 Back
BackGeneral Chemistry Exam Review Guidance: Electrolytes, Thermochemistry, Gas Laws, and More
Study Guide - Smart Notes

Q1. Which of the following are weak electrolytes?
Background
Topic: Electrolytes and Acid/Base Strength
This question tests your understanding of strong and weak electrolytes, including strong/weak acids and bases.
Key Terms:
Electrolyte: A substance that produces ions when dissolved in water.
Strong electrolyte: Completely dissociates in water (e.g., NaCl, HBr).
Weak electrolyte: Partially dissociates in water (e.g., CH3NH2, HC2H3O2).
Step-by-Step Guidance
Identify which compounds are strong acids, strong bases, weak acids, or weak bases.
Recall that strong acids and bases are strong electrolytes, while weak acids and bases are weak electrolytes.
Look for the answer choices that include only weak acids and/or weak bases.
Try solving on your own before revealing the answer!
Q14. Calculate the amount of heat (in kJ) necessary to raise the temperature of 55.8 g benzene by 48.8 K. The specific heat capacity of benzene is 1.05 J/g°C.
Background
Topic: Thermochemistry – Heat Calculations
This question tests your ability to use the formula for heat transfer involving mass, specific heat, and temperature change.
Key formula:
= heat absorbed (J)
= mass (g)
= specific heat capacity (J/g\cdot°C)
= change in temperature (°C or K)
Step-by-Step Guidance
Identify the mass (), specific heat (), and temperature change () from the question.
Plug these values into the formula .
Calculate in joules, then convert to kilojoules by dividing by 1000.
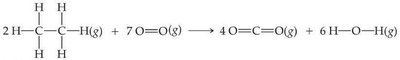
Try solving on your own before revealing the answer!
Q15. Use Table 5.4 to estimate ∆H for the following combustion reaction:
Background
Topic: Thermochemistry – Bond Enthalpy Calculations
This question tests your ability to estimate the enthalpy change of a reaction using average bond enthalpies.
Key formula:
Bonds broken: Reactants
Bonds formed: Products
Step-by-Step Guidance
Write out the balanced chemical equation and identify all bonds broken and formed.
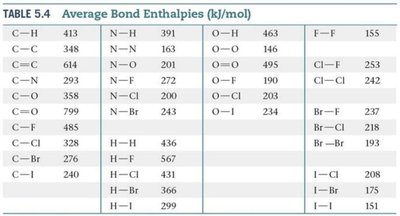
Use Table 5.4 to find the bond enthalpies for each bond involved.
Calculate the total energy required to break all bonds in the reactants.
Calculate the total energy released when new bonds are formed in the products.
Try solving on your own before revealing the answer!
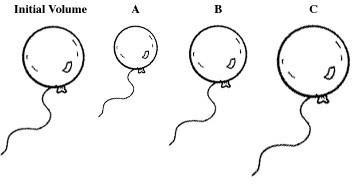
Q36. The pressure is changed from 0.98 atm to 1.2 atm at constant temperature. Select the letter of the balloon diagram that corresponds to the given change in conditions.
Background
Topic: Gas Laws – Boyle's Law
This question tests your understanding of how gas volume changes with pressure at constant temperature.
Key formula:
= initial pressure
= initial volume
= final pressure
= final volume
Step-by-Step Guidance
Recall that increasing pressure at constant temperature causes the volume of a gas to decrease (Boyle's Law).
Compare the initial and final pressures to determine which balloon diagram shows a decrease in volume.
Identify the diagram that matches the expected change.
Try solving on your own before revealing the answer!
