 Back
BackSolutions and Their Properties: General Chemistry Study Notes
Study Guide - Smart Notes

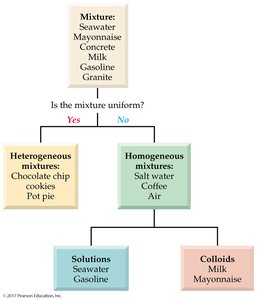
Solutions and Mixtures
Classification of Mixtures
Mixtures are combinations of two or more substances that retain their individual properties. They can be classified as heterogeneous or homogeneous based on uniformity of composition.
Heterogeneous mixtures: Not uniform throughout; different regions have different compositions. Examples: chocolate chip cookies, pot pie.
Homogeneous mixtures: Uniform throughout; same composition everywhere. Examples: salt water, coffee, air.
Homogeneous mixtures are further divided into solutions and colloids:
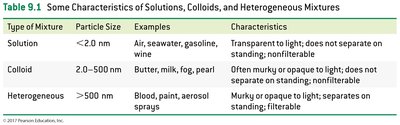
Solutions: Particle size < 2 nm; transparent, do not separate on standing.
Colloids: Particle size 2–500 nm; often murky or opaque, do not separate on standing.
Type of Mixture | Particle Size | Examples | Characteristics |
|---|---|---|---|
Solution | <2.0 nm | Air, seawater, gasoline, wine | Transparent to light; does not separate on standing; nonfilterable |
Colloid | 2.0–500 nm | Butter, milk, fog, pearl | Often murky or opaque to light; does not separate on standing; nonfilterable |
Heterogeneous | >500 nm | Blood, paint, aerosol sprays | Murky or opaque to light; separates on standing; filterable |
Types of Solutions
Solutions can be classified based on the physical state of their solute and solvent.
Type of Solution | Example |
|---|---|
Gas in gas | Air (O2, N2, Ar, other gases) |
Gas in liquid | Seltzer water (CO2 in water) |
Gas in solid | H2 in palladium metal |
Liquid in liquid | Gasoline (mixture of hydrocarbons) |
Liquid in solid | Dental amalgam (mercury in silver) |
Solid in liquid | Seawater (NaCl and other salts in water) |
Solid in solid | Metal alloys (e.g., 14-karat gold) |

The Solution Process
Solubility and Intermolecular Forces
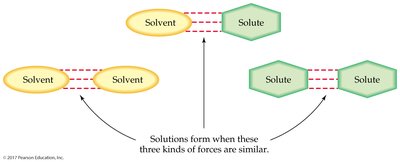
The ability of a solute to dissolve in a solvent depends on the strength of attraction between solute and solvent particles compared to the attractions within the pure substances. The principle "like dissolves like" guides solubility:
Polar solvents dissolve polar and ionic solutes.
Nonpolar solvents dissolve nonpolar solutes.
Example: Water (polar) dissolves NaCl (ionic), but oil (nonpolar) does not mix with water.
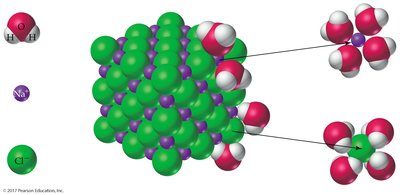
Solvation and Hydration
When ionic compounds dissolve in water, water molecules surround the ions, stabilizing them by electrical attraction. This process is called solvation (or hydration for water).
Physical and Enthalpy Changes
Dissolution is a physical change; the solute and solvent retain their chemical identities.
Dissolution involves an enthalpy change (ΔH):
Exothermic dissolution: Releases heat (e.g., some salts).
Endothermic dissolution: Absorbs heat, cooling the solution (e.g., instant cold packs).
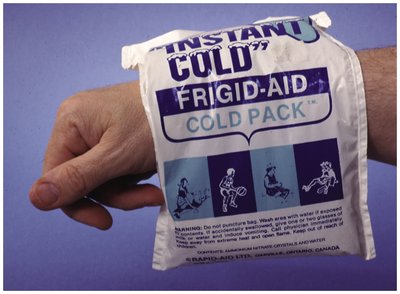
Solubility
Miscibility, Saturation, and Solubility
Miscible substances are mutually soluble in all proportions. Most substances have a solubility limit beyond which no more will dissolve. A saturated solution contains the maximum amount of dissolved solute at equilibrium.
Solubility: Maximum amount of solute that will dissolve in a given amount of solvent at a specified temperature.
Example: At 20°C, 35.8 g NaCl dissolves in 100 mL water; any excess remains undissolved.
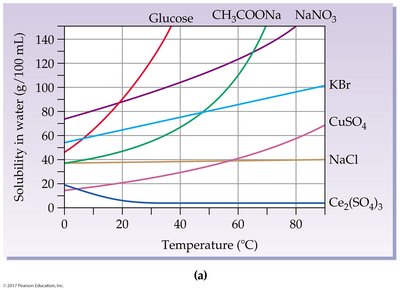
Effect of Temperature on Solubility
Solubility of Solids and Gases
Temperature affects solubility differently for solids and gases:
Most solids become more soluble as temperature increases.
Gases become less soluble as temperature increases.
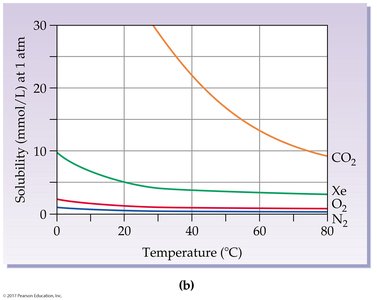
Supersaturated Solutions
Solids that are more soluble at high temperatures can form supersaturated solutions—unstable solutions containing more solute than a saturated solution. Disturbance causes rapid precipitation.

Worked Example: Solubility of Oxygen
Solubility of O2 at 25°C ≈ 8.3 mg/L; at 35°C ≈ 7.0 mg/L.
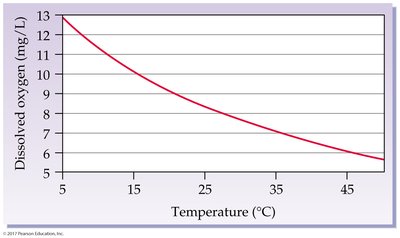
Percent change in solubility:
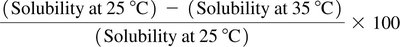
Effect of Pressure on Solubility: Henry’s Law
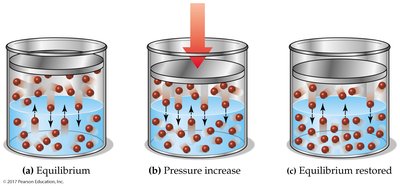
Henry’s Law
The solubility of a gas in a liquid is directly proportional to the partial pressure of the gas above the solution (at constant temperature).
Increasing pressure increases gas solubility.
Decreasing pressure decreases gas solubility.
Henry’s Law equation:
Example calculation:
Units of Concentration
Percent Concentrations
Percent concentrations express the amount of solute in 100 units of solution.
Concentration Measure | Solute Measure | Solution Measure |
|---|---|---|
Mass/mass percent, (m/m)% | Mass (g) | Mass (g) |
Volume/volume percent, (v/v)% | Volume (mL) | Volume (mL) |
Mass/volume percent, (m/v)% | Mass (g) | Volume (mL) |
Parts per million (ppm) | Parts* | 106 parts* |
Parts per billion (ppb) | Parts* | 109 parts* |
Molarity, M | Moles | Volume (L) |
Worked Examples
Mass/mass percent:

Volume/volume percent:

Mass/volume percent:

Parts per million (ppm) and parts per billion (ppb):


Molarity (M)
Molarity is the number of moles of solute per liter of solution.
Example:
Dilution
Calculating Dilutions
When diluting a solution, the amount of solute remains constant; only the volume changes. The dilution equation is:
Where and are the concentration and volume of the concentrated solution, and and are those of the diluted solution.
Example:
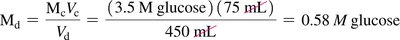
To prepare 750 mL of 0.32 M NaOH from 1.0 M NaOH:
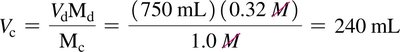
Ions in Solution: Electrolytes
Electrolytes and Conductivity
Ionic compounds in aqueous solution conduct electricity due to the movement of ions.
Strong electrolytes: Ionize completely in water.
Weak electrolytes: Partially ionize in water.
Nonelectrolytes: Do not produce ions in water.

Equivalents of Ions
Body fluids contain many ions. The equivalent (Eq) is the amount of ion equal to 1 mol of charge. 1 mEq = 0.001 Eq; 1 Eq = 1000 mEq.
Example: Normal concentration of Ca2+ in blood is 5.0 mEq/L. To find mg of Ca2+ in 1.00 L blood:
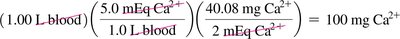
Properties of Solutions: Colligative Properties
Colligative Properties
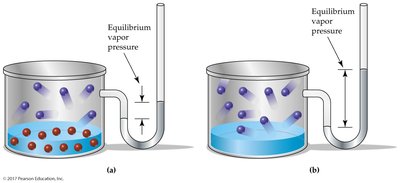
Colligative properties depend on the number of dissolved solute particles, not their chemical identity.
Vapor pressure lowering: Solutions have lower vapor pressure than pure solvents.
Boiling point elevation: Solutions boil at higher temperatures than pure solvents.
Freezing point depression: Solutions freeze at lower temperatures than pure solvents.
Osmosis: Solvent passes through a semipermeable membrane toward higher solute concentration.
Boiling Point Elevation
Each mole of solute particles raises the boiling point of 1 kg of water by 0.51°C.
Example: 0.75 mol KBr in 1.0 kg water increases boiling point to 100.77°C.
Freezing Point Depression
Each mole of nonvolatile solute particles lowers the freezing point of 1 kg water by 1.86°C.
Example: If a tomato freezes at –2.5°C, the concentration of dissolved particles is found using freezing point depression.
Osmosis and Osmotic Pressure
Osmosis
Osmosis is the passage of solvent through a semipermeable membrane separating two solutions of different concentration. Osmotic pressure is the external pressure needed to prevent net movement of solvent.
Osmolarity (osmol/L): Sum of molarities of all dissolved particles in 1.0 L solution.
Example: 5.0% (m/v) glucose solution has osmolarity equal to its molarity since glucose does not dissociate.
Isotonic, Hypotonic, and Hypertonic Solutions
Isotonic: Same osmolarity as blood plasma.
Hypotonic: Lower osmolarity than plasma or cells.
Hypertonic: Higher osmolarity than plasma or cells.
Dialysis
Dialysis and Applications
Dialysis uses a membrane with pores that allow solvent and small solute particles to pass through, but not large molecules like proteins. Hemodialysis is used to cleanse blood in patients with kidney malfunction.
Protein molecules do not cross semipermeable membranes and help regulate osmolarity of body fluids.
Osmosis regulates delivery of oxygen and nutrients and removal of waste products in cells.
Additional info: These notes cover Chapter 9: Solutions, including all major concepts relevant to general chemistry, such as types of mixtures, solution process, solubility, effects of temperature and pressure, concentration units, dilution, electrolytes, colligative properties, osmosis, and dialysis. All images included are directly relevant to the adjacent explanations.
