 Back
BackIntermolecular and Intramolecular Forces in Chemistry
Study Guide - Smart Notes

Intermolecular and Intramolecular Forces
Definitions and Overview
Understanding the forces that hold molecules together and those that act between molecules is essential in general chemistry. These forces determine many physical properties of substances, such as boiling and melting points, solubility, and physical state.
Intermolecular forces: Attract ive forces that occur among molecules. These are responsible for holding molecules together in the liquid and solid states.
Intramolecular forces: Attractive forces that occur within a molecule, such as covalent bonds between atoms.
Relative Strength of Forces
Intermolecular forces are generally much weaker than intramolecular (covalent) bonds.
Despite their weakness, intermolecular forces are crucial for determining the physical properties of substances.
Types of Intermolecular Forces
There are two main types of intermolecular forces discussed in introductory chemistry:
Van der Waals' forces: A general term that includes all intermolecular forces except those due to covalent or ionic bonding. These include:
London dispersion forces: Present in all molecules, especially significant in nonpolar molecules.
Dipole-dipole interactions: Occur between polar molecules.
Hydrogen bonding: A special, stronger type of dipole-dipole interaction that occurs when hydrogen is bonded to highly electronegative atoms (N, O, or F).
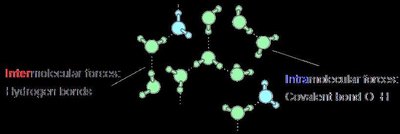
Example: Water Molecules
Water (H2O) is a classic example where both intermolecular and intramolecular forces are important:
Intramolecular forces: The covalent bonds between hydrogen and oxygen within a single water molecule.
Intermolecular forces: Hydrogen bonds between the hydrogen atom of one water molecule and the oxygen atom of another.
Comparison Table: Intermolecular vs. Intramolecular Forces
Type of Force | Acts Between | Relative Strength | Example |
|---|---|---|---|
Intramolecular | Atoms within a molecule | Strong | O-H bond in H2O |
Intermolecular | Separate molecules | Weaker | Hydrogen bond between H2O molecules |
Key Equations and Concepts
Hydrogen Bonding: Occurs when H is bonded to N, O, or F. The strength of hydrogen bonds is intermediate between typical dipole-dipole interactions and covalent bonds.
Boiling Point Trend: Substances with stronger intermolecular forces generally have higher boiling points.
Example: Water has a much higher boiling point than expected for its molecular weight due to strong hydrogen bonding between molecules.
Additional info: Van der Waals' forces is a collective term that includes London dispersion forces, dipole-dipole interactions, and dipole-induced dipole interactions. Hydrogen bonding is considered a particularly strong type of dipole-dipole interaction.
