 Back
BackIntroduction to Solutions and Aqueous Reactions: Solution Concentration, Preparation, and Types of Reactions
Study Guide - Smart Notes

Solution Concentration and Types of Solutions
Definitions and Key Concepts
Solutions are homogeneous mixtures composed of a solute (minority component) dissolved in a solvent (majority component). When water is the solvent, the solution is termed aqueous. The concentration of a solution quantifies the amount of solute relative to the solvent or total solution volume.
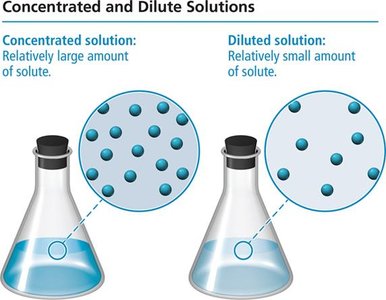
Concentrated solution: Contains a relatively large amount of solute.
Dilute solution: Contains a relatively small amount of solute.
Quantifying Solution Concentration: Molarity
Molarity (M) is the most common unit for expressing solution concentration in chemistry. It is defined as the number of moles of solute per liter of solution:
Note: Molarity is based on the total volume of the solution, not just the solvent.
Calculating Molarity: Example
To determine the molarity of a solution, first convert the mass of solute to moles, then divide by the total solution volume in liters.
Example: Calculate the molarity of a solution with 25.5 g KBr dissolved in 1.75 L of solution.
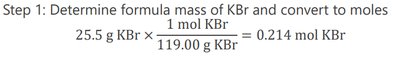
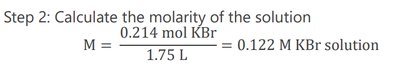
Preparing Solutions of Specified Concentration
To prepare a solution of a specific molarity, weigh out the required amount of solute, dissolve it in a portion of solvent, and dilute to the desired final volume.
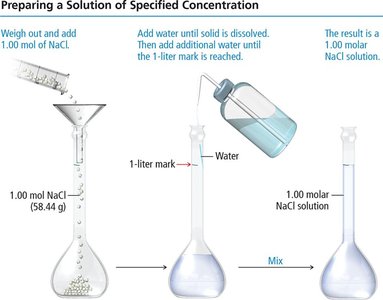
Solid solutes are weighed and dissolved in solvent.
For liquid solutes, a measured volume is mixed with solvent.
Using Molarity as a Conversion Factor
Molarity allows conversion between volume of solution and moles of solute:
To find moles:
To find volume:


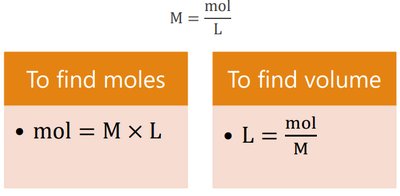
Preparing a Solution from a Solid Solute: Example
To prepare a solution of a given molarity and volume, calculate the required mass of solute:
Convert volume to liters.
Calculate moles of solute needed:
Convert moles to grams using molar mass.
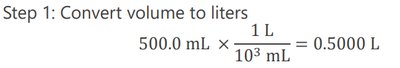
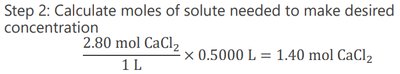
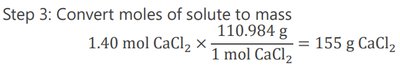
Solution Dilution
Concept and Calculation
Stock solutions are concentrated solutions stored in the laboratory. To prepare a less concentrated solution, dilute the stock solution by adding more solvent. The amount of solute remains constant:
= initial (stock) concentration
= volume of stock solution used
= final (diluted) concentration
= final total volume
Solute-Solvent Interactions and Dissolution
Forces in Solution Formation
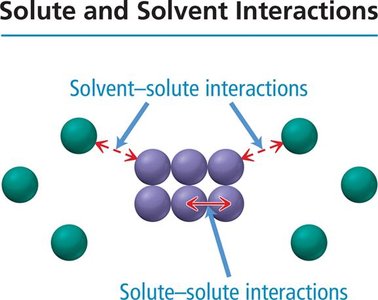
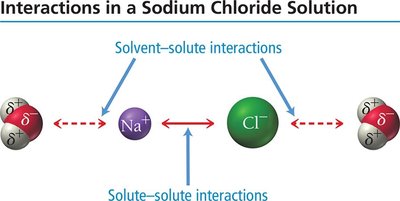
When a solute dissolves in a solvent, the process depends on the relative strengths of solute-solute, solvent-solvent, and solute-solvent interactions. If solute-solvent attractions are strong enough, the solute dissolves.
Charge Distribution in Water
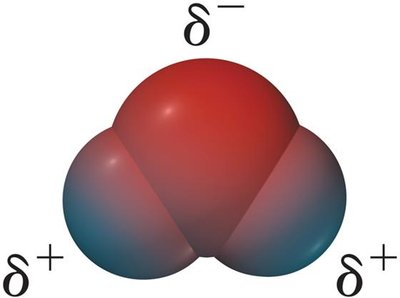
Water is a polar molecule with an uneven distribution of electron density, resulting in partial negative charge on oxygen and partial positive charges on hydrogen atoms.
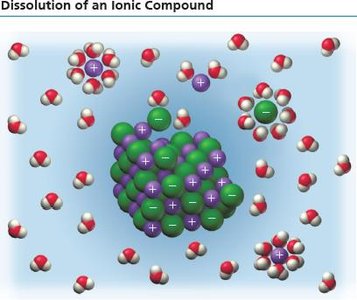
Solvation of Ionic Compounds
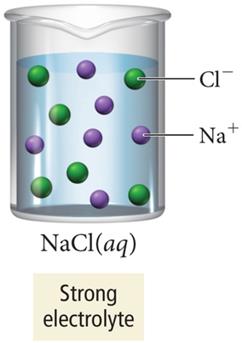
When ionic compounds dissolve, ions are separated and surrounded by water molecules, which insulate them from each other and allow the solution to conduct electricity.
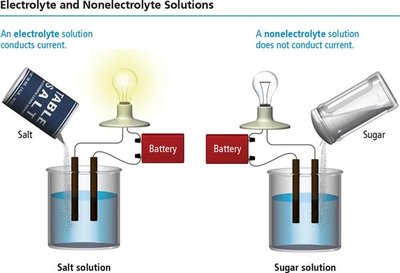
Electrolytes and Nonelectrolytes
Definitions and Examples
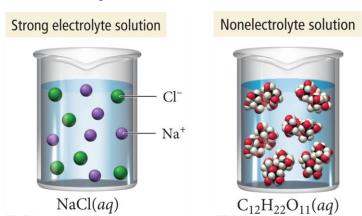
Electrolytes: Substances that dissolve in water to form ions and conduct electricity (e.g., NaCl).
Nonelectrolytes: Substances that dissolve as molecules and do not conduct electricity (e.g., sugar).
Strong and Weak Electrolytes
Strong electrolytes: Completely dissociate into ions (e.g., ionic compounds, strong acids).
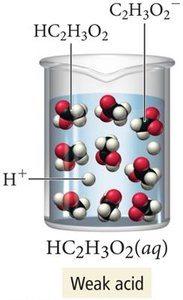
Weak electrolytes: Partially dissociate into ions (e.g., weak acids).
Solubility and Precipitation Reactions
Solubility of Salts
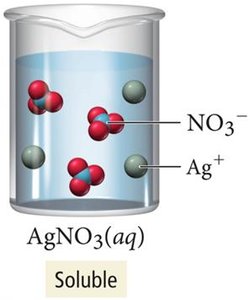
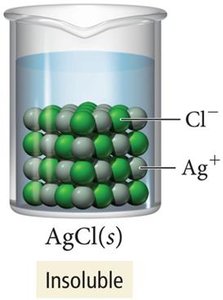
Not all ionic compounds are soluble in water. Solubility depends on the nature of the ions and can be predicted using solubility rules. For example, AgNO3 is soluble, while AgCl is insoluble.
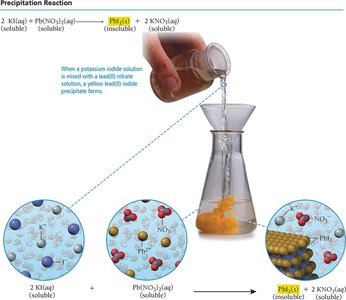
Precipitation Reactions
A precipitation reaction occurs when mixing two aqueous solutions produces an insoluble solid (precipitate). If all products are soluble, no reaction occurs.
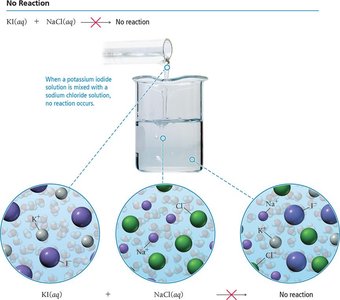
Predicting Precipitation Reactions
Identify ions in each reactant.
Exchange ions to form possible products.
Use solubility rules to determine if a precipitate forms.
Write the balanced equation, indicating (s) for solids and (aq) for aqueous species.
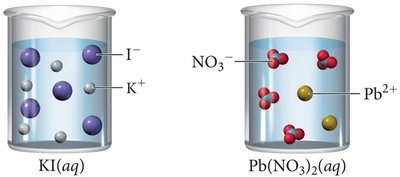
Representing Aqueous Reactions
Molecular equation: Shows complete formulas for all reactants and products.
Complete ionic equation: Shows all strong electrolytes as ions.
Net ionic equation: Shows only the species that actually change during the reaction (spectator ions are omitted).
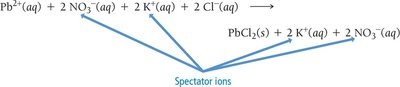
Acid–Base and Gas-Evolution Reactions
Acid–Base Reactions
In an acid–base (neutralization) reaction, an acid reacts with a base to produce water and a salt. The net ionic equation for a strong acid–strong base reaction is:
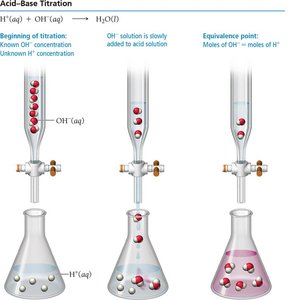
Acid–Base Titrations
Titration is a laboratory technique to determine the concentration of an unknown solution by reacting it with a solution of known concentration. The equivalence point is reached when moles of acid equal moles of base.
Gas-Evolving Reactions
Some reactions in aqueous solution produce a gas, either directly or through the decomposition of an unstable intermediate. Common gases evolved include H2S, CO2, SO2, and NH3.
Oxidation–Reduction (Redox) Reactions
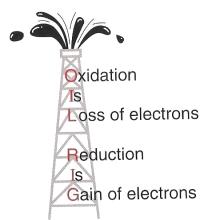
Definitions and Electron Transfer
Redox reactions involve the transfer of electrons between substances. Oxidation is the loss of electrons, and reduction is the gain of electrons. These processes always occur together.
Identifying Oxidation and Reduction
Oxidation states (numbers) are assigned to elements in compounds to track electron transfer. The element whose oxidation state increases is oxidized (reducing agent), and the one whose oxidation state decreases is reduced (oxidizing agent).
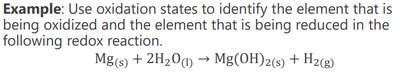

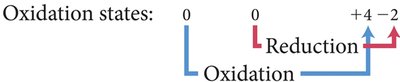
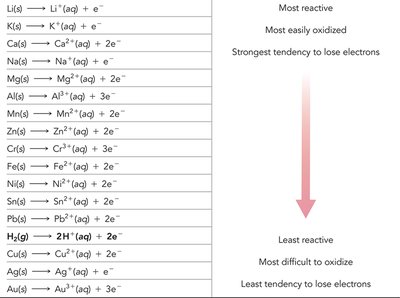
Activity Series of Metals
The activity series ranks metals by their tendency to lose electrons (be oxidized). Metals higher in the series are more easily oxidized and more reactive.
Summary: Understanding solution concentration, preparation, and the types of reactions that occur in aqueous solutions is fundamental to general chemistry. Mastery of these concepts enables prediction and analysis of chemical behavior in laboratory and real-world contexts.
