 Back
BackMolecular Compounds: Covalent Bonding, Structure, and Properties
Study Guide - Smart Notes

Covalent Bonds and Molecular Compounds
Nature of Covalent Bonds
Covalent bonds are chemical bonds formed by the sharing of electrons between atoms, typically nonmetals, to achieve stable electron configurations. Unlike ionic bonds, which involve the transfer of electrons, covalent bonds involve the mutual sharing of one or more pairs of electrons.
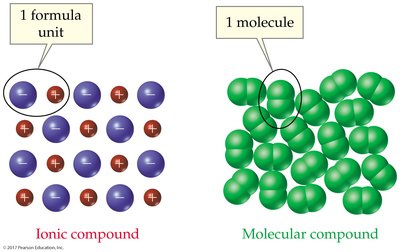
Molecule: A group of atoms held together by covalent bonds.
Octet Rule: Main group elements tend to react to achieve eight valence electrons (or two for hydrogen), resembling noble gas configurations.
Bond Length: The optimal distance between nuclei where attractive and repulsive forces balance.
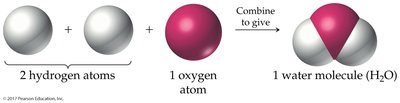
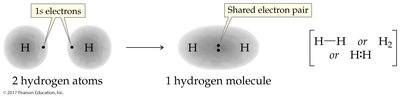
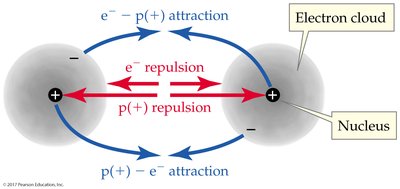
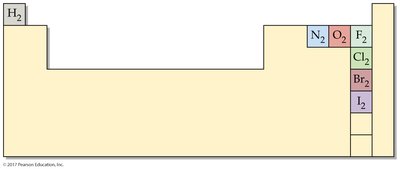
Diatomic Elements
Some elements naturally exist as diatomic molecules due to the stability provided by covalent bonding. These include hydrogen, nitrogen, oxygen, fluorine, chlorine, bromine, and iodine.
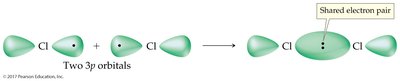
Covalent Bonds and the Periodic Table
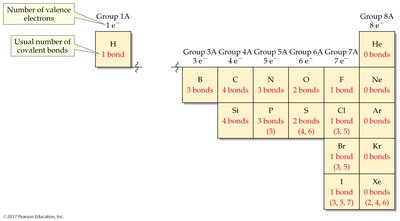
Predicting Number of Bonds
The number of covalent bonds an atom forms is generally determined by its group in the periodic table, as atoms seek to complete their valence shell.
Group 1A: 1 bond (e.g., H)
Group 4A: 4 bonds (e.g., C)
Group 5A: 3 bonds (e.g., N)
Group 6A: 2 bonds (e.g., O)
Group 7A: 1 bond (e.g., Cl)
Exceptions to the Octet Rule
Boron: Often forms only three bonds (six electrons).
Expanded Octet: Elements in the third period and below can have more than eight electrons due to available d orbitals (e.g., PCl5, SF6).

Multiple Covalent Bonds
Single, Double, and Triple Bonds
Atoms may share more than one pair of electrons to achieve octet configurations, resulting in single, double, or triple bonds.
Single Bond: One shared pair (e.g., H—H)
Double Bond: Two shared pairs (e.g., O=O)
Triple Bond: Three shared pairs (e.g., N≡N)
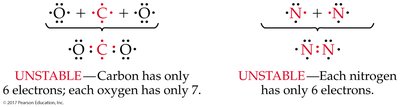
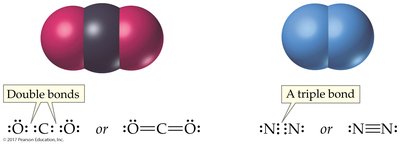
Common Elements in Multiple Bonds
Carbon, nitrogen, and oxygen frequently form multiple bonds.
Carbon forms four bonds, nitrogen three, and oxygen two, even in molecules with mu ltiple bonds.
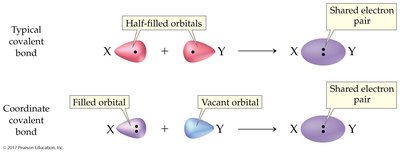
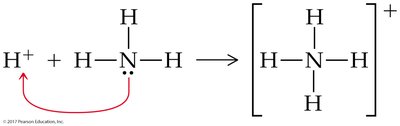
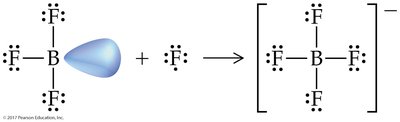
Coordinate Covalent Bonds
Definition and Examples
A coordinate covalent bond forms when both electrons in a shared pair originate from the same atom. Once formed, these bonds are indistinguishable from typical covalent bonds.
Common in polyatomic ions (e.g., NH4+, BF4-).
Characteristics of Molecular Compounds
Comparison with Ionic Compounds
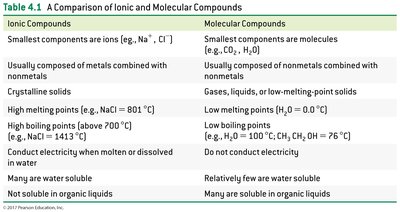
Molecular compounds differ from ionic compounds in structure, composition, and properties.
Ionic Compounds | Molecular Compounds |
|---|---|
Smallest components are ions | Smallest components are molecules |
Composed of metals and nonmetals | Composed of nonmetals |
Crystalline solids | Gases, liquids, or low-melting solids |
High melting/boiling points | Low melting/boiling points |
Conduct electricity in solution | Do not conduct electricity |
Many are water soluble | Few are water soluble |
Molecular Formulas and Lewis Structures
Definitions
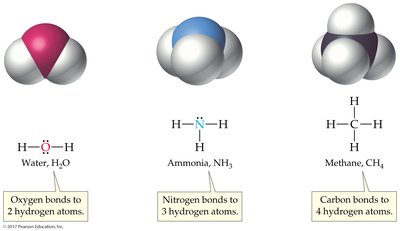
Molecular Formula: Indicates the number and type of atoms in a molecule (e.g., H2O).
Structural Formula: Shows how atoms are connected by bonds.
Lewis Structure: Shows both bonding pairs and lone pairs of electrons.
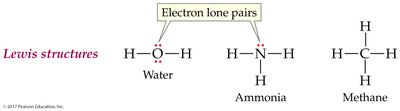
Drawing Lewis Structures
Follow these steps to draw Lewis structures:
Count total valence electrons (add for negative ions, subtract for positive ions).
Connect atoms with single bonds (lines).
Distribute remaining electrons as lone pairs to complete octets (except H).
Place extra electrons on the central atom if needed.
If the central atom lacks an octet, form multiple bonds as necessary.

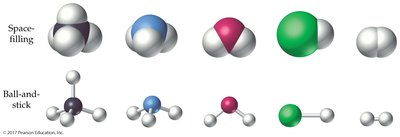
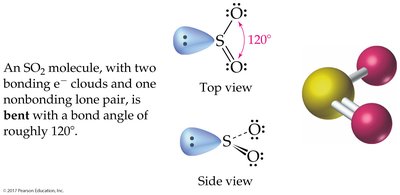
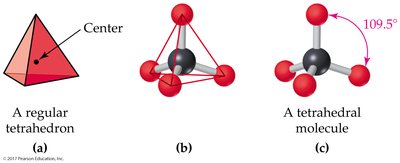
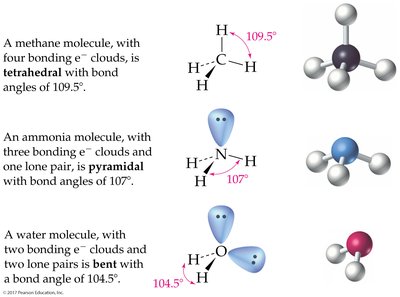
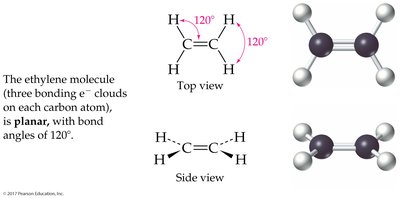
The Shapes of Molecules (VSEPR Theory)
Valence-Shell Electron-Pair Repulsion (VSEPR) Model
The VSEPR model predicts molecular geometry by considering the repulsion between electron charge clouds (bonding and lone pairs) around a central atom.
Draw the Lewis structure.
Count the number of charge clouds (bonds + lone pairs) around the central atom.
Predict the geometry that minimizes repulsion (maximizes separation).
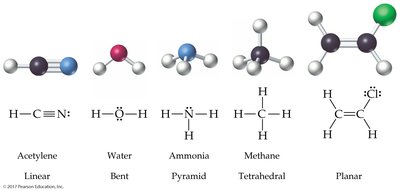
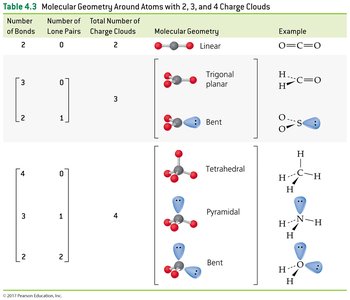
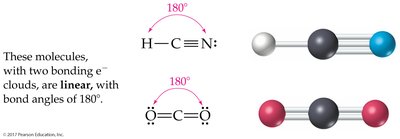
2 charge clouds: Linear (180°)
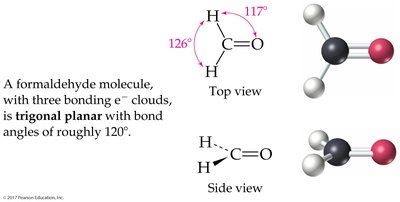
3 charge clouds: Trigonal planar (120°); bent if one is a lone pair
4 charge clouds: Tetrahedral (109.5°); pyramidal or bent if lone pairs are present
Polar Covalent Bonds and Electronegativity
Bond Polarity
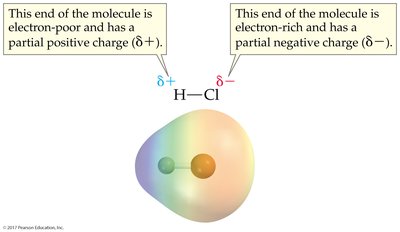
When atoms with different electronegativities form a bond, electrons are shared unequally, resulting in a polar covalent bond. The atom with higher electronegativity attracts electrons more strongly, acquiring a partial negative charge (δ−), while the other becomes partially positive (δ+).
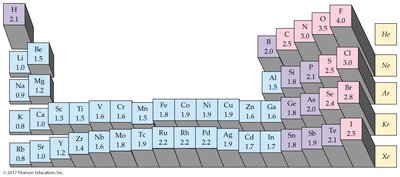
Electronegativity: A measure of an atom's ability to attract electrons in a bond. Fluorine is the most electronegative element (4.0).
Electronegativity decreases down a group and from right to left across a period.
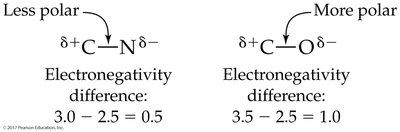
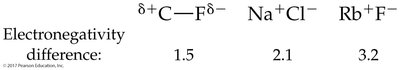
Bond Type by Electronegativity Difference
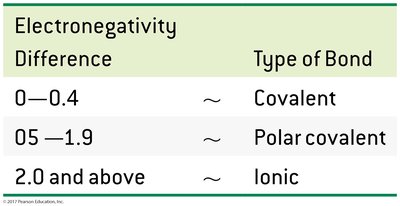
Electronegativity Difference | Type of Bond |
|---|---|
0–0.4 | Nonpolar covalent |
0.5–1.9 | Polar covalent |
2.0 and above | Ionic |
Polar Molecules
Molecular Polarity
A molecule is polar if it has a net dipole moment, resulting from the vector sum of individual bond dipoles and lone pair contributions. Molecular shape (geometry) is crucial in determining overall polarity.
Symmetrical molecules with polar bonds can be nonpolar overall if dipoles cancel.
Asymmetrical molecules with polar bonds are usually polar.
Naming Binary Molecular Compounds
Naming Rules
Name the first element in the formula, using a prefix if needed to indicate the number of atoms.
Name the second element, modifying its ending to –ide and using a numerical prefix as appropriate.
The prefix mono- is omitted for the first element unless needed for clarity.
Examples: CO2 is carbon dioxide, N2O3 is dinitrogen trioxide.
Summary Table: Common Prefixes for Naming
Number | Prefix |
|---|---|
1 | mono- |
2 | di- |
3 | tri- |
4 | tetra- |
5 | penta- |
6 | hexa- |
