 Back
BackPreparation and Reactions of Alkyl Halides: Free Radical Halogenation and SN2 Mechanism
Study Guide - Smart Notes

Preparation of Alkyl Halides
Free Radical Halogenation of Alkanes
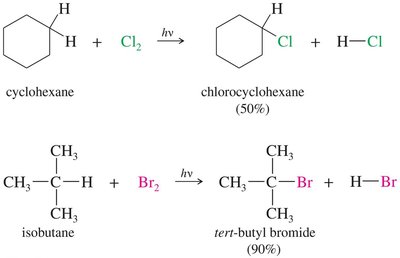
Alkyl halides can be synthesized by the halogenation of alkanes, a process that involves the substitution of a hydrogen atom with a halogen atom via a free radical mechanism. The selectivity and efficiency of this reaction depend on the halogen used.
Chlorination produces a mixture of products due to low selectivity. It is mainly useful for alkanes where all hydrogens are equivalent.
Bromination is highly selective, favoring the substitution at more substituted carbon atoms: 3° > 2° > 1°.
Example: Chlorination of cyclohexane and bromination of isobutane illustrate the difference in selectivity and product distribution.
Free Radical Allylic Halogenation
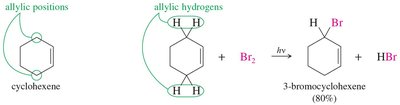
Allylic halogenation targets the carbon atom adjacent to a double bond (the allylic position). This process is especially efficient with bromine due to the resonance stabilization of the allylic radical intermediate.
Allylic radicals are stabilized by resonance, making allylic halogenation highly selective and efficient.
Bromination at the allylic position yields high product selectivity and yield.
Example: Bromination of cyclohexene at the allylic position forms 3-bromocyclohexene.
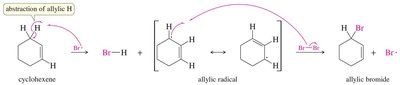
Mechanism of Allylic Bromination
The mechanism involves the abstraction of an allylic hydrogen atom to form a resonance-stabilized allylic radical, which then reacts with bromine to yield the allylic bromide.
Both possible allylic radicals can react with bromine, leading to product formation at the allylic position.
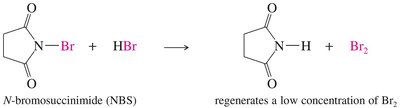
N-Bromosuccinimide (NBS) as an Allylic Brominating Agent
N-Bromosuccinimide (NBS) is commonly used for allylic bromination because it maintains a low, steady concentration of Br2 in the reaction mixture, preventing unwanted side reactions.
NBS reacts with HBr (formed during bromination) to regenerate Br2 in situ.
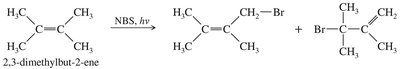
Example: Allylic bromination of 2,3-dimethylbut-2-ene with NBS and light yields the corresponding allylic bromide.
Reactions of Alkyl Halides
The SN2 Reaction (Bimolecular Nucleophilic Substitution)
The SN2 reaction is a fundamental mechanism in organic chemistry where a nucleophile replaces a leaving group (such as a halide) in a single, concerted step. This reaction is second order overall, depending on the concentrations of both the alkyl halide and the nucleophile.
Mechanism: The nucleophile attacks the electrophilic carbon from the opposite side of the leaving group, resulting in inversion of configuration at the carbon center.
Rate Law:
Leaving Group: The halogen atom (e.g., I−) departs with its electron pair.
Example: Hydroxide ion reacts with iodomethane to produce methanol and iodide ion.
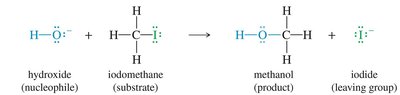
SN2 Reaction Mechanism
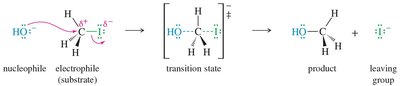
The SN2 mechanism is concerted, meaning bond formation and bond breaking occur simultaneously through a single transition state. The nucleophile approaches the substrate from the side opposite the leaving group, leading to a stereochemical inversion (Walden inversion).
Transition State: Both the nucleophile and leaving group are partially bonded to the central carbon in the transition state.
Product: The nucleophile replaces the leaving group, and the configuration at the carbon is inverted.
Additional info: The SN2 reaction is most efficient with primary alkyl halides and less hindered substrates. Tertiary alkyl halides are generally unreactive in SN2 reactions due to steric hindrance.
