 Back
BackQuantum Numbers, Atomic Orbitals, and Electron Configurations
Study Guide - Smart Notes

Quantum Numbers and Atomic Orbitals
Principal Quantum Number (n) and Shell Structure
The principal quantum number (n) determines the main energy level or shell of an electron in an atom. Each shell contains one or more subshells, and each subshell contains one or more orbitals.
n = 1: One subshell (1s), one orbital
n = 2: Two subshells (2s, 2p), four orbitals
n = 3: Three subshells (3s, 3p, 3d), nine orbitals
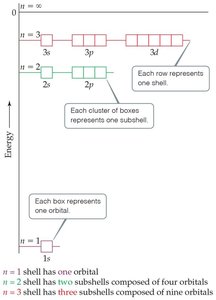
Each box represents one orbital, and each cluster of boxes represents a subshell.
Degeneracy and Energy Levels in Hydrogen and Multi-Electron Atoms
In a hydrogen atom (one electron), all orbitals with the same principal quantum number (n) have the same energy and are called degenerate orbitals. In multi-electron atoms, electron-electron repulsion causes energy splitting, so not all orbitals with the same n are degenerate. However, orbitals within the same subshell (same l) remain degenerate.
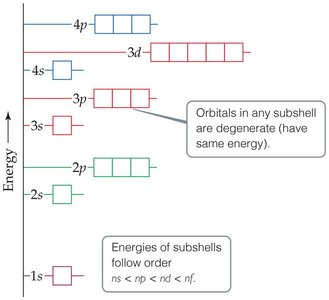
Order of subshell energies:
Quantum Numbers Overview
Principal quantum number (n): Energy level (n = 1, 2, 3, ...)
Angular momentum quantum number (l): Subshell type (l = 0 to n-1; s, p, d, f...)
Magnetic quantum number (m_l): Orientation of orbital (m_l = -l to +l)
Spin quantum number (m_s): Electron spin (+1/2 or -1/2)
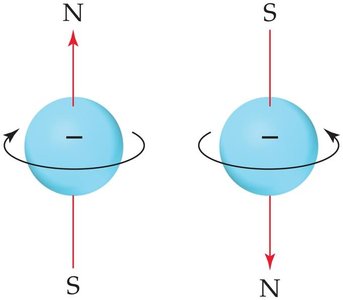
Spin quantum number describes the two possible orientations of an electron's magnetic field.
Pauli Exclusion Principle
The Pauli Exclusion Principle states that no two electrons in the same atom can have the same set of four quantum numbers. This means each orbital can hold a maximum of two electrons with opposite spins.
General Rules for Assigning Electrons to Atomic Orbitals
Each shell (n) contains n subshells.
Each subshell (l) contains (2l + 1) orbitals.
Each orbital can hold up to two electrons.
Total number of orbitals in a shell:
Maximum electrons in a shell:
Electron Configurations
Notation and Aufbau Principle
The electron configuration of an atom describes the distribution of electrons among the orbitals. The Aufbau principle states that electrons fill orbitals in order of increasing energy.
Notation: n (energy level), l (orbital type), superscript (number of electrons)
Example: 4p5 means 5 electrons in the 4p orbitals
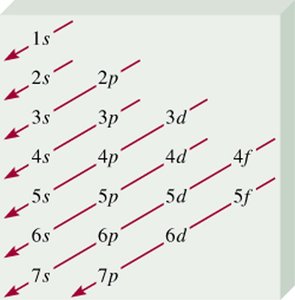
Order of filling: 1s < 2s < 2p < 3s < 3p < 4s < 3d < 4p < 5s < 4d < 5p < 6s ...
Orbital Diagrams and Hund's Rule
Orbital diagrams use boxes to represent orbitals and arrows for electrons. Hund's Rule states that electrons occupy degenerate orbitals singly with parallel spins before pairing up.
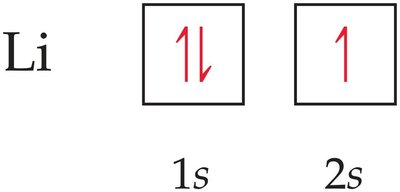
Example: Lithium (Li) has the configuration 1s22s1.
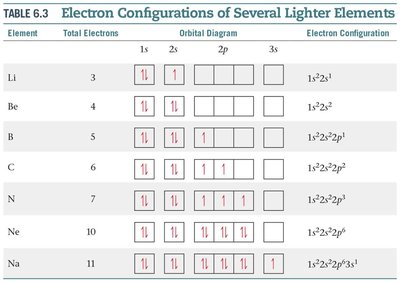
Electron Configurations of Lighter Elements
The table below summarizes the electron configurations and orbital diagrams for several lighter elements:
Element | Total Electrons | Orbital Diagram | Electron Configuration |
|---|---|---|---|
Li | 3 | 1s22s1 | 1s22s1 |
Be | 4 | 1s22s2 | 1s22s2 |
B | 5 | 1s22s22p1 | 1s22s22p1 |
C | 6 | 1s22s22p2 | 1s22s22p2 |
N | 7 | 1s22s22p3 | 1s22s22p3 |
Ne | 10 | 1s22s22p6 | 1s22s22p6 |
Na | 11 | 1s22s22p63s1 | 1s22s22p63s1 |
Condensed (Abbreviated) Electron Configurations
To simplify electron configurations, use the symbol of the previous noble gas in brackets to represent core electrons, followed by the valence electron configuration.
Example: Na: [Ne]3s1
Example: Cl: [Ne]3s23p5
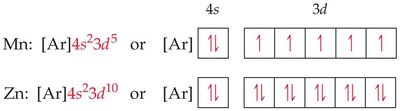
Transition Metals, Lanthanides, and Actinides
Transition metals fill d orbitals after the s orbital of the next higher shell. Lanthanides and actinides fill f orbitals. Some anomalies occur due to the close energy of s, d, and f orbitals, leading to unexpected electron configurations (e.g., Cr: [Ar]4s13d5).
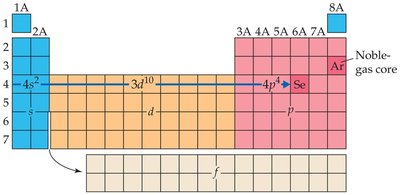
Periodic Table and Electron Configuration
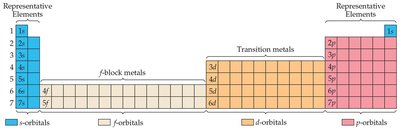
Blocks of the Periodic Table
The periodic table is divided into blocks corresponding to the filling of s, p, d, and f orbitals. The s and p blocks are called main-group elements, d block is for transition metals, and f block for inner transition elements.
Using the Periodic Table to Write Electron Configurations
Electron configurations can be determined by following the order of filling across the periodic table. The position of an element indicates its valence electron configuration.
Quantum Numbers and Allowed Orbitals
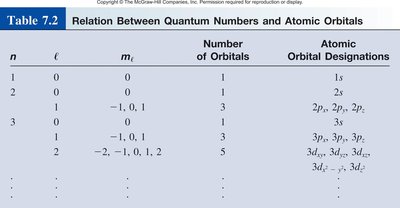
Relation Between Quantum Numbers and Atomic Orbitals
The table below summarizes the relationship between quantum numbers and the number of orbitals:
n | l | ml | Number of Orbitals | Atomic Orbital Designations |
|---|---|---|---|---|
1 | 0 | 0 | 1 | 1s |
2 | 0 | 0 | 1 | 2s |
2 | 1 | -1, 0, 1 | 3 | 2px, 2py, 2pz |
3 | 0 | 0 | 1 | 3s |
3 | 1 | -1, 0, 1 | 3 | 3px, 3py, 3pz |
3 | 2 | -2, -1, 0, 1, 2 | 5 | 3dxy, 3dyz, 3dzx, 3dx2-y2, 3dz2 |
Sample Questions and Answers
Which set of quantum numbers is not possible? For l = 0, ml must be 0.
Maximum number of electrons for n = 4, l = 3, ml = -2, ms = +1/2? Only one electron can have this set.
Number of orbitals in a d subshell? 5 (l = 2, ml = -2, -1, 0, 1, 2)
Difference in electron configuration between carbon-14 and carbon-12? None; isotopes have the same electron configuration.
Example electron configurations:
O (8e): 1s22s22p4
Ne (10e): 1s22s22p6
Mg (12e): 1s22s22p63s2
Si (14e): 1s22s22p63s23p2
Cl- (18e): 1s22s22p63s23p6
Sr2+ (36e): 1s22s22p63s23p64s23d104p6
