 Back
BackIonic Compounds and Ion Formation: Structured Study Notes for General Chemistry
Study Guide - Smart Notes

Ions and Ionic Compounds
Introduction to Ions
Ions are charged particles formed when atoms gain or lose electrons. They play critical roles in biological and chemical processes, such as nerve signal transmission and maintaining cellular functions.
Cation: A positively charged ion formed by the loss of electrons.
Anion: A negatively charged ion formed by the gain of electrons.
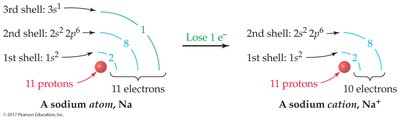
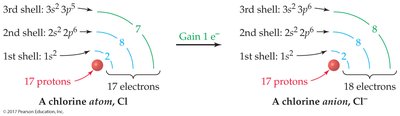
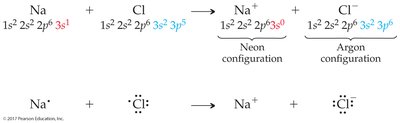
Example: Sodium (Na) loses one electron to become Na+; chlorine (Cl) gains one electron to become Cl–.

Formation of Ions: Electron Configurations
Atoms form ions to achieve stable electron configurations, often resembling noble gases. Alkali metals (group 1A) lose electrons, while halogens (group 7A) gain electrons.
Alkali metals: ns1 configuration; lose one electron to form cations.
Halogens: ns2np5 configuration; gain one electron to form anions.
The Octet Rule
The octet rule states that main group elements tend to react to achieve eight valence electrons, resulting in stable ions.
Metals: Lose electrons to form cations with noble gas configurations.
Nonmetals: Gain electrons to form anions with noble gas configurations.
Worked Examples: Electron Configurations and Ion Formation
Magnesium (Mg): Electron configuration: 1s22s22p63s2. Loses two electrons to form Mg2+ with a neon configuration.
Nitrogen (N): Electron configuration: 1s22s22p3. Gains three electrons to form N3– with a neon configuration.
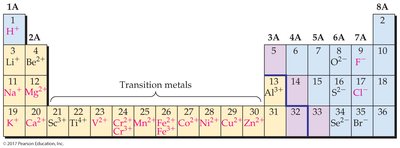
Periodic Properties and Ion Formation
Predicting Ionic Charges
The periodic table helps predict the charges of ions formed by main group elements.
Group 1A: M → M+ + e–
Group 2A: M → M2+ + 2e–
Group 6A: X + 2e– → X2–
Group 7A: X + e– → X–
Transition metals: Can form cations of multiple charges; octet rule not always followed.
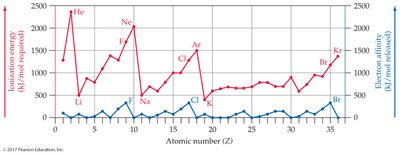
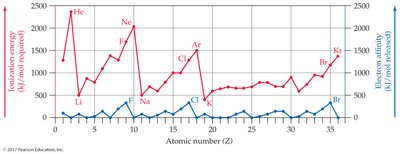
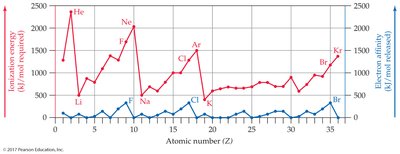
Ionization Energy and Electron Affinity
Ionization energy is the energy required to remove an electron; electron affinity is the energy released when an atom gains an electron. These properties influence ion formation.
Alkali metals: Low ionization energy, lose electrons easily.
Halogens: High electron affinity, gain electrons easily.
Noble gases: Do not form ions easily.
Naming Ions and Ionic Compounds
Naming Monoatomic Ions
Monoatomic ions are named based on their element and charge.
Main group cations: Element name + "ion" (e.g., potassium ion).
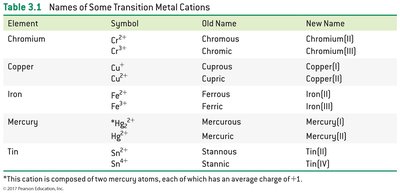
Transition metal cations: Old system (-ous, -ic) and new system (roman numerals).
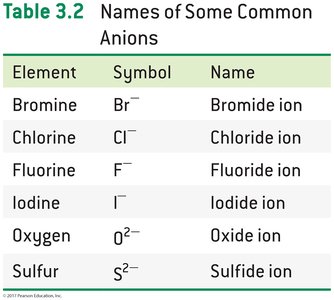
Anions: Element name ending replaced with "-ide" (e.g., chloride ion).
Polyatomic Ions
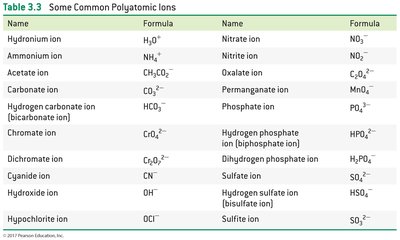
Polyatomic ions are composed of multiple atoms covalently bonded, carrying a net charge. Their names and formulas must be memorized.
Examples: Nitrate (NO3–), sulfate (SO42–), ammonium (NH4+).
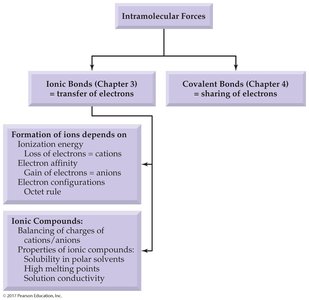
Ionic Bonds and Compound Formation
Nature of Ionic Bonds
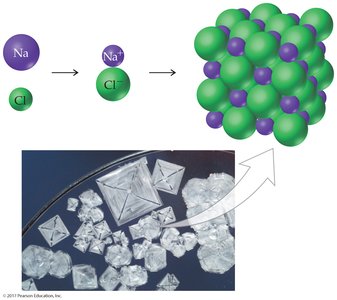
Ionic bonds are formed by the electrostatic attraction between oppositely charged ions. These bonds hold ions together in a crystal lattice, resulting in ionic solids.
Example: Sodium chloride (NaCl) forms a crystalline structure held by ionic bonds.

Formulas of Ionic Compounds
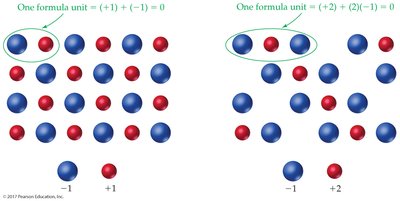
Ionic compounds are electrically neutral. The formula shows the ratio of cations to anions needed to balance charges.
Rule: List cation first, anion second; do not write charges; use parentheses for polyatomic ions with subscripts.
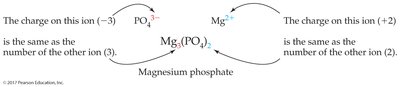
Example: Ca2+ and NO3– combine to form Ca(NO3)2.
Worked Examples: Writing Formulas
Sodium bromide: NaBr
Calcium bromide: CaBr2
Sodium sulfate: Na2SO4
Calcium phosphate: Ca3(PO4)2
Properties and Applications of Ionic Compounds
Physical Properties
Ionic compounds have distinct physical properties due to their strong ionic bonds.
High melting and boiling points: e.g., NaCl melts at 801 °C.
Electrical conductivity: Conduct electricity when dissolved in water.
Brittleness: Ionic solids shatter when struck.
Solubility: Many ionic compounds dissolve in water.

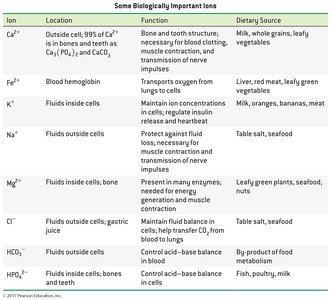
Biologically Important Ions
Many ions are essential for biological functions, such as nerve transmission, blood clotting, and maintaining fluid balance.
Ion | Location | Function | Dietary Source |
|---|---|---|---|
Ca2+ | Outside cells; bones and teeth | Bone structure, blood clotting | Milk, leafy vegetables |
Fe2+ | Blood hemoglobin | Oxygen transport | Liver, red meat |
K+ | Fluids inside cells | Maintain ion concentrations | Milk, bananas |
Na+ | Fluids outside cells | Fluid balance | Table salt |
Mg2+ | Fluids inside cells, bone | Enzyme function | Leafy plants, nuts |
Cl– | Fluids outside cells, gastric juice | Fluid balance | Table salt |
HCO3– | Fluids outside cells | Acid-base balance | Metabolism by-product |
HPO42– | Fluids inside cells, bones | Acid-base balance | Fish, poultry |
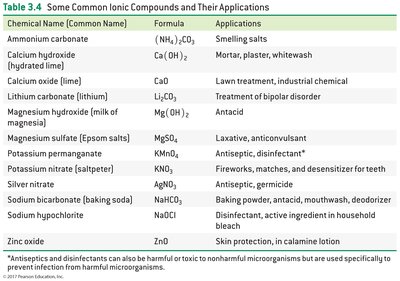
Common Ionic Compounds and Their Applications
Ionic compounds are widely used in everyday life and industry.
Chemical Name | Formula | Applications |
|---|---|---|
Ammonium carbonate | (NH4)2CO3 | Smelling salts |
Calcium hydroxide | Ca(OH)2 | Mortar, plaster |
Magnesium sulfate | MgSO4 | Laxative |
Potassium nitrate | KNO3 | Fireworks, matches |
Sodium bicarbonate | NaHCO3 | Baking powder |
Sodium chloride | NaCl | Table salt, bleach |
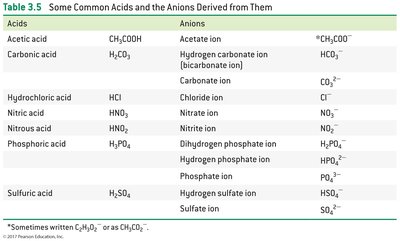
Acids and Bases: H+ and OH– Ions
Acids
An acid is a substance that provides H+ ions in water. The hydrogen cation (H+) is simply a proton, which forms a hydronium ion (H3O+) in water.
Example: Hydrochloric acid (HCl) provides one H+ per molecule.
Equation:
Bases
A base is a substance that provides OH– ions in water. The hydroxide ion is a polyatomic ion consisting of oxygen and hydrogen.
Example: Sodium hydroxide (NaOH) provides one OH– per formula unit.
Equation:
Acids and Their Derived Anions
Acid | Anion |
|---|---|
Acetic acid (CH3COOH) | Acetate ion (CH3COO–) |
Hydrochloric acid (HCl) | Chloride ion (Cl–) |
Sulfuric acid (H2SO4) | Sulfate ion (SO42–) |
Summary
Ionic compounds are formed by the transfer of electrons between metals and nonmetals, resulting in cations and anions held together by ionic bonds. Their properties, naming conventions, and biological importance are fundamental concepts in general chemistry.
