by-Step Guidance
Recall the definition of enthalpy and under what conditions it equals heat.
Identify the correct condition from the options provided.
Try solving on your own before revealing the answer!
Q25. The transition shown by the line from C to E is…
Background
Topic: Phase Diagrams
This question tests your ability to interpret phase diagrams and identify phase transitions.
Key Terms:
Phase transition: Change from one state of matter to another (e.g., sublimation, deposition, boiling).
Step-by-Step Guidance
Locate points C and E on the phase diagram.
Determine which phases are present at each point.
Identify the phase change represented by the line from C to E.
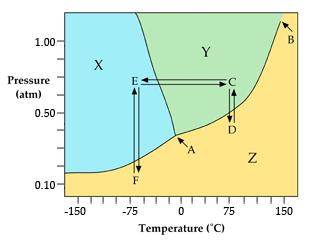
Try solving on your own before revealing the answer!
Q26. The line that runs from point A to point B is known as the…
Background
Topic: Phase Diagrams
This question tests your ability to identify the different curves on a phase diagram (e.g., fusion, vaporization, sublimation curves).
Key Terms:
Fusion curve: Boundary between solid and liquid phases.
Vaporization curve: Boundary between liquid and gas phases.
Sublimation curve: Boundary between solid and gas phases.
Step-by-Step Guidance
Locate points A and B on the phase diagram.
Identify which phases are separated by the line from A to B.
Determine the name of the curve based on the phases involved.

Try solving on your own before revealing the answer!
