 Back
BackStates of Matter, Intermolecular Forces, and Gas Laws: Core Concepts in General Chemistry
Study Guide - Smart Notes

States of Matter and Their Changes
Phases of Matter and Phase Changes
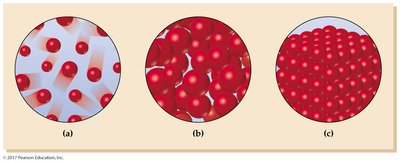
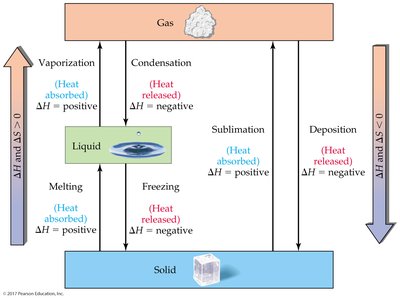
Matter exists in three primary states: solid, liquid, and gas. The state of a substance depends on the balance between the strength of attractive forces among particles and their kinetic energy. Changes between these states are called phase changes and are always reversible, involving changes in both enthalpy (ΔH) and entropy (ΔS).
Melting (solid to liquid): Endothermic (ΔH > 0), increases disorder (ΔS > 0).
Freezing (liquid to solid): Exothermic (ΔH < 0), decreases disorder (ΔS < 0).
Vaporization (liquid to gas): Endothermic (ΔH > 0), increases disorder (ΔS > 0).
Condensation (gas to liquid): Exothermic (ΔH < 0), decreases disorder (ΔS < 0).
Sublimation (solid to gas): Endothermic (ΔH > 0), increases disorder (ΔS > 0).
Deposition (gas to solid): Exothermic (ΔH < 0), decreases disorder (ΔS < 0).
The melting point is the temperature where solid and liquid phases are in equilibrium; the boiling point is where liquid and gas phases are in equilibrium.
Intermolecular Forces
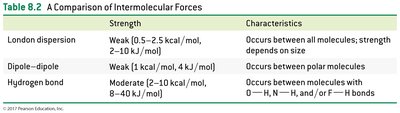
Types and Effects of Intermolecular Forces
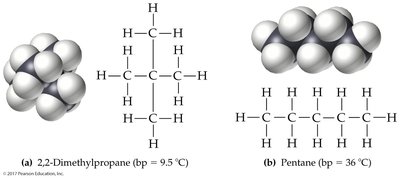
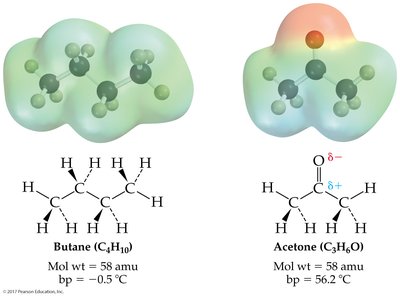
Intermolecular forces (IMFs) are attractions between molecules or atoms, crucial for determining physical properties such as boiling and melting points. There are three main types:
London dispersion forces: Present in all molecules, arising from temporary shifts in electron density. Strength increases with molecular size and surface area.
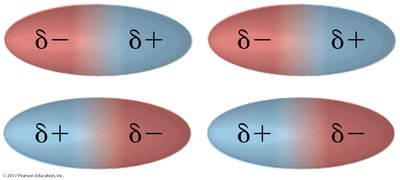
Dipole-dipole forces: Occur between polar molecules, where positive and negative ends attract each other.
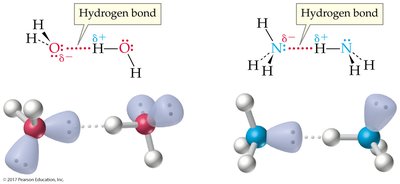
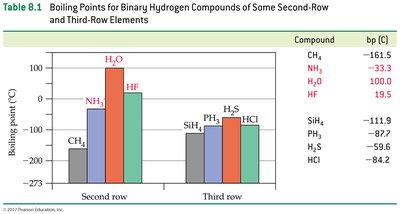
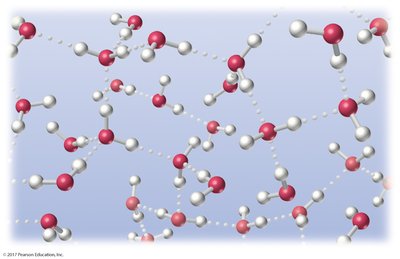
Hydrogen bonding: A strong dipole-dipole interaction between hydrogen bonded to O, N, or F and another O, N, or F atom.

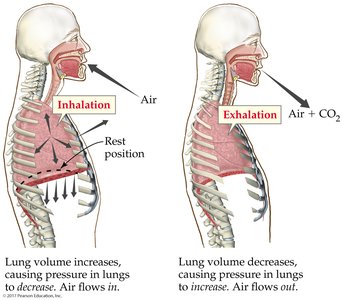
Gases and the Kinetic-Molecular Theory
Kinetic-Molecular Theory of Gases
The kinetic-molecular theory explains gas behavior based on the following assumptions:
Gas particles move randomly and independently, with negligible attractive forces.
The volume of gas particles is much smaller than the space between them.
Average kinetic energy is proportional to temperature (in Kelvin).
Collisions are elastic, conserving total kinetic energy.
A gas that perfectly follows these assumptions is called an ideal gas.
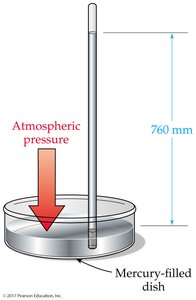
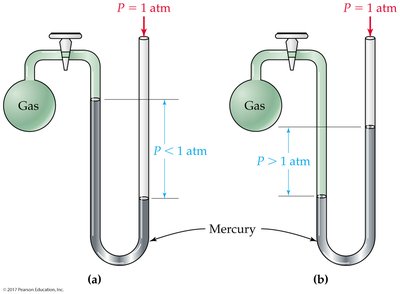
Pressure
Definition and Measurement of Pressure
Pressure (P) is the force per unit area exerted on a surface. Atmospheric pressure at sea level is 1 atm (14.7 psi, 760 mmHg, or 101,325 Pa). Pressure can be measured using barometers (for atmospheric pressure) and manometers (for gas samples in containers).

Gas Laws
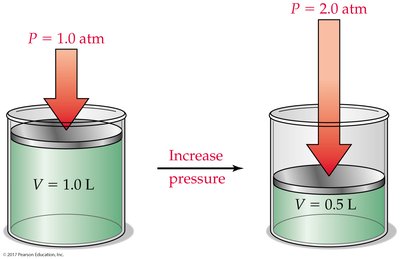
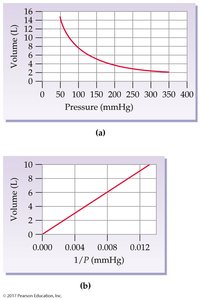
Boyle’s Law: Pressure-Volume Relationship
At constant temperature and amount of gas, the volume of a gas is inversely proportional to its pressure:
If pressure doubles, volume halves.
Mathematically:
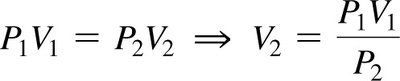
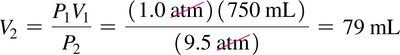
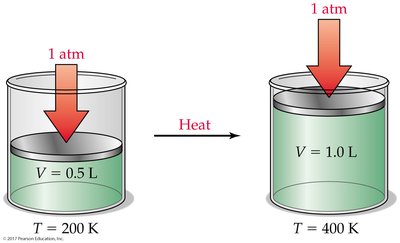
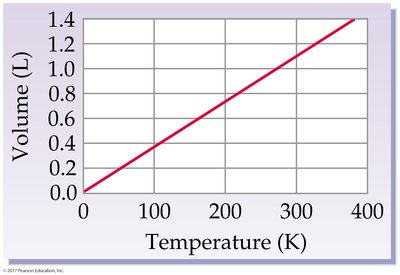
Charles’s Law: Volume-Temperature Relationship
At constant pressure and amount of gas, the volume of a gas is directly proportional to its Kelvin temperature:
If temperature doubles, volume doubles.
Mathematically:


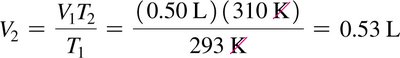
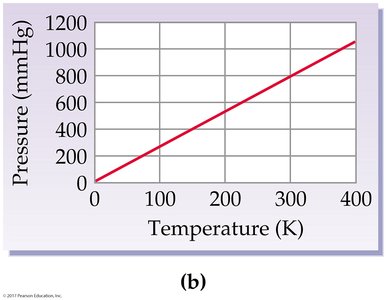
Gay-Lussac’s Law: Pressure-Temperature Relationship
At constant volume and amount of gas, the pressure of a gas is directly proportional to its Kelvin temperature:
If temperature doubles, pressure doubles.
Mathematically:
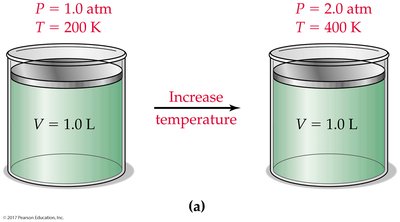
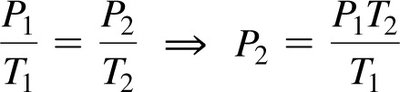
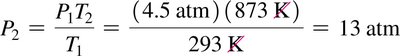
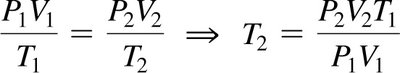
The Combined Gas Law
Combines Boyle’s, Charles’s, and Gay-Lussac’s laws for a fixed amount of gas:
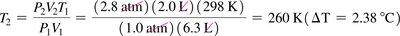
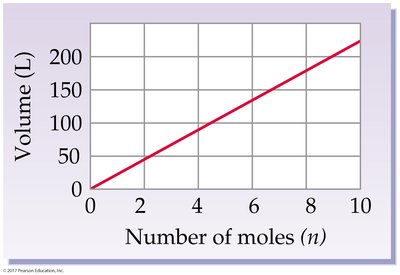
Avogadro’s Law: Volume-Mole Relationship
At constant temperature and pressure, the volume of a gas is directly proportional to the number of moles:
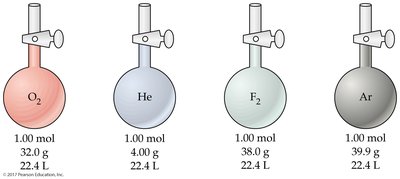
Standard Temperature and Pressure (STP): 0°C (273 K), 1 atm
Standard molar volume: 22.4 L/mol for any ideal gas at STP
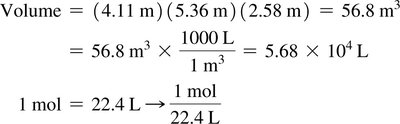
The Ideal Gas Law
Relates pressure, volume, temperature, and number of moles for an ideal gas:
R (gas constant): 0.0821 L·atm/mol·K or 62.4 L·mmHg/mol·K
Partial Pressure and Dalton’s Law
Dalton’s Law of Partial Pressures
In a mixture of gases, each gas exerts a partial pressure as if it were alone. The total pressure is the sum of the partial pressures:
Partial pressure is proportional to the mole fraction or percent composition of each gas in the mixture.
Liquids
Properties of Liquids
Liquids are characterized by constant molecular motion. Molecules with enough energy can escape into the gas phase (vaporization). In a closed container, vaporization and condensation reach equilibrium, and the vapor pressure is established. Vapor pressure increases with temperature and depends on the strength of intermolecular forces.
Boiling point: Temperature at which vapor pressure equals atmospheric pressure.
Viscosity: Resistance to flow, increases with stronger intermolecular forces.
Surface tension: Caused by differences in intermolecular forces at the surface versus the interior of the liquid.
Solids
Types of Solids
Solids are classified as crystalline or amorphous:
Crystalline solids: Ordered, repeating structures. Types include:
Ionic solids: Composed of ions in a regular lattice (e.g., NaCl).
Molecular solids: Molecules held by intermolecular forces (e.g., ice).
Covalent network solids: Atoms linked by covalent bonds in a giant network (e.g., diamond).
Metallic solids: Metal cations in a sea of electrons, allowing conductivity and malleability.
Amorphous solids: Disordered, non-repeating structures (e.g., glass, tar).
Changes of State Calculations
Calculating Heat for Phase Changes
When a substance is heated, energy increases molecular motion. At phase change temperatures, energy is used to overcome intermolecular forces rather than to increase temperature.
Heat of fusion: Energy required to melt 1 g of a substance at its melting point.
Heat of vaporization: Energy required to vaporize 1 g of a substance at its boiling point.
Strong intermolecular forces result in higher heats of fusion and vaporization.
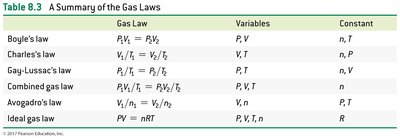
Summary Table: Gas Laws
Gas Law | Equation | Variables | Constant |
|---|---|---|---|
Boyle's law | P, V | n, T | |
Charles's law | V, T | n, P | |
Gay-Lussac's law | P, T | n, V | |
Combined gas law | P, V, T | n | |
Avogadro's law | V, n | P, T | |
Ideal gas law | P, V, T, n | R |
