 Back
BackIonic Compounds and Chemical Bonding: Structure, Formation, and Properties
Study Guide - Smart Notes

Chemical Bonding
Introduction to Chemical Bonds
Chemical bonds are the attractive forces that hold atoms or ions together in compounds. These bonds are formed through the interaction of electrons, particularly those in the outermost shell, known as valence electrons. The type of bond formed depends on the nature of the atoms involved and their tendency to lose, gain, or share electrons.
Chemical bonds are essential for the formation of molecules and compounds.
Valence electrons play a critical role in bond formation.
Atoms bond to achieve a more stable electron configuration, often resembling that of noble gases.
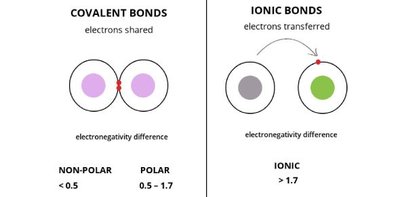
Types of Chemical Bonds
Ionic, Covalent, and Metallic Bonds
There are three primary types of chemical bonds: ionic, covalent, and metallic. Each type involves different mechanisms of electron interaction and occurs between specific types of elements.
Ionic bonds occur between metals and nonmetals, involving the transfer of electrons from one atom to another, resulting in the formation of ions.
Covalent bonds occur between nonmetals, involving the sharing of electrons between atoms.
Metallic bonds occur between metals, where electrons are delocalized and shared among a lattice of atoms.
Examples: Salt (ionic), sugar (covalent), copper wire (metallic).
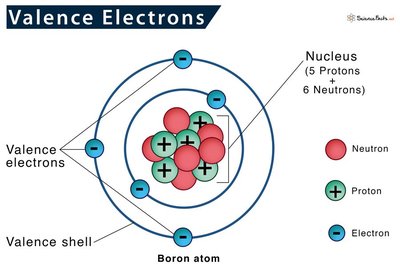
Valence Electrons and the Octet Rule
Valence Electrons
Valence electrons are the electrons in the outermost shell of an atom. They determine the chemical properties and reactivity of an element. The periodic table can be used to predict the number of valence electrons for main group elements.
Elements in the same group have the same number of valence electrons.
Valence electrons are involved in bond formation.

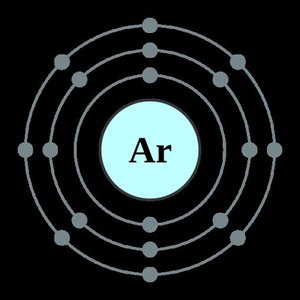
The Octet Rule
The octet rule states that atoms tend to gain, lose, or share electrons to achieve a full set of eight valence electrons, similar to the electron configuration of noble gases. This drive for stability explains the formation of many chemical bonds.
Atoms with a full octet are particularly stable (e.g., noble gases).
Metals tend to lose electrons, while nonmetals tend to gain or share electrons to satisfy the octet rule.
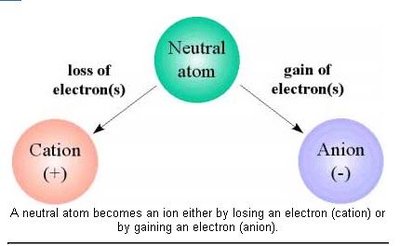
Ions and Ionic Compounds
Formation of Ions
An ion is an atom or group of atoms that has gained or lost electrons, resulting in a net charge. Cations are positively charged ions (formed by losing electrons), and anions are negatively charged ions (formed by gaining electrons).
Cation: Positively charged ion (e.g., Na+).
Anion: Negatively charged ion (e.g., Cl-).
Neutral atoms become ions to achieve a stable electron configuration.
Formation of Ionic Bonds
Ionic bonds are formed when electrons are transferred from a metal atom to a nonmetal atom, resulting in the formation of oppositely charged ions. The electrostatic attraction between these ions holds them together in an ionic compound.
Metals lose electrons to form cations; nonmetals gain electrons to form anions.
The resulting compound is electrically neutral overall.
Predicting Ion Charges
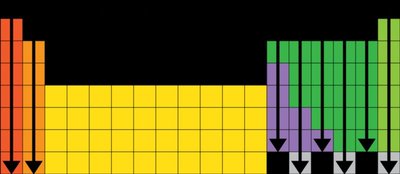
The charge of an ion can often be predicted based on its position in the periodic table:
Group | Common Ion Charge |
|---|---|
1 (Alkali metals) | +1 |
2 (Alkaline earth metals) | +2 |
13 | +3 |
15 | -3 |
16 | -2 |
17 (Halogens) | -1 |
18 (Noble gases) | 0 |
3-12 (Transition metals) | Variable |
Representing Ionic Compounds
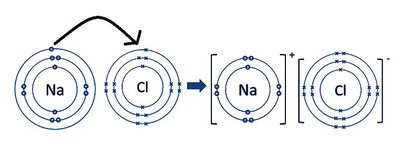
Bohr-Rutherford Diagrams
Bohr-Rutherford diagrams visually represent the arrangement of electrons in atoms and ions. These diagrams are useful for illustrating the transfer of electrons during ionic bond formation.
Show the nucleus and electron shells.
Indicate the movement of electrons from metal to nonmetal.

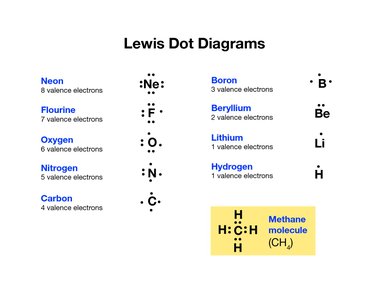
Lewis Dot Symbols and Diagrams
Lewis symbols represent the valence electrons of an atom as dots around the chemical symbol. Lewis diagrams can also show the transfer of electrons in ionic bonding and the resulting ions.
Each dot represents a valence electron.
For ions, brackets and charges are used to indicate the ion formed.
Naming and Writing Formulas for Ionic Compounds
Naming Ionic Compounds
The name of an ionic compound consists of the name of the metal (cation) followed by the name of the nonmetal (anion), with the anion's ending changed to "-ide." For compounds with polyatomic ions or multivalent metals, specific rules apply.
Metals keep their name (e.g., sodium).
Nonmetals change their ending to "-ide" (e.g., chloride).
For transition metals with multiple possible charges, the charge is indicated in Roman numerals (e.g., Iron(III) chloride).
Writing Chemical Formulas
To write the formula for an ionic compound:
Write the symbol of the cation first, then the anion.
Determine the charge of each ion.
Balance the charges so the total is zero (criss-cross method).
Reduce subscripts to the lowest whole numbers.
Example: Magnesium chloride is MgCl2 because Mg2+ and Cl- combine in a 1:2 ratio.
Polyatomic Ions
Polyatomic ions are groups of atoms bonded together that carry a charge. Most are anions, but some are cations. Common examples include sulfate (SO42-), nitrate (NO3-), and ammonium (NH4+).
Polyatomic ions have unique names and charges.
They combine with ions of opposite charge to form ionic compounds.
Structure and Properties of Ionic Compounds
Crystal Lattice Structure
Ionic compounds form a regular, repeating three-dimensional structure known as a crystal lattice. This arrangement maximizes the attractive forces between oppositely charged ions and minimizes repulsion.
The smallest repeating unit is called a formula unit.
Examples: NaCl, CaCO3, HgS.
Physical Properties of Ionic Compounds
Ionic compounds have characteristic physical properties due to their strong ionic bonds and crystal lattice structure.
Hard, brittle solids at room temperature.
High melting and boiling points.
Conduct electricity when melted or dissolved in water (electrolytes).
Soluble in water to varying degrees.

Summary Table: Types of Bonds and Key Properties
Bond Type | Participants | Electron Behavior | Physical State | Example |
|---|---|---|---|---|
Ionic | Metal + Nonmetal | Transferred | Solid (crystal) | NaCl |
Covalent | Nonmetal + Nonmetal | Shared | Liquid/Gas/Solid | H2O, CO2 |
Metallic | Metal + Metal | Delocalized | Solid | Cu, Fe |
Success Criteria
Explain chemical bonds and their types.
Determine if a compound is ionic or covalent.
Explain the octet rule and its importance.
Describe how ionic bonds are formed.
Identify neutral atoms, cations, and anions.
Use diagrams to represent ionic compound formation.
Explain the structure and properties of ionic compounds.
Name and write formulas for ionic compounds.
