 Back
BackMolecular Shapes, VSEPR Theory, and Molecular Polarity: Study Notes
Study Guide - Smart Notes

Chemical Bonding II: Molecular Shapes, VSEPR Theory, and Molecular Orbital Theory
Valence Shell Electron Pair Repulsion (VSEPR) Theory
VSEPR theory is a fundamental model used to predict the shapes of molecules based on the repulsions between electron groups around a central atom. The arrangement of these groups determines the geometry and bond angles of the molecule.
Electron groups include lone pairs, single, double, and triple bonds.
Electron groups repel each other due to coulombic forces, seeking maximum separation for stability.
The shape of the molecule is determined by the number and type of electron groups.
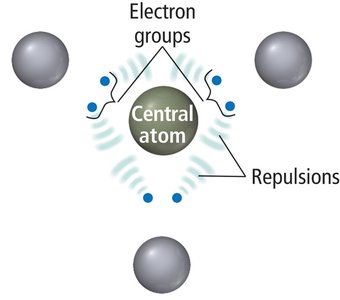
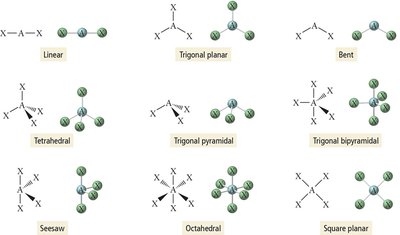
Basic Electron Group Geometries
There are five basic arrangements of electron groups around a central atom, each corresponding to a specific geometry and ideal bond angles.
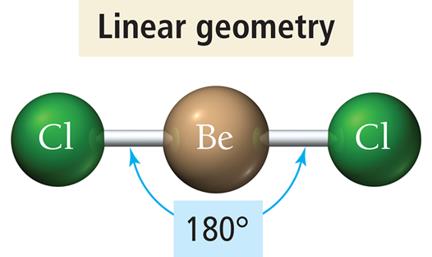
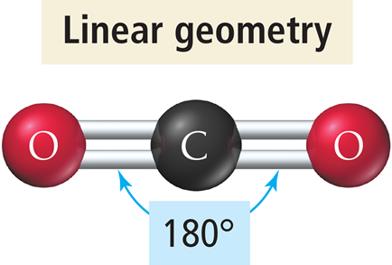
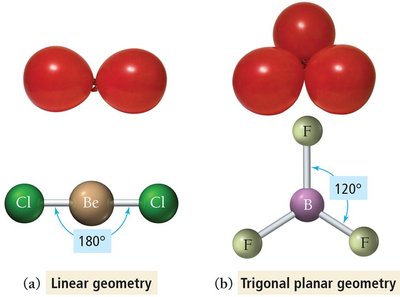
Linear Geometry
Two electron groups around the central atom.
Groups are positioned opposite each other, resulting in a bond angle of 180°.
Example: BeCl2, CO2.
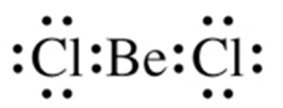
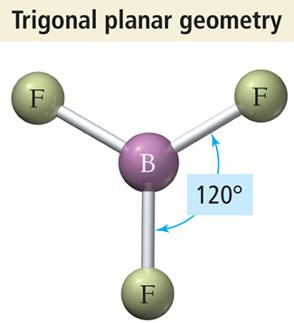
Trigonal Planar Geometry
Three electron groups around the central atom.
Groups form a flat triangle with bond angles of 120°.
Example: BF3, formaldehyde (CH2O).

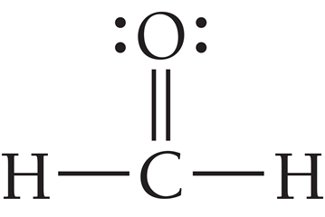
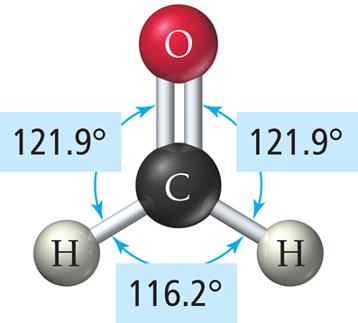
Comparison of Linear and Trigonal Planar Geometries
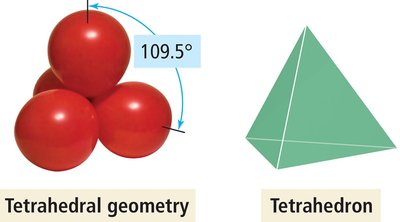
Tetrahedral Geometry
Four electron groups around the central atom.
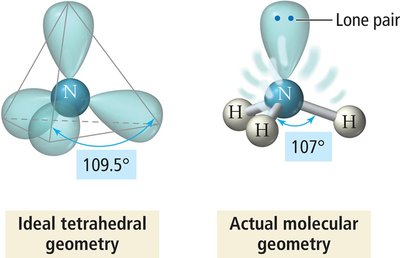
Groups form a tetrahedron with bond angles of 109.5°.
Example: CH4.
Effect of Lone Pairs on Molecular Geometry
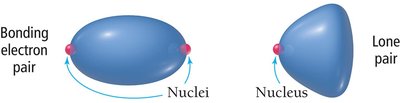
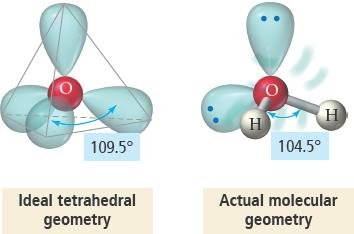
Lone pairs exert greater repulsion than bonding pairs, causing bond angles to deviate from ideal values and altering molecular geometry.
Lone pairs are more spread out, as they are attracted to only one nucleus.
Bonding pairs are attracted to two nuclei, making them less repulsive.
Repulsion order: lone pair–lone pair > lone pair–bonding pair > bonding pair–bonding pair.
Examples of Lone Pair Effects
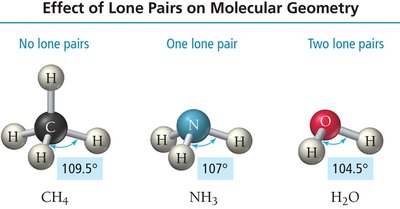
Trigonal pyramidal: Four electron groups, one lone pair (e.g., NH3).
Bent: Four electron groups, two lone pairs (e.g., H2O).
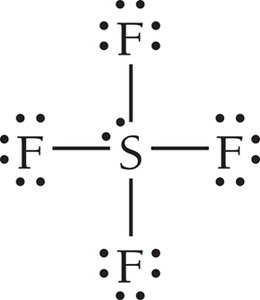
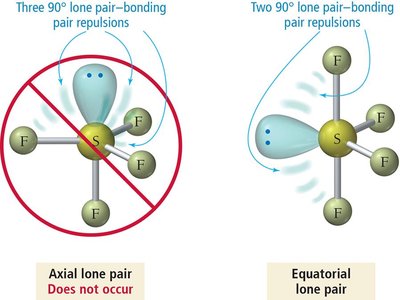
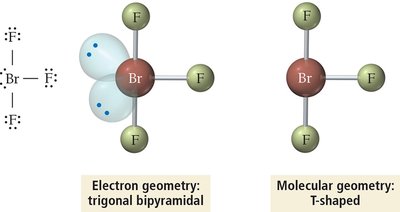
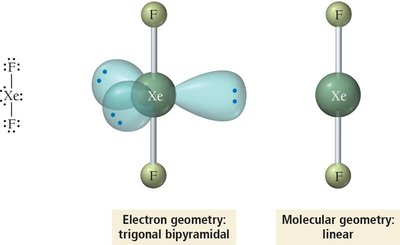
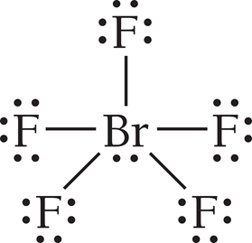
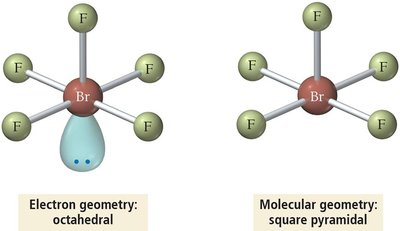
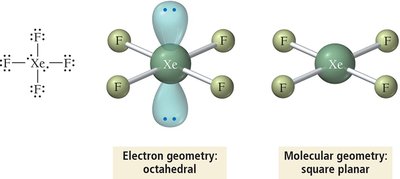
Summary Table: Electron and Molecular Geometries
The following table summarizes the relationship between electron groups, bonding groups, lone pairs, electron geometry, molecular geometry, and bond angles.
Electron Groups | Bonding Groups | Lone Pairs | Electron Geometry | Molecular Geometry | Approx. Bond Angles | Example |
|---|---|---|---|---|---|---|
2 | 2 | 0 | Linear | Linear | 180° | CO2 |
3 | 3 | 0 | Trigonal planar | Trigonal planar | 120° | BF3 |
3 | 2 | 1 | Trigonal planar | Bent | <120° | SO2 |
4 | 4 | 0 | Tetrahedral | Tetrahedral | 109.5° | CH4 |
4 | 3 | 1 | Tetrahedral | Trigonal pyramidal | <109.5° | NH3 |
4 | 2 | 2 | Tetrahedral | Bent | <109.5° | H2O |
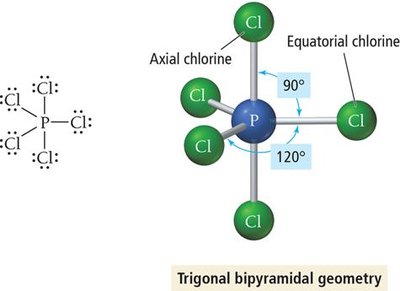
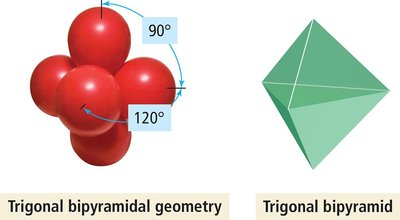
5 | 5 | 0 | Trigonal bipyramidal | Trigonal bipyramidal | 120°, 90° | PCl5 |
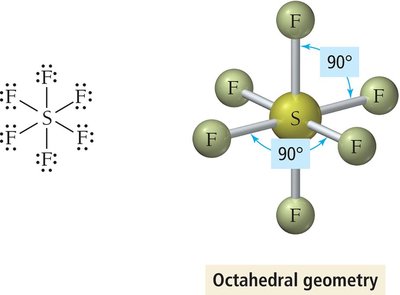
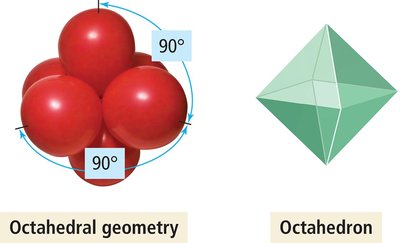
6 | 6 | 0 | Octahedral | Octahedral | 90° | SF6 |
Representing Three-Dimensional Shapes on Paper
To depict molecular geometry, chemists use conventions for bonds:
Straight line: bond in the plane of paper
Solid wedge: bond coming out of the page
Hashed wedge: bond going into the page

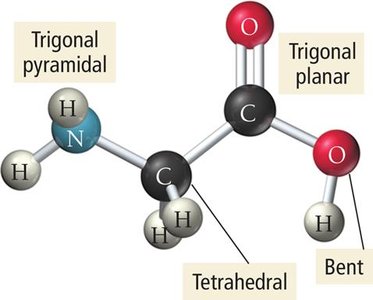
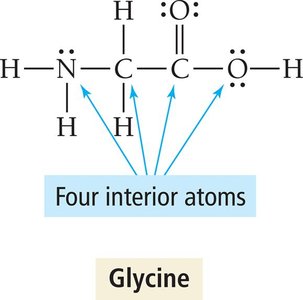
Molecules with Multiple Central Atoms
Complex molecules may have several interior atoms, each with its own geometry. The overall shape is described by analyzing each central atom in sequence.
Molecular Polarity
Molecular polarity arises from the presence of polar bonds and the overall shape of the molecule. Polarity affects properties such as solubility and intermolecular interactions.
A polar bond occurs when electrons are unequally shared between atoms of different electronegativities.
The net dipole moment depends on the vector sum of individual bond dipoles.
If dipoles cancel, the molecule is nonpolar; if they add, the molecule is polar.
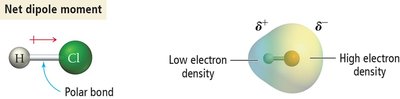
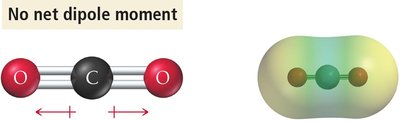
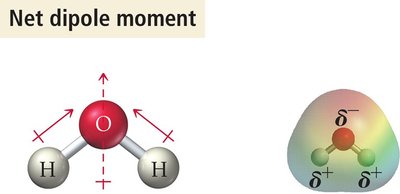
Determining Molecular Polarity: Steps
Draw the Lewis structure and determine molecular geometry.
Identify polar bonds and draw vectors toward more electronegative atoms.
Sum the vectors to determine if there is a net dipole moment.
Vector Addition in Molecular Polarity
Vector addition is used to determine the net dipole moment in molecules.
Vectors pointing in opposite directions cancel (nonpolar).
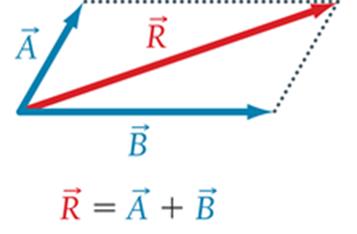
Vectors pointing in the same direction add (polar).
Three identical polar bonds in trigonal planar geometry cancel (nonpolar).

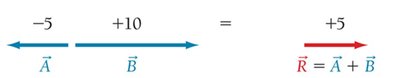
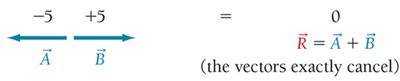
Molecular Polarity and Solubility
Polar molecules dissolve well in polar solvents like water, while nonpolar molecules do not. Some molecules have both polar and nonpolar regions, affecting their solubility and biological function.
Valence Bond Theory and Hybridization
Valence bond theory explains chemical bonding as the overlap of atomic or hybrid orbitals, with spin-pairing of electrons. Hybridization adjusts the number and orientation of orbitals to match observed molecular shapes.
Hybrid orbitals are formed by mixing atomic orbitals (e.g., s and p).
The type of hybridization depends on the electron group geometry (see Table below).
Hybridization schemes: sp (linear), sp2 (trigonal planar), sp3 (tetrahedral), sp3d (trigonal bipyramidal), sp3d2 (octahedral).
Electron Groups | Electron Geometry | Hybridization Scheme |
|---|---|---|
2 | Linear | sp |
3 | Trigonal planar | sp2 |
4 | Tetrahedral | sp3 |
5 | Trigonal bipyramidal | sp3d |
6 | Octahedral | sp3d2 |
Types of Bonds: Sigma (σ) and Pi (π)
σ bond: Formed by head-on overlap of orbitals along the internuclear axis; stronger and allows free rotation.
π bond: Formed by side-by-side overlap of unhybridized p orbitals; weaker and restricts rotation.
Molecular Orbital (MO) Theory
MO theory applies quantum mechanics to molecules, combining atomic orbitals to form molecular orbitals that are delocalized over the entire molecule.
Bonding molecular orbitals (constructive combination) are lower in energy.
Antibonding molecular orbitals (destructive combination) are higher in energy.
Bond order =
Paramagnetic molecules have unpaired electrons in MO diagrams; diamagnetic molecules have all electrons paired.
Summary and Applications
VSEPR theory predicts molecular shapes based on electron group repulsions.
Molecular geometry and polarity influence physical and chemical properties.
Valence bond and MO theories provide deeper understanding of bonding and molecular behavior.
