 Back
BackMolecules and Compounds: Polyatomic Ions and Compound Naming
Study Guide - Smart Notes

Ch.3 - Molecules and Compounds
Polyatomic Ions
Polyatomic ions are groups of atoms covalently bonded together that carry an overall charge. These ions are fundamental in general chemistry, as they frequently appear in ionic compounds and acids.
Definition: A polyatomic ion is a charged species (ion) composed of several atoms bound together.
Example: The nitrate ion, NO3-, is a polyatomic ion.
Charge: Polyatomic ions can be either positively or negatively charged, but most are anions (negatively charged).
Common Polyatomic Cations: NH4+ (ammonium) and Hg22+ (mercury(I)).
Polyatomic Oxyanions
Oxyanions are polyatomic ions containing oxygen. Their names and formulas follow systematic patterns based on the number of oxygen atoms present.
Trioxides: Ions ending with -ate typically have three oxygens (e.g., nitrate, NO3-).
Tetraoxides: Ions ending with -ate may have four oxygens (e.g., sulfate, SO42-).
Deriving Oxyanions: Decreasing the number of oxygens by one changes the ending to -ite (e.g., nitrite, NO2-).
Prefixes: per- and hypo- are used for ions with more or fewer oxygens, respectively.

Halogen Oxyanions
Halogen oxyanions are polyatomic ions containing halogens (F, Cl, Br, I) and oxygen. Their names are based on the halogen's base name and the number of oxygens.
Base Name: The root of the halogen's name (e.g., chlor- for chlorine).
Charge: All halogen oxyanions possess a charge of -1.
Naming: The number of oxygens determines the prefix and suffix:
per-...ate: Most oxygens (e.g., perchlorate, ClO4-)
...ate: Standard number (e.g., chlorate, ClO3-)
...ite: One fewer oxygen (e.g., chlorite, ClO2-)
hypo-...ite: Least oxygens (e.g., hypochlorite, ClO-)

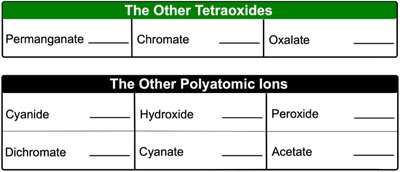
Other Polyatomic Ions
Some polyatomic ions do not fit predictable patterns and must be memorized. These include ions like cyanide, hydroxide, peroxide, dichromate, and acetate.
Cyanide: CN-
Hydroxide: OH-
Peroxide: O22-
Dichromate: Cr2O72-
Acetate: CH3COO- or C2H3O2-
Naming Ionic Compounds
Ionic compounds consist of a positive ion (cation) and a negative ion (anion). The naming follows a systematic approach:
Cation: The metal keeps its name and is written first. If the metal can have multiple charges, a Roman numeral is used to indicate its charge (e.g., iron(III) chloride).
Anion: The nonmetal keeps its base name but changes its ending to -ide (e.g., chloride).
Polyatomic Ion: If present, the polyatomic ion keeps its name (e.g., sodium nitrate).
Writing Ionic Compound Formulas
To write the formula for an ionic compound:
Step 1: Write the ions involved.
Step 2: Combine the ions so that the total positive and negative charges balance to zero.
Rule: If the charges are the same, simply combine the elements. If different, use subscripts to balance the charges.
Example: Magnesium sulfate:
Naming Ionic Hydrates
Ionic hydrates are ionic compounds linked to water molecules. The number of water molecules is indicated by a numerical prefix.
Step 4: The water portion is called "hydrate" and is preceded by a prefix (e.g., trihydrate for three water molecules).
Example: is named potassium dichromate trihydrate.
Naming Acids
Acids are covalent compounds beginning with hydrogen. They are classified as binary acids or oxyacids.
Binary Acids: Contain H+ and a nonmetal anion (not oxygen). Named with "hydro-" prefix and "-ic" suffix (e.g., hydrochloric acid).
Oxyacids: Contain H+ and a polyatomic ion with oxygen. If the ion ends in "-ate", the acid ends in "-ic"; if "-ite", the acid ends in "-ous" (e.g., sulfuric acid from sulfate, sulfurous acid from sulfite).
Naming Molecular (Covalent) Compounds
Molecular compounds contain only nonmetals. Numerical prefixes indicate the number of each element.
First Nonmetal: Named normally, uses all prefixes except "mono-".
Second Nonmetal: Uses any prefix and changes ending to "-ide".
Example: is dinitrogen tetroxide.
Numerical Prefixes: mono (1), di (2), tri (3), tetra (4), penta (5), hexa (6), hepta (7), octa (8), nona (9), deca (10).
Summary Table: Polyatomic Ion Naming Patterns
Halogen | Base Name |
|---|---|
Fluorine (F) | fluor- |
Chlorine (Cl) | chlor- |
Bromine (Br) | brom- |
Iodine (I) | iod- |
# of Oxygens | Base Name |
|---|---|
Most | per___ate |
Standard | ___ate |
One fewer | ___ite |
Least | hypo___ite |
The Other Tetraoxides | |
|---|---|
Permanaganate | MnO4- |
Chromate | CrO42- |
Oxalate | C2O42- |
The Other Polyatomic Ions | |
|---|---|
Cyanide | CN- |
Hydroxide | OH- |
Peroxide | O22- |
Dichromate | Cr2O72- |
Cyanate | OCN- |
Acetate | CH3COO- |
