 Back
BackMatter and Measurements: Foundations of General Chemistry
Study Guide - Smart Notes

Chemistry: The Central Science
Definition and Scope
Chemistry is the study of the nature, properties, and transformations of matter. It is considered the central science because it connects and underpins many other scientific disciplines, including biology, physics, environmental science, and medicine. - Matter: Anything that has mass and occupies space. - Scientific Method: The systematic process of observation, hypothesis, and experimentation used to expand scientific knowledge. - Property: A characteristic useful for identifying a substance or object, such as size, color, temperature, chemical composition, and chemical reactivity. 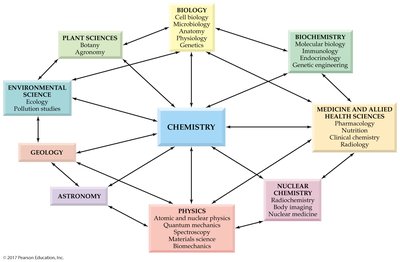
Chemical vs. Physical Changes
- Physical Change: Alters the form or appearance of matter without changing its chemical identity (e.g., melting ice, dissolving sugar in water). - Chemical Change: Results in the formation of new substances with different properties (e.g., burning sugar to make caramel). 
Type of Change | Description | Example |
|---|---|---|
Physical Change | No change in chemical makeup | Sugar dissolving in water |
Chemical Change | Change in chemical makeup; new substance formed | Sugar heated to caramel |
States of Matter
Solid, Liquid, and Gas
Matter exists in three primary states: - Solid: Definite shape and volume. - Liquid: Definite volume, shape conforms to container. - Gas: Neither definite shape nor volume; fills container. 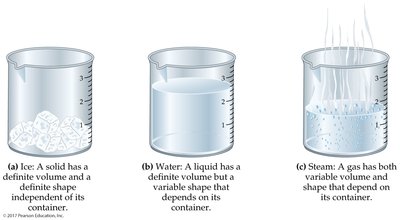 - Change of State: Conversion from one state to another (e.g., melting, boiling).
- Change of State: Conversion from one state to another (e.g., melting, boiling).
Classification of Matter
Pure Substances and Mixtures
Matter can be classified as either a pure substance or a mixture. - Pure Substance: Uniform chemical composition throughout. - Mixture: Blend of two or more substances, each retaining its chemical identity. 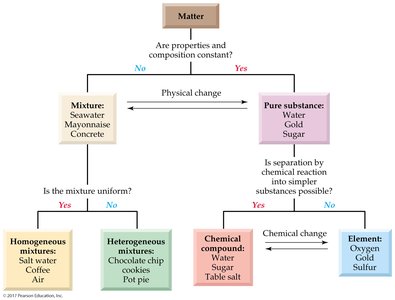
Homogeneous Mixture: Uniform composition (e.g., salt water).
Heterogeneous Mixture: Non-uniform composition (e.g., chocolate chip cookies).
Element: Fundamental substance that cannot be broken down chemically.
Chemical Compound: Pure substance that can be broken down into simpler substances by chemical reactions.
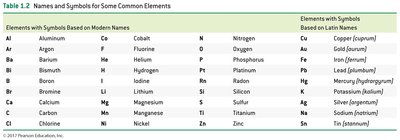
Chemical Elements and Symbols
Element Names and Symbols
Chemists use one- or two-letter symbols to represent elements. - The first letter is always capitalized; the second letter, if present, is lowercase. - Most symbols are based on English names; some are based on Latin names (e.g., Na for sodium).
Element | Symbol |
|---|---|
Aluminum | Al |
Carbon | C |
Sodium | Na |
Iron | Fe |
Gold | Au |
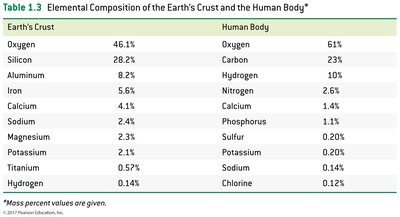
Elemental Abundance
Elements occur with varying abundance in the Earth's crust and the human body.
Element | Earth's Crust (%) | Human Body (%) |
|---|---|---|
Oxygen | 46.1 | 61 |
Silicon | 28.2 | — |
Carbon | — | 23 |
Hydrogen | 0.14 | 10 |
Chemical Formulas
Chemical formulas use element symbols and subscripts to show the number of atoms in a compound. - Example: (2 H atoms, 1 O atom), (1 C atom, 4 H atoms), (12 C atoms, 22 H atoms, 11 O atoms). 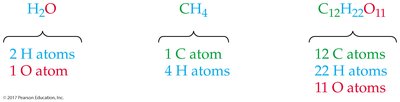
Chemical Reactions
Definition and Representation
A chemical reaction is a process in which the identity and composition of substances are changed. - Reactant: Starting substance. - Product: Substance formed as a result. - Chemical reactions are represented with reactants on the left, products on the right, and an arrow indicating the transformation. 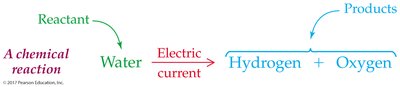

Physical Quantities: Units and Scientific Notation
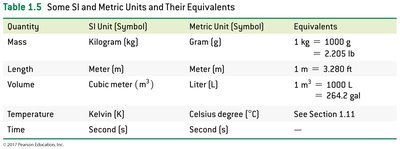
Measurement and Units
Physical quantities such as mass, volume, and temperature are described by both a number and a unit. - SI units: kilogram (kg), meter (m), cubic meter (m3), kelvin (K), second (s). - Metric units: gram (g), liter (L), Celsius degree (°C). 
Quantity | SI Unit | Metric Unit | Equivalents |
|---|---|---|---|
Mass | kg | g | 1 kg = 1000 g = 2.205 lb |
Length | m | m | 1 m = 3.280 ft |
Volume | m3 | L | 1 m3 = 1000 L |
Temperature | K | °C | See Section 1.11 |
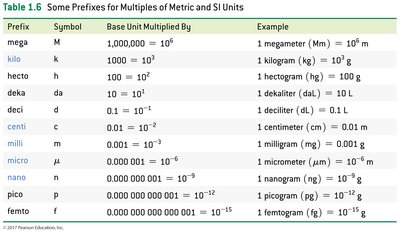
Prefixes and Scientific Notation
Metric and SI units can be modified by prefixes to refer to smaller or larger quantities.
Prefix | Symbol | Multiplier | Example |
|---|---|---|---|
kilo | k | 1000 | 1 kilogram (kg) = 103 g |
milli | m | 0.001 | 1 milligram (mg) = 10-3 g |
micro | μ | 0.000001 | 1 micrometer (μm) = 10-6 m |
nano | n | 0.000000001 | 1 nanogram (ng) = 10-9 g |
 - Scientific Notation: Expresses numbers as the product of a number between 1 and 10, times 10 raised to a power. Example:
- Scientific Notation: Expresses numbers as the product of a number between 1 and 10, times 10 raised to a power. Example: 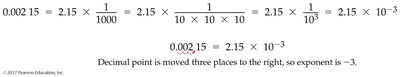
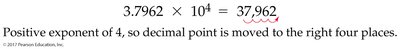
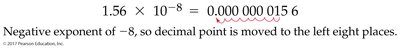
Measuring Mass, Length, and Volume
Mass vs. Weight
- Mass: Amount of matter in an object. - Weight: Gravitational force exerted on an object. 
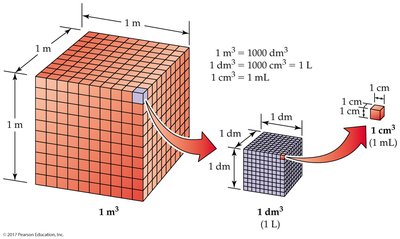
Length and Volume
- Meter: Standard unit of length. - Volume: Amount of space occupied; commonly measured in liters (L) or cubic centimeters (cm3). 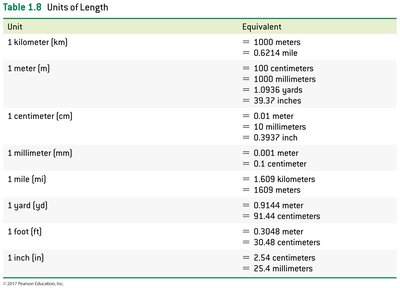
Measurement and Significant Figures
Precision and Uncertainty
Every measurement has a degree of uncertainty. - Significant Figures: The number of meaningful digits used to express a value. 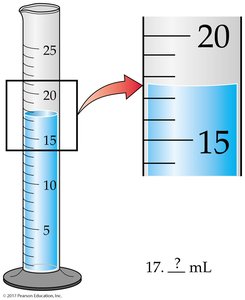
Rule 1: Zeroes in the middle of a number are significant.
Rule 2: Leading zeroes are not significant.
Rule 3: Trailing zeroes after a decimal point are significant.
Rule 4: Trailing zeroes before an implied decimal point may or may not be significant.
Rounding Off Numbers
Rules for Rounding
- In multiplication/division, the answer cannot have more significant figures than the original numbers.  - In addition/subtraction, the answer cannot have more digits after the decimal point than the original numbers.
- In addition/subtraction, the answer cannot have more digits after the decimal point than the original numbers. 
Problem Solving: Unit Conversions
Factor-Label Method
The factor-label method uses conversion factors to cancel unwanted units and obtain the desired units. - Conversion Factor: Expression of the numerical relationship between two units. 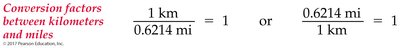

Temperature, Heat, and Energy
Temperature Scales
- Kelvin (K): SI unit; absolute zero is 0 K. - Celsius (°C): 0 °C is freezing point of water. - Fahrenheit (°F): 32 °F is freezing point of water. 
Energy and Specific Heat
- Energy: Capacity to do work or supply heat. - Specific Heat: Amount of heat needed to raise the temperature of 1 g of a substance by 1 °C.
Density and Specific Gravity
Definitions and Calculations
- Density: Mass per unit volume, usually expressed in g/cm3 or g/mL. - Specific Gravity: Density of a substance divided by the density of water at the same temperature.
Concept Map
Additional info: This study guide covers foundational concepts in general chemistry, including matter, measurement, classification, chemical reactions, units, significant figures, and basic calculations. It is suitable for exam preparation and introductory coursework.
