 Back
BackStudy Guide: Cubic Unit Cells and Solid-State Chemistry
Study Guide - Smart Notes

Q1. How much of a corner atom is in a single unit cell?
Background
Topic: Unit Cell Geometry
This question tests your understanding of how atoms are shared between adjacent unit cells in a crystal lattice, specifically at the corners.
Key Terms:
Corner atom: An atom located at the corner of a unit cell.
Unit cell: The smallest repeating unit in a crystal lattice.
Step-by-Step Guidance
Visualize the cubic unit cell and note that each corner atom is shared by multiple adjacent unit cells.
Recall that a cube has 8 corners, and each corner atom is shared among 8 unit cells.
Set up the fraction of the atom that belongs to a single unit cell: .
Try solving on your own before revealing the answer!
Q2. How much of an atom in the body of a unit cell is inside the cell?
Background
Topic: Unit Cell Geometry
This question focuses on the body-centered cubic (bcc) structure, where an atom is located at the center of the cube.
Key Terms:
Body atom: An atom located at the center of the unit cell.
Body-centered cubic (bcc): A type of cubic unit cell with one atom at the center.
Step-by-Step Guidance
Consider the position of the atom at the center of the cube.
Recognize that the body atom is not shared with any other unit cell.
Set up the fraction: .
Try solving on your own before revealing the answer!
Q3. How much of an atom in the face of a unit cell is inside the cell?
Background
Topic: Unit Cell Geometry
This question deals with face-centered cubic (fcc) structures, where atoms are located at the faces of the cube.
Key Terms:
Face atom: An atom located at the center of a face of the unit cell.
Face-centered cubic (fcc): A type of cubic unit cell with atoms at each face.
Step-by-Step Guidance
Visualize the cube and note that each face atom is shared between two adjacent unit cells.
Set up the fraction: .
Try solving on your own before revealing the answer!
Q4. How many atoms are in each cell for simple cubic, bcc, and fcc?
Background
Topic: Counting Atoms in Unit Cells
This question tests your ability to calculate the total number of atoms per unit cell for different cubic structures.
Key Terms:
Simple cubic: Atoms only at corners.
Body-centered cubic (bcc): Atoms at corners and one at the center.
Face-centered cubic (fcc): Atoms at corners and faces.
Step-by-Step Guidance
For simple cubic: 8 corners × per corner = 1 atom per cell.
For bcc: 8 corners × + 1 body atom = 2 atoms per cell.
For fcc: 8 corners × + 6 faces × = 4 atoms per cell.
Try solving on your own before revealing the answer!
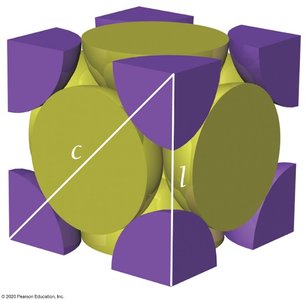
Q5. For the face-centered cubic structure, what is the length of the diagonal (c) across one face in terms of r (atomic radius)?
Background
Topic: Geometry of Cubic Unit Cells
This question tests your ability to relate the atomic radius to the geometry of the unit cell, specifically the face diagonal.
Key Formula:
Pythagorean theorem:
Face diagonal in terms of edge length:
Step-by-Step Guidance
Visualize the face-centered cubic unit cell and identify the face diagonal.
Recall that the face diagonal passes through the centers of two atoms on the face.
Set up the relationship: (since the diagonal spans four radii).
Relate the diagonal to the edge length: .
Try solving on your own before revealing the answer!
Q6. Use the answer to part a and the Pythagorean theorem to derive the expression for the edge length (l) in terms of r.
Background
Topic: Geometry of Cubic Unit Cells
This question asks you to use the relationship between the face diagonal and the edge length to express the edge length in terms of the atomic radius.
Key Formula:
Face diagonal:
Atomic radius relationship:
Step-by-Step Guidance
Set the two expressions for the face diagonal equal to each other: .
Rearrange to solve for in terms of : .
Try solving on your own before revealing the answer!
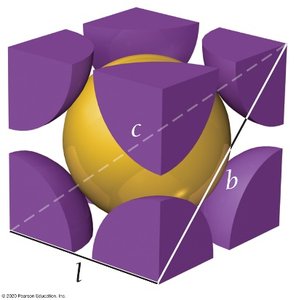
Q7. For the body-centered cubic structure, what is the length of the line (c) that runs from one corner diagonally through the center to the opposite corner in terms of r?
Background
Topic: Geometry of Cubic Unit Cells
This question tests your understanding of the body diagonal in a body-centered cubic unit cell and its relationship to the atomic radius.
Key Formula:
Body diagonal:
Atomic radius relationship: (for bcc, the diagonal passes through two atoms and the center atom)
Step-by-Step Guidance
Visualize the body-centered cubic unit cell and identify the body diagonal.
Recall that the body diagonal passes through four radii.
Set up the relationship: .
Relate the diagonal to the edge length: .
Try solving on your own before revealing the answer!
Q8. Use the Pythagorean theorem to derive an expression for the length of the line (b) that runs along the face of the cube in terms of the edge length (l).
Background
Topic: Geometry of Cubic Unit Cells
This question asks you to use the Pythagorean theorem to relate the face diagonal to the edge length in a cube.
Key Formula:
Pythagorean theorem:
Face diagonal:
Step-by-Step Guidance
Identify the right triangle formed by two edges and the face diagonal.
Apply the Pythagorean theorem: .
Solve for : .
Try solving on your own before revealing the answer!
Q9. Use the answers to parts a and b along with the Pythagorean theorem to derive the expression for the edge length (l) in terms of r for bcc.
Background
Topic: Geometry of Cubic Unit Cells
This question combines previous relationships to express the edge length in terms of the atomic radius for a body-centered cubic unit cell.
Key Formula:
Body diagonal:
Atomic radius relationship:
Step-by-Step Guidance
Set the two expressions for the body diagonal equal: .
Rearrange to solve for in terms of : .
Try solving on your own before revealing the answer!
Q10. When told the type of unit cell, what other information is implied?
Background
Topic: Unit Cell Properties
This question tests your understanding of what structural information is implied by knowing the unit cell type (simple cubic, bcc, fcc).
Key Terms:
Unit cell type: Determines the number of atoms per cell, packing efficiency, and geometric relationships.
Step-by-Step Guidance
Recall the number of atoms per unit cell for each type.
Consider the geometric relationships (edge length, atomic radius).
Think about packing efficiency and coordination number.
Try solving on your own before revealing the answer!
Q11. If you know the species (e.g., Molybdenum), what do you know?
Background
Topic: Elemental Properties in Solids
This question tests your ability to connect the identity of the element to its physical and structural properties in the solid state.
Key Terms:
Atomic mass
Atomic radius
Crystal structure
Step-by-Step Guidance
Look up the atomic mass and radius for the element.
Determine the typical crystal structure (e.g., bcc for Molybdenum).
Use this information to calculate properties like density.
Try solving on your own before revealing the answer!
Q12. If we are told the radius (136 pm), what can that be related to?
Background
Topic: Atomic Radius and Unit Cell Geometry
This question tests your ability to relate the atomic radius to the edge length and other geometric properties of the unit cell.
Key Formula:
Edge length in terms of radius (depends on unit cell type): for bcc, for fcc.
Step-by-Step Guidance
Identify the unit cell type.
Use the appropriate formula to relate radius to edge length.
Apply this relationship to calculate other properties (e.g., volume).
Try solving on your own before revealing the answer!
Q13. Write an expression for the volume of a cube.
Background
Topic: Geometry of Cubes
This question tests your ability to recall and apply the formula for the volume of a cube.
Key Formula:
Volume of a cube:
Step-by-Step Guidance
Recall that the volume of a cube is the edge length cubed.
Write the formula: .
Try solving on your own before revealing the answer!
Q14. (Tro 13.34) Molybdenum crystallized with a body-centered unit cell. The radius of a molybdenum atom is 136 pm. Calculate the density of molybdenum.
Background
Topic: Density Calculation for Crystalline Solids
This question tests your ability to calculate the density of a metal using its unit cell structure and atomic radius.
Key Formula:
Density:
Edge length for bcc:
Volume:
Step-by-Step Guidance
Calculate the edge length using the atomic radius and the formula for bcc.
Convert the edge length from pm to cm.
Calculate the volume of the unit cell: .
Determine the number of atoms per unit cell (2 for bcc).
Calculate the mass of atoms in the unit cell using molar mass and Avogadro's number.
Try solving on your own before revealing the answer!
Q15. Polonium crystallizes with a simple cubic structure. It has a density of 9.3 g/cm3. What is the radius of a polonium atom (in pm)?
Background
Topic: Density and Atomic Radius in Simple Cubic Structures
This question tests your ability to relate density, unit cell geometry, and atomic radius for a simple cubic structure.
Key Formula:
Density:
Edge length for simple cubic:
Volume:
Step-by-Step Guidance
Set up the formula for density using the mass of one atom and the volume of the unit cell.
Express the edge length in terms of atomic radius: .
Write the volume formula: .
Rearrange the density formula to solve for .
Try solving on your own before revealing the answer!
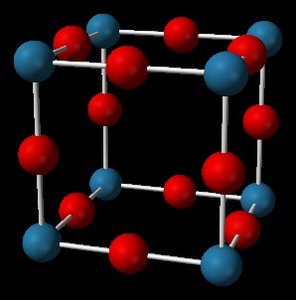
Q16. Given the unit cells shown, determine the formula for the compound.
Background
Topic: Stoichiometry and Unit Cell Analysis
This question tests your ability to deduce the chemical formula of a compound based on the arrangement of atoms in the unit cell.
Key Terms:
Stoichiometry: The ratio of atoms in the unit cell.
Unit cell: The smallest repeating unit in a crystal lattice.
Step-by-Step Guidance
Count the number of each type of atom in the unit cell.
Consider how atoms are shared between adjacent cells (corners, faces, edges, center).
Calculate the total number of each atom per unit cell.
Write the empirical formula based on the ratio.
Try solving on your own before revealing the answer!
Q17. (Tro 13.40 and 39) Identify each solid as molecular, ionic, or atomic.
Background
Topic: Classification of Solids
This question tests your ability to classify solids based on their composition and bonding.
Key Terms:
Molecular solid: Composed of molecules held together by intermolecular forces.
Ionic solid: Composed of ions held together by ionic bonds.
Atomic solid: Composed of atoms held together by metallic or covalent bonds.
Step-by-Step Guidance
For each compound, identify the type of particles present (ions, molecules, atoms).
Determine the bonding type (ionic, covalent, metallic).
Classify each solid accordingly.
Try solving on your own before revealing the answer!
Q18. (Tro 13.82) The density of an unknown metal is 12.3 g/cm3, and its atomic radius is 0.134 nm. It has a face-centered cubic lattice. Find the atomic mass of this metal.
Background
Topic: Density and Atomic Mass Calculation for FCC Metals
This question tests your ability to use density, atomic radius, and unit cell structure to calculate the atomic mass of a metal.
Key Formula:
Density:
Edge length for fcc:
Volume:
Atoms per cell (fcc): 4
Step-by-Step Guidance
Calculate the edge length using the atomic radius and the formula for fcc.
Convert the edge length from nm to cm.
Calculate the volume of the unit cell: .
Set up the density formula and solve for the mass of one atom.
Use Avogadro's number to convert the mass of one atom to molar mass.
Try solving on your own before revealing the answer!
