 Back
BackPeriodicity and Chemical Reactions: Structure, Trends, and Types of Reactions
Study Guide - Smart Notes

Periodicity and the Periodic Table
Classification of Elements
The periodic table organizes elements based on atomic number and recurring chemical properties. Elements are grouped into categories with similar properties, which helps predict their behavior in chemical reactions.
Main group elements (Groups 1A–8A): Also called representative elements, these have predictable properties and their valence electrons are in s or p orbitals.
Transition metals (Groups 3B–2B): These elements have d electrons and show variable oxidation states.
Inner transition metals: Lanthanides and actinides, with f electrons.
Group 2B elements (Zn, Cd, Hg, Cn): Often classified as transition metals but do not exhibit typical transition metal characteristics.
Groups 1A and 2A: Alkali and Alkaline Earth Metals
Alkali metals (Group 1A) and alkaline earth metals (Group 2A) are highly reactive metals, especially with water, and form characteristic cations.
Alkali metals: Li, Na, K, Rb, Cs, Fr; form +1 cations, react violently with water.
Alkaline earth metals: Be, Mg, Ca, Sr, Ba, Ra; form +2 cations, less reactive than alkali metals.


Groups 7A and 8A: Halogens and Noble Gases
Halogens (Group 7A) are highly reactive nonmetals, forming diatomic molecules and -1 anions. Noble gases (Group 8A) are inert, existing as monoatomic gases and rarely forming compounds.
Halogens: F, Cl, Br, I; reactivity decreases down the group; form ionic and molecular compounds.
Noble gases: He, Ne, Ar, Kr, Xe, Rn; very low reactivity due to full valence shells.


Formation of Ions and Lewis Dot Symbols
Ion Formation and Isoelectronic Species
Atoms form ions to achieve stable electron configurations, often matching the nearest noble gas (octet or duet rule). Metals lose electrons to form cations; nonmetals gain electrons to form anions.
Duet rule: Stability with 2 valence electrons (e.g., He configuration).
Octet rule: Stability with 8 valence electrons (e.g., Ne, Ar configuration).
Isoelectronic species: Ions/atoms with the same electron configuration.

Types of Chemical Reactions
Combination and Decomposition Reactions
Combination reactions involve two or more substances forming a single product. Decomposition reactions are the reverse, breaking a compound into simpler substances, often requiring energy input.
Combination: A + B → C (e.g., 2Na + Cl2 → 2NaCl)
Decomposition: C → A + B (e.g., 2HgO → 2Hg + O2)



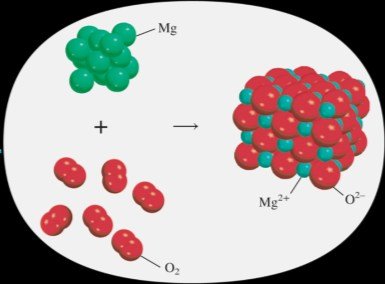

Oxidation-Reduction (Redox) Reactions
Redox reactions involve the transfer of electrons, changing the oxidation states of elements. Oxidation is the loss of electrons; reduction is the gain of electrons. Mnemonics: LEO (Loss of Electrons is Oxidation), GER (Gain of Electrons is Reduction).
Oxidizing agent: Accepts electrons, gets reduced.
Reducing agent: Donates electrons, gets oxidized.


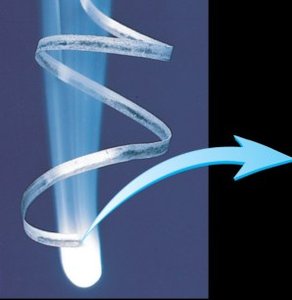
Combustion Reactions
Combustion is a redox reaction where a substance reacts with oxygen, releasing energy, carbon dioxide, and water. It is essential in biological and industrial processes.
General form: CxHyOz + O2 → CO2 + H2O + heat
Reactions in Aqueous Solutions
Types of Displacement and Double Displacement Reactions
Single displacement reactions involve one element replacing another in a compound. Double displacement reactions involve the exchange of ions between two compounds, often forming a precipitate or water.
Single displacement: A + BC → AC + B
Double displacement: AB + CD → AD + CB
Precipitation and Acid-Base Reactions
Precipitation reactions form an insoluble product (precipitate) when two aqueous solutions are mixed. Acid-base reactions (neutralizations) produce water and a salt.
Precipitation: Formation of an insoluble compound from soluble reactants.
Acid-base: HA + BOH → H2O + BA
Periodic Trends in Physical Properties
Effective Nuclear Charge (Zeff)
The effective nuclear charge is the net positive charge experienced by valence electrons, accounting for shielding by inner electrons. It increases across a period and slightly down a group.
Formula:
Atomic and Ionic Radii
Atomic radius decreases across a period (due to increasing Zeff) and increases down a group (due to higher principal quantum number n). Cations are smaller than their parent atoms; anions are larger.
Trend: Left to right: decreases; top to bottom: increases.
Ionization Energy
Ionization energy is the energy required to remove an electron from a gaseous atom. It increases across a period and decreases down a group. Exceptions occur due to electron configurations and subshell stability.
First ionization energy:
Electron Affinity and Electronegativity
Electron affinity is the energy change when an atom gains an electron. Electronegativity is the tendency of an atom in a bond to attract electrons. Both generally increase across a period and decrease down a group.
Electron affinity:
Electronegativity: Relative scale (Pauling scale) for bonded atoms.
Summary Table: Periodic Trends
Property | Trend Across Period | Trend Down Group |
|---|---|---|
Atomic radius | Decreases | Increases |
Ionization energy | Increases | Decreases |
Electron affinity | Increases | Decreases |
Electronegativity | Increases | Decreases |
Key Definitions
Oxidation number: The hypothetical charge an atom would have if all bonds were ionic.
Precipitate: An insoluble solid formed from a reaction in solution.
Spectator ion: An ion that does not participate in the actual chemical change.
Isoelectronic: Species with the same number of electrons.
