 Back
BackGeneral Chemistry Final Exam & Midterm Study Guidance
Study Guide - Smart Notes

Q1. How are the ‘actual nuclear charge’ and the ‘effective nuclear charge’ experienced by an electron in a many-electron atom related to one another?
Background
Topic: Atomic Structure & Periodicity
This question tests your understanding of the concepts of actual nuclear charge (Z) and effective nuclear charge (Zeff), and how electron shielding affects the attraction between the nucleus and electrons in multi-electron atoms.
Key Terms and Formulas:
Actual Nuclear Charge (Z): The total positive charge of the nucleus, equal to the number of protons.
Effective Nuclear Charge (Zeff): The net positive charge experienced by an electron, accounting for both the attraction from the nucleus and the repulsion from other electrons (shielding).
Shielding: The phenomenon where inner electrons partially block the attraction between the nucleus and outer electrons.
Key formula: Where is the atomic number and is the shielding constant.
Step-by-Step Guidance
Recall that the actual nuclear charge () is simply the number of protons in the nucleus for a given element.
Understand that electrons in inner shells repel outer electrons, reducing the full effect of the nuclear charge. This is called shielding.
Apply the formula to see that the effective nuclear charge is always less than the actual nuclear charge, except for hydrogen (where there is only one electron and no shielding).
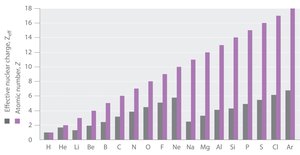
Examine the provided bar graph: the purple bars (actual nuclear charge) are always higher than the gray bars (effective nuclear charge), illustrating this relationship across the periodic table.
Try solving on your own before revealing the answer!
Q2. What is the hybridization? What is the shape at each carbon atom? How many sigma? How many pi bonds?
Background
Topic: Chemical Bonding & Molecular Structure
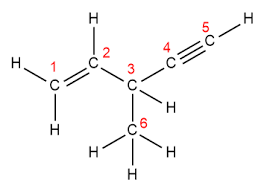
This question asks you to analyze a hydrocarbon molecule (structure provided) to determine the hybridization and geometry of each carbon atom, as well as to count the number of sigma (σ) and pi (π) bonds present.
Key Terms and Formulas:
Hybridization: The mixing of atomic orbitals to form new hybrid orbitals suitable for bonding.
Common Hybridizations:
sp3: tetrahedral geometry, 4 sigma bonds
sp2: trigonal planar geometry, 3 sigma bonds, 1 pi bond
sp: linear geometry, 2 sigma bonds, 2 pi bonds
Sigma (σ) bond: A single covalent bond formed by the direct overlap of orbitals.
Pi (π) bond: A bond formed by the sideways overlap of p orbitals, present in double and triple bonds.
Step-by-Step Guidance
For each carbon atom, count the number of regions of electron density (bonds and lone pairs) to determine its hybridization.
Assign the geometry based on the hybridization: sp3 (tetrahedral), sp2 (trigonal planar), sp (linear).
Identify all single, double, and triple bonds in the molecule. Remember: each single bond is a sigma bond; in double bonds, one is sigma and one is pi; in triple bonds, one is sigma and two are pi.
Count the total number of sigma and pi bonds in the molecule by analyzing each bond type.
Try solving on your own before revealing the answer!
