 Back
BackInternal Energy and Enthalpy in Chemical Systems
Study Guide - Smart Notes

Internal Energy and Enthalpy
The First Law of Thermodynamics
The First Law of Thermodynamics states that energy cannot be created or destroyed, only transferred or transformed. In chemistry, this law is applied to understand how energy moves between a system and its surroundings during chemical reactions.
Internal energy (E) is the sum of all kinetic and potential energies of the particles in a system.
Energy transfer can occur as heat (q) or work (w).
The change in internal energy is given by:
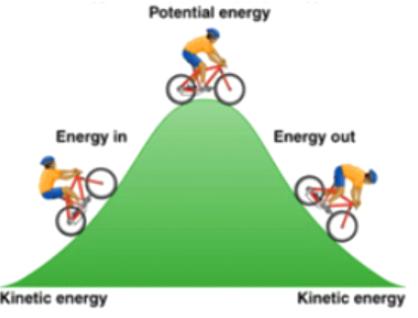
Example: A cyclist going up a hill increases potential energy (energy in), while going down converts it back to kinetic energy (energy out).
Defining Chemical Systems
In thermochemistry, it is essential to distinguish between the system (the part of the universe under study) and the surroundings (everything else).
Open system: Can exchange both matter and energy with surroundings.
Closed system: Can exchange energy but not matter.
Isolated system: Cannot exchange either matter or energy.
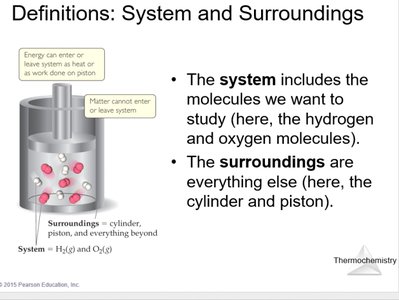
Example: In a reaction vessel, the gases inside are the system, while the container and everything outside are the surroundings.
Internal Energy of a System
The internal energy of a system is the sum of all kinetic and potential energies of its components. Measuring the absolute internal energy is impractical, so chemists focus on changes in internal energy ().
Energy can be lost to or gained from the surroundings.
Sign conventions help track these changes.
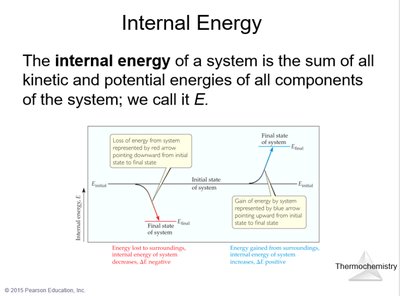
Example: If a system loses energy to its surroundings, is negative; if it gains energy, $\Delta E$ is positive.
Sign Conventions for Energy Changes
Sign conventions are used to indicate the direction of energy transfer:
Quantity | Positive (+) | Negative (-) |
|---|---|---|
q (heat) | System gains heat | System loses heat |
w (work) | Work done on system | Work done by system |
Net gain of energy by system | Net loss of energy by system |

Work in Chemical Systems
Work is often associated with changes in volume, especially in reactions involving gases. The mathematical model for work done by a system is:
Where P is pressure and is the change in volume.
Work is negative when the system does work on the surroundings (expansion).
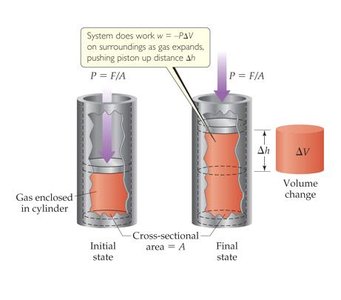
Example: When a gas expands in a cylinder, it pushes the piston up, doing work on the surroundings.
Enthalpy: Definition and Properties
Enthalpy (H) is a thermodynamic quantity that is particularly useful for reactions occurring at constant pressure. It is defined as:
Change in enthalpy:
At constant pressure:
Enthalpy change () is easier to measure than absolute enthalpy, and it corresponds to the heat exchanged at constant pressure.
Enthalpy Under Constant Volume and Pressure
Reactions can occur under constant volume or constant pressure conditions:
Constant volume: No work is done (), so (heat at constant volume).
Constant pressure: (heat at constant pressure).
Example: Most laboratory reactions are performed at constant pressure, so enthalpy change is the relevant quantity.
Enthalpy of Reaction and Energy Diagrams
The enthalpy of reaction () is the enthalpy change associated with a chemical reaction. It can be visualized using energy diagrams, which show the relative enthalpy of reactants and products.
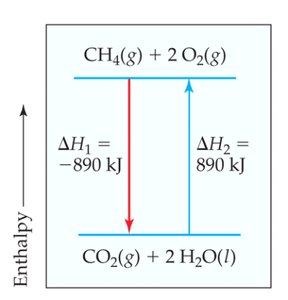
Example: The combustion of methane: , kJ (exothermic).
Endothermic vs. Exothermic Reactions
Breaking chemical bonds requires energy (endothermic), while forming bonds releases energy (exothermic). The overall enthalpy change depends on the balance between these processes.
Endothermic reaction: Absorbs heat,
Exothermic reaction: Releases heat,
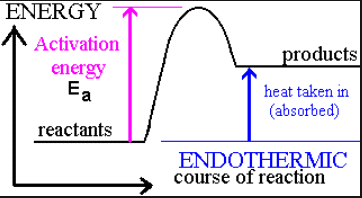
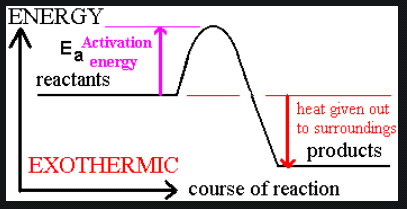
Example: Photosynthesis is endothermic; combustion is exothermic.
Quantitative Assessment of Enthalpy of Reaction
Enthalpy changes can be measured experimentally or calculated using standard enthalpies of formation. These values are essential for predicting reaction energetics and designing chemical processes.
Standard enthalpy of formation () is the enthalpy change for forming 1 mole of a compound from its elements in their standard states.
Enthalpy of reaction can be calculated using:
Example: Calculating the enthalpy change for the combustion of methane using standard enthalpies of formation.
