 Back
BackStoichiometry and Chemical Quantities: Study Notes
Study Guide - Smart Notes

Stoichiometry: Chemical Quantities and Calculations
Introduction to Stoichiometry
Stoichiometry is the branch of chemistry that deals with the quantitative relationships between reactants and products in a chemical reaction. It allows chemists to predict the amounts of substances consumed and produced in a given reaction.
Stoichiometric calculations are based on balanced chemical equations.
Key concepts include mole, molar mass, Avogadro's number, and percent composition.
Stoichiometry is essential for laboratory work, industrial processes, and understanding chemical reactions.
Molecular and Formula Weights
The molecular weight (or molar mass) of a compound is the sum of the atomic masses of all atoms in its chemical formula.
Molecular weight is measured in atomic mass units (amu) or grams per mole (g/mol).
To calculate, add the atomic masses of each element multiplied by the number of atoms present.
Formula weight applies to ionic compounds, while molecular weight applies to covalent compounds.
Example: The formula weight of NaCl is calculated as: 
Percent Composition
Percent composition expresses the mass percentage of each element in a compound.
Formula:
Used to determine the empirical formula and analyze purity.
Example: For :
Calculate mass of each element in 1 mol.
Divide by total molar mass and multiply by 100%.
The Mole Concept and Avogadro's Number
A mole is a unit that represents particles (atoms, molecules, ions).
Avogadro's number is .
1 mole of any substance contains Avogadro's number of particles.
Used to convert between mass, moles, and number of particles.
Example: mol of Cu contains: atoms of Cu 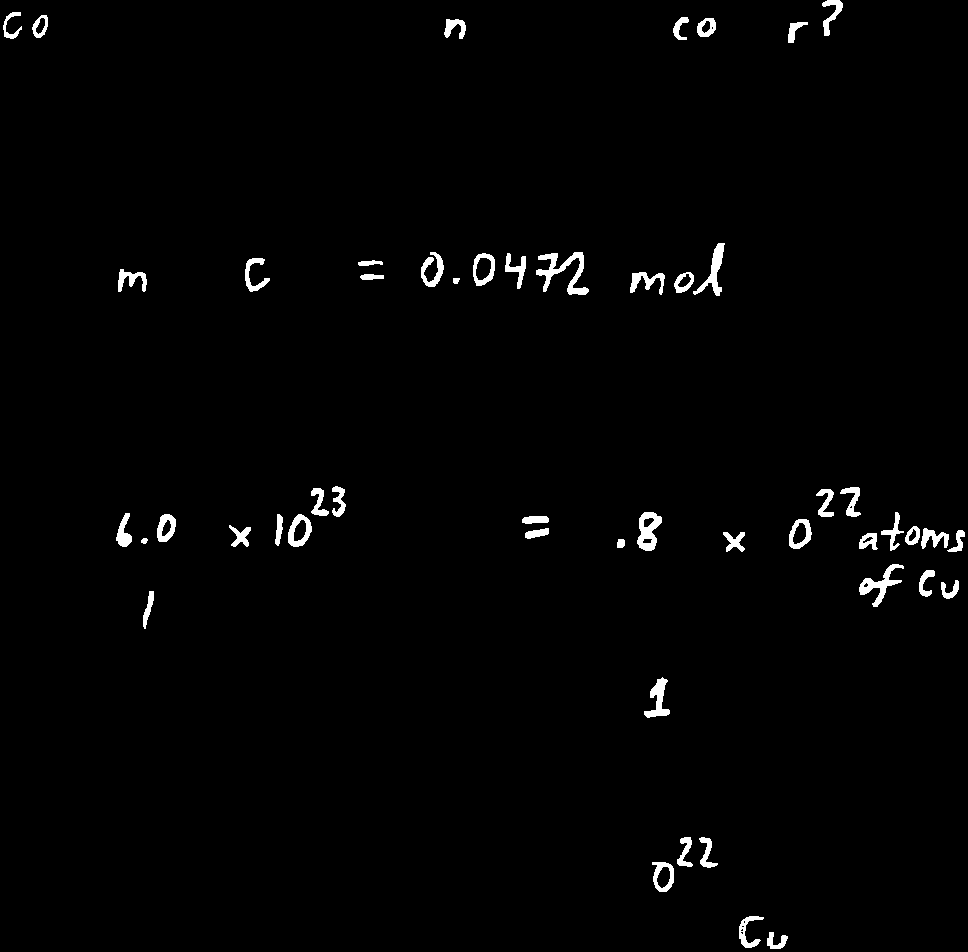
Stoichiometric Calculations: Mole-to-Mole and Mass-to-Mass
Stoichiometric calculations use balanced equations to relate moles and masses of reactants and products.
Steps: (1) Write balanced equation, (2) Convert mass to moles, (3) Use mole ratio, (4) Convert moles to mass.
Mole ratios are derived from coefficients in the balanced equation.
Example: For :
Given mass of NaO$_2$, calculate moles.
Use mole ratio to find moles of O produced.
Convert moles of O to grams.

Limiting Reactant and Theoretical Yield
The limiting reactant is the substance that is completely consumed first, limiting the amount of product formed.
Identify limiting reactant by comparing mole ratios.
Theoretical yield is the maximum amount of product possible.
Actual yield is often less due to losses; percent yield is calculated as:
Example: If 20 g of SO is produced from a reaction with a theoretical yield of 28.04 g: 
Worked Examples and Practice Problems
Calculating number of atoms in a given mass.
Finding mass of product from a given mass of reactant.
Determining limiting reactant and percent yield.
Example: How many moles of oxygen are in 0.12 mol of Al(NO)$_3$?
Each mole of Al(NO)$_3$ contains 9 moles of O.
mol O
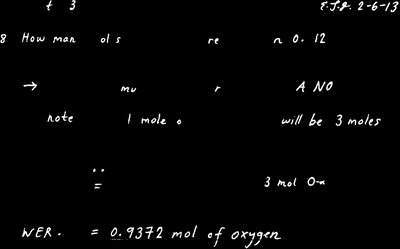
Summary Table: Key Stoichiometric Relationships
Concept | Formula | Example |
|---|---|---|
Mole to Mass | 2 mol HO × 18.02 g/mol = 36.04 g | |
Mass to Mole | 36.04 g HO ÷ 18.02 g/mol = 2 mol | |
Mole to Particles | 1 mol Na = atoms | |
Percent Composition | H in HO: | |
Percent Yield | 20 g / 28.04 g × 100% = 71.4% |
Additional info:
Some equations and examples were inferred for clarity and completeness.
Images included only where they directly reinforce calculations or concepts.
