 Back
BackStoichiometry, Limiting Reactant, and Molecular Calculations Worksheet Guidance
Study Guide - Smart Notes

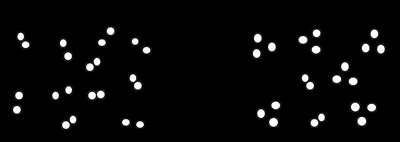
Q1. The following box represents a chemical reaction between AB2 and B2:
Background
Topic: Stoichiometry and Limiting Reactant
This question tests your understanding of how to interpret molecular diagrams, write balanced chemical equations, identify limiting reactants, and perform stoichiometric calculations based on given quantities.
Key Terms and Formulas:
Balanced Equation: A chemical equation with equal numbers of each atom on both sides.
Limiting Reactant: The reactant that is completely consumed first, limiting the amount of product formed.
Stoichiometry: The calculation of reactants and products in chemical reactions using mole ratios.
Step-by-Step Guidance
Examine the molecular diagram to identify the reactants (AB2 and B2) and products. Count the number of each type of molecule on both sides of the reaction.
Write a balanced chemical equation for the reaction, making sure the number of each atom is the same on both sides. Include the physical states (e.g., (g), (s), (aq)) if possible.
To determine the limiting reactant, compare the mole ratio of AB2 to B2 from the balanced equation with the quantities provided or depicted in the diagram.
For part (c), use the balanced equation to set up a stoichiometric calculation: convert moles of each reactant to moles of product, and identify which reactant limits the amount of product formed.
For part (d), express the amount of product formed in terms of a variable (G mol B2), using the stoichiometric ratio from the balanced equation.
Try solving on your own before revealing the answer!
Q2. Calculate the number of grams of O2 required to burn 72.0 g of C2H6 to CO2 and H2O.
Background
Topic: Stoichiometry and Combustion Reactions
This question tests your ability to use balanced equations to convert between mass of reactant and mass of another reactant.
Key Terms and Formulas:
Molar Mass: The mass of one mole of a substance (g/mol).
Stoichiometric Ratio: The ratio of coefficients from the balanced equation.
Step-by-Step Guidance
Write down the balanced equation: $2\,\mathrm{C_2H_6\,(g)} + 7\,\mathrm{O_2\,(g)} \rightarrow 4\,\mathrm{CO_2\,(g)} + 6\,\mathrm{H_2O\,(g)}$
Calculate the molar mass of C2H6 ($2 \times 12.01 + 6 \times 1.01$ g/mol).
Convert 72.0 g of C2H6 to moles using its molar mass.
Use the stoichiometric ratio from the balanced equation to find moles of O2 required per mole of C2H6.
Set up the calculation to convert moles of O2 to grams using the molar mass of O2 ($2 \times 16.00$ g/mol).
Try solving on your own before revealing the answer!
Q3. Calculate the number of grams of zinc chloride that can be prepared from 26.5 g of zinc.
Background
Topic: Stoichiometry and Limiting Reactant
This question tests your ability to use a balanced equation to convert mass of a reactant to mass of a product.
Key Terms and Formulas:
Molar Mass: The mass of one mole of a substance.
Stoichiometric Ratio: The ratio of coefficients from the balanced equation.
Step-by-Step Guidance
Write the balanced equation: $\mathrm{Zn\,(s) + 2\,HCl\,(aq) \rightarrow ZnCl_2\,(aq) + H_2\,(g)}$
Calculate the molar mass of Zn ($65.38$ g/mol) and ZnCl2 ($65.38 + 2 \times 35.45$ g/mol).
Convert 26.5 g of Zn to moles using its molar mass.
Use the stoichiometric ratio (1:1) to find moles of ZnCl2 produced.
Set up the calculation to convert moles of ZnCl2 to grams using its molar mass.
Try solving on your own before revealing the answer!
Q4. A 50.0 g sample of CaCO3 is allowed to react with 35.0 g of H3PO4:
3 CaCO3 + 2 H3PO4 → Ca3(PO4)2 + 3 CO2 + 3 H2O
Background
Topic: Limiting Reactant, Stoichiometry, and Percent Yield
This question tests your ability to determine the limiting reactant, calculate theoretical yield, and percent yield.
Key Terms and Formulas:
Limiting Reactant: The reactant that is used up first in a reaction.
Theoretical Yield: The maximum amount of product that can be formed from the limiting reactant.
Percent Yield: $\text{Percent Yield} = \frac{\text{Actual Yield}}{\text{Theoretical Yield}} \times 100\%$
Step-by-Step Guidance
Calculate the molar masses of CaCO3, H3PO4, and Ca3(PO4)2.
Convert the given masses of CaCO3 and H3PO4 to moles.
Use the stoichiometric ratios from the balanced equation to determine which reactant is limiting.
Calculate the theoretical yield (in grams) of Ca3(PO4)2 based on the limiting reactant.
For part (b), determine the moles of excess reagent remaining after the reaction.
For part (c), use the actual yield and theoretical yield to calculate percent yield.
Try solving on your own before revealing the answer!
Q5. Ethyl butyrate (C6H12O2): Molar Mass, Molecules, and Atoms Calculations
Background
Topic: Molar Mass, Mole-to-Mass, and Molecule Calculations
This question tests your ability to calculate molar mass, convert between moles, mass, and molecules, and count atoms in a compound.
Key Terms and Formulas:
Molar Mass: Sum of atomic masses in a molecule.
Avogadro's Number: $6.022 \times 10^{23}$ molecules/mol.
Mass of One Molecule: $\text{Mass of one molecule} = \frac{\text{Molar mass}}{6.022 \times 10^{23}}$
Number of Atoms: Multiply the number of moles by Avogadro's number and the number of atoms per molecule.
Step-by-Step Guidance
Calculate the molar mass of C6H12O2 ($6 \times 12.01 + 12 \times 1.01 + 2 \times 16.00$ g/mol).
For each part, use the appropriate conversion: moles to grams, grams to moles, moles to molecules, and molecules to mass.
For atom counting, multiply the number of moles by Avogadro's number and the number of hydrogen atoms per molecule.
Set up the calculation for each part, but stop before plugging in the final numbers.
Try solving on your own before revealing the answer!
