 Back
BackGeneral Chemistry Final Exam Study Guidance
Study Guide - Smart Notes

Q1. Van der Waals equation: The volumes of real gases often exceed those calculated by the ideal gas equation. These deviations are best attributed to:
Background
Topic: Real Gases vs. Ideal Gases
This question tests your understanding of why real gases deviate from ideal behavior, specifically regarding the Van der Waals equation.
Key Terms:
Ideal Gas Law:
Van der Waals Equation:
"a" accounts for intermolecular attractions; "b" accounts for the volume of gas molecules.
Step-by-Step Guidance
Recall that the ideal gas law assumes gas molecules have no volume and no intermolecular forces.
Understand that real gases have molecules with finite volume and experience intermolecular attractions.
Consider which factor (volume, attractions, kinetic energy, dissociation) would cause the measured volume to be greater than predicted.
Try solving on your own before revealing the answer!
Q2. Identifying a system: ΔT is measured inside a constant pressure calorimeter while 0.50 g of MgSO4 is dissolved in 10.0 ml of water. In this case, the system is:
Background
Topic: Thermodynamics – System and Surroundings
This question tests your ability to identify the "system" in a calorimetry experiment.
Key Terms:
System: The part of the universe being studied.
Surroundings: Everything outside the system.
Calorimeter: Device used to measure heat changes.
Step-by-Step Guidance
Identify what is undergoing change (dissolution of MgSO4).
Consider whether the system is the chemical process, the solution, or the calorimeter.
Recall that the system is typically the reactants and products involved in the process being measured.
Try solving on your own before revealing the answer!
Q3. Types of systems: Which of the following depicts an open system? Note: double walls indicate insulation that prevents heat flow.
Background
Topic: Thermodynamics – Types of Systems
This question tests your ability to distinguish between open, closed, and isolated systems based on diagrams.
Key Terms:
Open System: Can exchange both matter and energy with surroundings.
Closed System: Can exchange energy but not matter.
Isolated System: Cannot exchange matter or energy.
Step-by-Step Guidance
Examine each diagram for features that allow exchange of matter and/or energy.
Look for openings or lack of insulation to identify an open system.
Recall that double walls indicate insulation (no heat flow).


Try solving on your own before revealing the answer!
Q10. Calorimetry: 84.12 g of gold at 256.3 ˚C is placed in 106.4 g of H2O at 25.0 ˚C. What is the final temperature of this system, in °C?
Background
Topic: Calorimetry – Heat Transfer
This question tests your ability to calculate the final temperature when two substances at different temperatures are mixed.
Key Terms and Formulas:
Specific Heat (): Amount of heat required to raise 1 g of a substance by 1°C.
Heat Transfer Formula:
At equilibrium, (heat lost by gold = heat gained by water).

Step-by-Step Guidance
Write the heat transfer equations for both gold and water: , .
Set up the equation: .
Plug in the masses, specific heats, and initial temperatures for both substances.
Combine the equations to solve for , but stop before the final calculation.
Try solving on your own before revealing the answer!
Q17. Pressure-reading devices: The device above is specifically a(n):
Background
Topic: Measuring Gas Pressure
This question tests your ability to identify types of manometers and barometers used in chemistry.
Key Terms:
Manometer: Device for measuring gas pressure.
Open-end Manometer: One end open to atmosphere, one to gas sample.
Barometer: Measures atmospheric pressure.
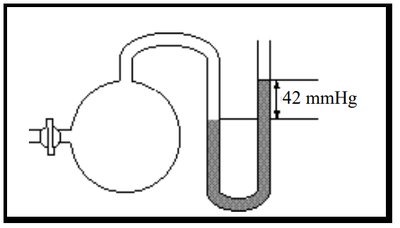
Step-by-Step Guidance
Observe the device: one end is connected to a bulb (gas sample), the other is open to the atmosphere.
Note the difference in mercury levels (42 mmHg) between the two sides.
Recall the definitions of open-end manometer and barometer.
Try solving on your own before revealing the answer!
Q19. Surface tension, IMFs, Viscosity: Based on the droplets sitting on a surface, which substance, A or B, do you expect to be less viscous?
Background
Topic: Intermolecular Forces – Surface Tension and Viscosity
This question tests your ability to interpret droplet shapes in terms of surface tension and viscosity.
Key Terms:
Surface Tension: The energy required to increase the surface area of a liquid.
Viscosity: Resistance to flow.
Droplet Shape: More spherical = higher surface tension.
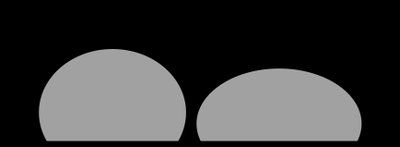
Step-by-Step Guidance
Compare the shapes of droplets A and B: which is more spherical?
Recall that higher surface tension leads to more spherical droplets.
Consider how viscosity affects the spreading of droplets.
Try solving on your own before revealing the answer!
