 Back
BackGeneral Chemistry Study Guide: Chemical Kinetics, Equilibrium, Thermodynamics, Acids/Bases, and Electrochemistry
Study Guide - Smart Notes

Chemical Kinetics
Reaction Rates and Monitoring Progress
Chemical kinetics studies the speed at which chemical reactions occur and the factors affecting them. The rate of a reaction is defined as the change in concentration of reactants or products per unit time.
Reaction Rate: for disappearance of reactant A; for appearance of product B.
Monitoring Progress: Rates can be measured by tracking concentration changes over time.
Example: For the reaction , A is consumed at twice the rate of C formation.

Rate Laws and Experimental Determination
Rate laws express the relationship between reaction rate and reactant concentrations.
General Form: For , .
Experimental Determination: Rate laws must be determined experimentally, as they may not directly match reaction stoichiometry for complex mechanisms.
Example: Iodine clock reaction: .

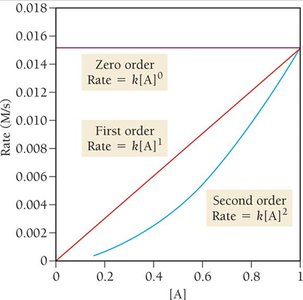
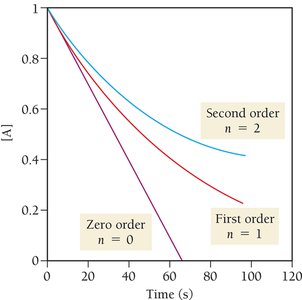
Reaction Order and Integrated Rate Laws
The order of a reaction describes how the rate depends on reactant concentrations.
Zero Order: ;
First Order: ;
Second Order: ;
Half-life: For first order,
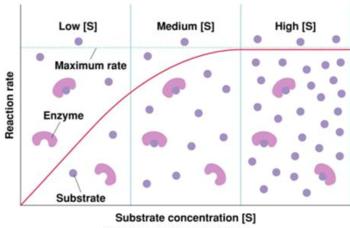
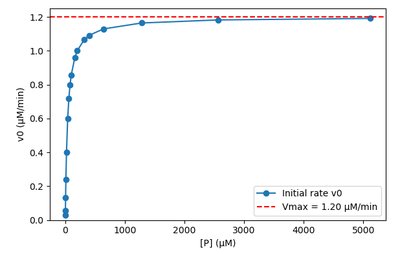
Enzyme Kinetics and Michaelis-Menten Model
Enzyme-catalyzed reactions often display saturation behavior, leading to zero-order kinetics at high substrate concentrations.
Michaelis-Menten Equation:
Zero Order: At high substrate, rate is independent of [S].
Example: Ethanol metabolism in the liver follows zero-order kinetics at high concentrations.

Chemical Equilibrium
Dynamic Equilibrium and the Equilibrium Constant
Chemical equilibrium occurs when the rates of the forward and reverse reactions are equal, resulting in constant concentrations of reactants and products.
Equilibrium Constant (K): For ,
Law of Mass Action: The equilibrium expression depends on the balanced equation.
ICE Tables: Used to calculate equilibrium concentrations.
Le Châtelier’s Principle
Le Châtelier’s Principle states that a system at equilibrium will shift to counteract any imposed change.
Adding Reactant: Shifts equilibrium toward products.
Adding Product: Shifts equilibrium toward reactants.
Changing Temperature: Endothermic reactions: increasing temperature shifts equilibrium toward products; exothermic: toward reactants.
Thermodynamics
Spontaneity, Enthalpy, and Entropy
Thermodynamics determines whether a reaction is spontaneous and how much energy is exchanged.
Enthalpy (ΔH): Heat exchanged at constant pressure.
Entropy (ΔS): Measure of disorder;
Spontaneous Process: Occurs naturally;
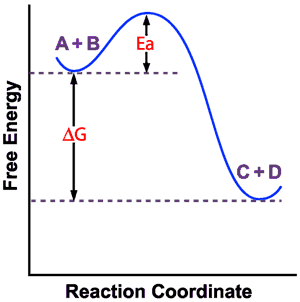
Gibbs Free Energy
Gibbs free energy combines enthalpy and entropy to predict spontaneity.
Equation:
Standard State:
Relationship to K:
Acids, Bases, and Buffers
Acid/Base Definitions and pH
Acids and bases are defined by their ability to donate or accept protons or electron pairs.
Arrhenius: Acids produce H+, bases produce OH- in water.
Brønsted-Lowry: Acids donate protons, bases accept protons.
Lewis: Acids accept electron pairs, bases donate electron pairs.
pH Calculation:
Strong and Weak Acids/Bases
Strong Acids: Completely dissociate (e.g., HCl, HNO3)
Weak Acids: Partially dissociate (e.g., CH3COOH)
Ka and Kb: Acid and base dissociation constants; relate to strength.
Buffer Solutions and Henderson-Hasselbalch Equation
Buffers resist changes in pH by neutralizing added acid or base.
Buffer Criteria: Mixture of weak acid and its conjugate base.
Henderson-Hasselbalch:
Electrochemistry
Redox Reactions and Oxidation States
Redox reactions involve electron transfer.
Oxidation: Loss of electrons; increase in oxidation state.
Reduction: Gain of electrons; decrease in oxidation state.
Assigning Oxidation States: Based on electronegativity and bonding.
Electrochemical Cells
Electrochemical cells convert chemical energy to electrical energy (voltaic/galvanic) or vice versa (electrolytic).
Anode: Site of oxidation.
Cathode: Site of reduction.
Cell Potential (Ecell):
Standard Electrode Potentials: Measured against the Standard Hydrogen Electrode (SHE).
Relationship Between Gibbs Energy and Cell Potential
Equation:
Nernst Equation:
Applications: Batteries, Fuel Cells, and Electrolysis
Batteries: Engineered voltaic cells (e.g., lead-acid, Li-ion).
Fuel Cells: Convert fuel (e.g., H2, CH4) to electricity.
Electrolysis: Uses electrical energy to drive non-spontaneous reactions.
Radioactivity and Nuclear Chemistry
Types of Radioactive Decay
Alpha Decay: Emission of He nucleus.
Beta Decay: Emission of electron or positron.
Gamma Decay: Emission of high-energy photon.
Kinetics of Radioactive Decay
Radioactive decay follows first-order kinetics.
Rate Law:
Half-life:
Example: Carbon-14 dating, medical tracers.
Tables
Summary Table: Rate Laws for Zero, First, and Second Order Reactions
Reaction Order | Rate Law | Integrated Form |
|---|---|---|
Zero | Rate = k | (A) – (A0) = -kt |
First | Rate = k(A) | ln[(A)/(A0)] = -kt |
Second | Rate = k(A)2 | (1/A) – (1/A0) = kt |
Summary Table: Common Strong Acids and Bases
Strong Acids | Strong Bases |
|---|---|
HI, HBr, HCl, HClO4, H2SO4, HNO3 | LiOH, NaOH, KOH, Ba(OH)2, Ca(OH)2, Sr(OH)2 |
Summary Table: Standard Electrode Potentials (Selected)
Half-Reaction | Eo (V) |
|---|---|
2 H+ + 2 e- → H2 | 0.00 |
F2 + 2 e- → 2 F- | +2.87 |
Cu2+ + 2 e- → Cu | +0.34 |
Zn2+ + 2 e- → Zn | -0.76 |
Additional info:
Some context and examples were inferred for completeness, such as the use of ICE tables and the application of the Nernst equation.
Images included are directly relevant to the explanation of the adjacent paragraphs, such as reaction coordinate diagrams, enzyme kinetics, and radioactive decay.
