 Back
BackSolutions: Formation, Properties, and Solubility
Study Guide - Smart Notes

Solutions
Introduction to Solutions
A solution is a homogeneous mixture composed of two or more substances. Solutions can exist in solid, liquid, or gaseous states. The substance present in the greatest amount is called the solvent, while the other substances are referred to as solutes. Each component in a solution is known as a solution component.
Solvent: The major component in a solution.
Solute: The minor component(s) in a solution.
Solvation: The process by which solute particles are surrounded by solvent molecules.
Hydration: Solvation when the solvent is water.
Energetics of Solution Formation
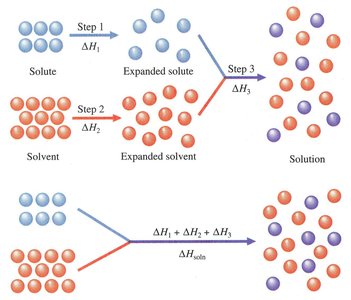
The formation of a solution involves several energetic steps. The overall enthalpy change for solution formation, denoted as , is the sum of three distinct steps:
: Separation of solute molecules (always endothermic).
: Separation of solvent molecules (always endothermic).
: Formation of solute-solvent interactions (always exothermic).
The overall enthalpy change is given by:
Depending on the relative magnitudes of these terms, the formation of a solution can be either endothermic or exothermic.
Spontaneity and Solution Formation
Whether a solution forms spontaneously depends on both enthalpy and entropy:
Exothermic processes () are generally spontaneous.
Endothermic processes () can be spontaneous if they result in a significant increase in entropy (randomness).
Entropy always increases during solution formation, favoring spontaneity unless solute-solute or solvent-solvent interactions are too strong.
Solubility and "Like Dissolves Like"
Solubility refers to the ability of a solute to dissolve in a solvent. Substances with similar intermolecular forces tend to be soluble in one another, summarized by the phrase "like dissolves like."
Non-polar substances are soluble in non-polar solvents.
Ionic and polar substances are soluble in polar solvents.
Network solids (e.g., diamond) are generally insoluble in both types of solvents.
Miscible liquids: Mix in all proportions.
Immiscible liquids: Do not dissolve in one another.
Types of Solutions
Solutions can be classified based on their saturation:
Saturated solution: Contains the maximum amount of solute that can dissolve at a given temperature; excess solute remains undissolved.
Supersaturated solution: Contains more solute than can normally dissolve at a given temperature, typically formed by heating and then cooling.
Effect of Pressure on Solubility
Pressure affects the solubility of gases but not solids or liquids. The solubility of a gas in a liquid increases in direct proportion to its partial pressure above the solution, described by Henry's Law:
= solubility of the gas
= Henry's Law constant
= partial pressure of the gas
Example: For CO2, M/atm at 25°C. If the partial pressure of CO2 in the atmosphere is atm, the expected concentration in lake water is:
M
Effect of Temperature on Solubility
Temperature has different effects on the solubility of solids and gases:
As temperature increases, the solubility of most solids in water increases.
As temperature increases, the solubility of gases in water decreases.
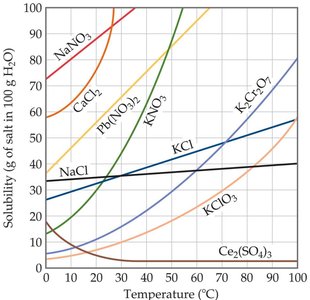
Example: The solubility curves show that salts like KNO3 and NaNO3 become much more soluble as temperature rises, while Ce2(SO4)3 decreases in solubility with increasing temperature.
Summary Table: Types of Solutions and Solubility Factors
Type | Description | Solubility Factors |
|---|---|---|
Saturated | Equilibrium with undissolved solute | Temperature, nature of solute/solvent |
Supersaturated | More solute than equilibrium allows | Formed by heating then cooling |
Unsaturated | Less solute than equilibrium allows | Can dissolve more solute |
Additional info: Colligative properties, concentration units, and colloids are also important aspects of solution chemistry, but were not covered in detail in the provided materials.
