 Back
BackPeriodic Trends: Melting Point and Structure in the Periodic Table
Study Guide - Smart Notes

Periodic Trends
Melting Point and Atomic Structure
The melting point of elements varies systematically across the periodic table and is closely related to the type of atomic structure and bonding present in each element. Understanding these trends helps explain the physical properties of elements and their chemical behavior.
Types of Atomic Structures
Giant Metallic Structure: Found in metals, where atoms are arranged in a lattice and valence electrons are delocalized, forming a 'sea of electrons' that holds the metal ions together.
Giant Covalent Structure: Atoms are bonded by strong covalent bonds in a continuous network (e.g., diamond, graphite).
Simple Molecular Structure: Elements or compounds consist of discrete molecules held together by weak intermolecular forces (e.g., O2, S8).
Melting Point Trends in the Periodic Table
The melting point of elements shows periodic trends that can be explained by their atomic structure and bonding:
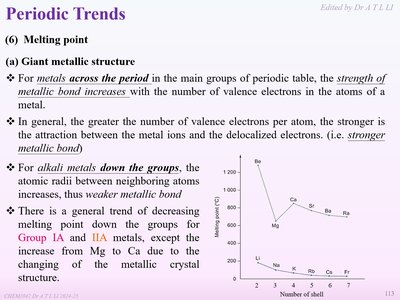
Across a Period: In the main groups, the strength of metallic bonding increases with the number of valence electrons per atom, leading to higher melting points. This is because more delocalized electrons strengthen the attraction between metal ions and electrons.
Down a Group (Alkali Metals): The atomic radius increases, so the metallic bond becomes weaker, resulting in a decrease in melting point down the group. An exception is observed from Mg to Ca due to changes in crystal structure.
Example: Sodium (Na) has a lower melting point than magnesium (Mg) because Mg has more valence electrons, resulting in stronger metallic bonding.
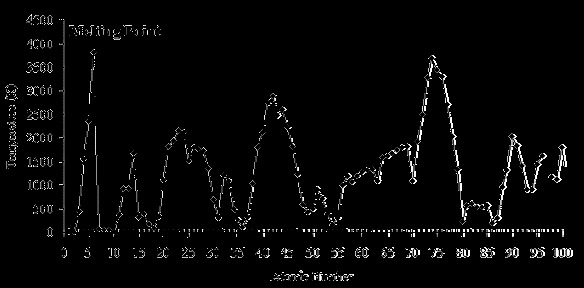
Graphical Representation of Melting Points
The melting points of elements, when plotted against atomic number, show periodic peaks and troughs. Peaks often correspond to elements with strong bonding (e.g., carbon, tungsten), while troughs correspond to elements with weak intermolecular forces (e.g., noble gases).
Class Practice: Periodic Trends in Period 3 Elements
Students are often asked to predict and explain periodic trends for elements in a period, such as:
Electronegativity: Increases across a period due to increasing nuclear charge and decreasing atomic radius.
Atomic Radii: Decreases across a period as the effective nuclear charge increases, pulling electrons closer to the nucleus.
Melting Point: Generally increases from Group 1 to Group 14 (due to stronger metallic/covalent bonding), then decreases towards Group 18 (due to weaker molecular forces).
Physical Properties of Allotropes of Carbon
Allotropes are different structural forms of the same element. Carbon has several allotropes, each with distinct physical properties:
Graphene: Single layer of carbon atoms in a hexagonal lattice; excellent conductor of electricity.
Graphite: Layers of carbon atoms; good conductor due to delocalized electrons between layers.
Buckminsterfullerene (Fullerene): Molecules of 60 carbon atoms (C60) arranged in a spherical shape; poor conductor and has a low melting point compared to other allotropes.
Carbon Nanotube: Cylindrical tubes of carbon atoms; excellent conductor and very strong.
Example: Buckminsterfullerene has the lowest melting point and the poorest electrical conductivity among the listed allotropes due to its molecular structure and lack of delocalized electrons.

Summary Table: Melting Point and Conductivity of Carbon Allotropes
Allotrope | Melting Point | Electrical Conductivity |
|---|---|---|
Graphene | Very high | Excellent |
Graphite | High | Good |
Buckminsterfullerene | Low | Poor |
Carbon Nanotube | Very high | Excellent |
Key Equations and Concepts
Metallic Bond Strength: Increases with the number of delocalized electrons per atom.
Melting Point Trend (Metals):
Atomic Radius Trend: across a period, down a group
