 Back
BackLiquids and Intermolecular Forces: States of Matter and Their Properties
Study Guide - Smart Notes

Liquids and Intermolecular Forces
Introduction to States of Matter
The physical state of a substance—gas, liquid, or solid—is determined by the balance between the kinetic energy of its particles and the strength of the intermolecular forces acting between them. Understanding these forces is essential for explaining the properties and behaviors of materials in different phases.
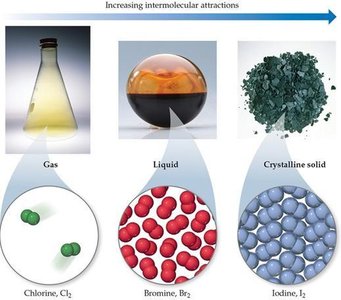
Gases: Particles are far apart, move freely, and have weak intermolecular forces.
Liquids: Particles are closer together, move past each other, and have moderate intermolecular forces.
Solids: Particles are tightly packed, vibrate in place, and have strong intermolecular forces.
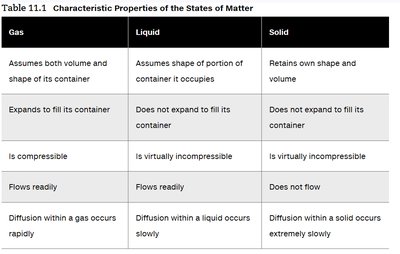
Gas | Liquid | Solid | |
|---|---|---|---|
Shape & Volume | Assumes both volume and shape of its container | Assumes shape of portion of container it occupies | Retains own shape and volume |
Compressibility | Is compressible | Is virtually incompressible | Is virtually incompressible |
Flow | Flows readily | Flows readily | Does not flow |
Diffusion | Diffusion within a gas occurs rapidly | Diffusion within a liquid occurs slowly | Diffusion within a solid occurs extremely slowly |
Intermolecular vs. Intramolecular Forces
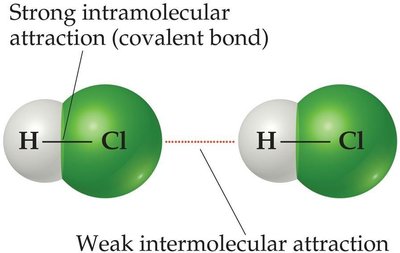
Intramolecular forces (chemical bonds) hold atoms together within a molecule, while intermolecular forces are the attractions between molecules. Intermolecular forces are generally much weaker than intramolecular forces, but they are crucial in determining the physical properties of substances.
Ionic bonds: Electrostatic attraction between ions (e.g., NaCl).
Covalent bonds: Sharing of electrons (e.g., H2O).
Metallic bonds: Delocalized electrons among metal atoms (e.g., Fe).

Physical properties such as boiling point, melting point, viscosity, and surface tension are determined by the strength of intermolecular forces.
Types of Intermolecular Forces
Dispersion Forces (London Dispersion Forces)
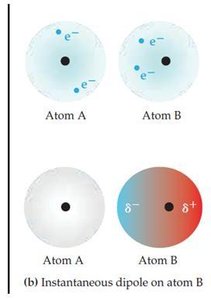
Dispersion forces are present in all molecules, whether polar or nonpolar. They arise from temporary fluctuations in electron distribution, creating instantaneous dipoles that induce dipoles in neighboring molecules. The strength of dispersion forces increases with molecular size and polarizability.
Polarizability: The ease with which the electron cloud of a molecule can be distorted. Larger, heavier atoms/molecules are more polarizable.
Dispersion forces are significant only when molecules are very close together.


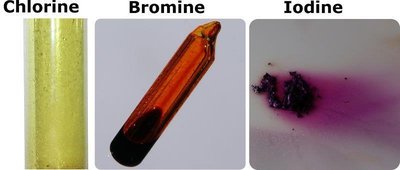
Example: The halogens at room temperature: Cl2 (gas), Br2 (liquid), I2 (solid) reflect increasing dispersion forces with increasing molecular size.
Dipole–Dipole Interactions
Dipole–dipole forces occur between polar molecules with permanent dipole moments. The positive end of one molecule is attracted to the negative end of another. These forces are stronger than dispersion forces for molecules of similar size and mass.
Effective only when molecules are very close together.
For molecules of similar mass, higher polarity leads to higher boiling points.
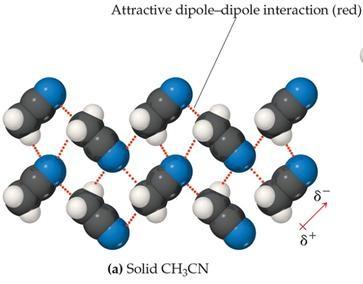
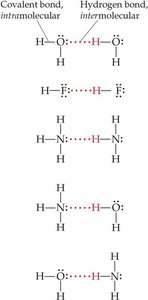
Hydrogen Bonding
Hydrogen bonding is a special, strong type of dipole–dipole interaction that occurs when hydrogen is bonded to highly electronegative atoms (N, O, or F). The nearly bare hydrogen nucleus interacts strongly with lone pairs on nearby electronegative atoms.
Hydrogen bonds are not true chemical bonds but are much stronger than typical dipole–dipole interactions.
Hydrogen bonding leads to anomalously high boiling points for compounds like H2O, NH3, and HF.
Ion–Dipole Interactions
Ion–dipole forces exist between an ion and a polar molecule. These are especially important in solutions of ionic compounds in polar solvents (e.g., NaCl in water). The strength of ion–dipole interactions increases with the charge of the ion and the magnitude of the dipole moment.
Properties of Liquids Affected by Intermolecular Forces
Viscosity
Viscosity is the resistance of a liquid to flow. It increases with stronger intermolecular forces and decreases with higher temperature.
Cohesion and Adhesion
Cohesive forces: Intermolecular forces binding similar molecules together.
Adhesive forces: Forces between different types of molecules (e.g., water and glass).
Surface Tension
Surface tension is the energy required to increase the surface area of a liquid. It results from extra inward forces on surface molecules, causing liquids to form droplets and "bead up" on nonpolar surfaces.



Capillary Action
Capillary action is the rise of liquids in narrow tubes due to the interplay of cohesive and adhesive forces. Water rises in glass due to strong adhesive forces, while mercury does not due to stronger cohesive forces.
Phase Changes and Energy
Phase Changes
Phase changes are transitions between solid, liquid, and gas states. They involve energy changes:
Melting (fusion): Solid to liquid
Freezing: Liquid to solid
Vaporization: Liquid to gas
Condensation: Gas to liquid
Sublimation: Solid to gas
Deposition: Gas to solid
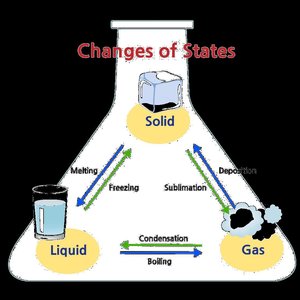
The energy required for these changes is quantified as heat of fusion, heat of vaporization, and heat of sublimation.
Heating Curves
A heating curve is a graph of temperature versus heat added. During a phase change, temperature remains constant as energy is used to change the phase rather than increase temperature. The specific heat capacity determines how much energy is needed to change the temperature of a substance.
Vapor Pressure and Boiling Point
Vapor Pressure
At any temperature, some molecules in a liquid have enough energy to escape into the gas phase, creating vapor pressure. As temperature increases, vapor pressure increases. When vapor pressure equals atmospheric pressure, the liquid boils.
The Clausius–Clapeyron equation relates vapor pressure and temperature:
= vapor pressure
= enthalpy of vaporization
= gas constant (8.314 J/mol·K)
= absolute temperature (K)
= constant
Phase Diagrams
Phase Diagrams
A phase diagram summarizes the conditions of temperature and pressure under which different phases of a substance exist. It shows the triple point (where all three phases coexist) and the critical point (beyond which the liquid and gas phases are indistinguishable).
For water, the melting curve has a negative slope, indicating that ice melts at lower temperatures under higher pressure.
For CO2, it sublimes at normal pressures because the triple point is above atmospheric pressure.
Liquid Crystals
Liquid Crystals
Some substances form liquid crystals, an intermediate phase between solid and liquid. Liquid crystals flow like liquids but retain some ordered structure of solids. They are often composed of rod-shaped molecules and have unique optical and flow properties.
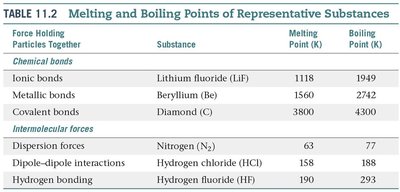
Summary Table: Melting and Boiling Points
Force Holding Particles Together | Substance | Melting Point (K) | Boiling Point (K) |
|---|---|---|---|
Ionic bonds | Lithium fluoride (LiF) | 1118 | 1949 |
Metallic bonds | Beryllium (Be) | 1560 | 2742 |
Covalent bonds | Diamond (C) | 3800 | 4300 |
Dispersion forces | Nitrogen (N2) | 63 | 77 |
Dipole-dipole interactions | Hydrogen chloride (HCl) | 158 | 188 |
Hydrogen bonding | Hydrogen fluoride (HF) | 190 | 293 |
Key Equations
Clausius–Clapeyron Equation:
Heat for temperature change:
Heat for phase change:
Conclusion
Understanding the nature and strength of intermolecular forces is essential for explaining the physical properties and behaviors of substances in different states. These concepts are foundational for further study in chemistry, including solution chemistry, thermodynamics, and materials science.
