 Back
BackStudy Guide: Ionic Compounds and Electron Configurations
Study Guide - Smart Notes

Ionic Compounds
Formation and Properties of Ionic Compounds
Ionic compounds are a fundamental class of chemical substances formed by the electrostatic attraction between cations (positively charged ions, usually metals) and anions (negatively charged ions, usually nonmetals). The process involves the transfer of electrons from the cation to the anion, resulting in a compound with a net charge of zero.
Cation: A positively charged ion formed by the loss of electrons (typically metals).
Anion: A negatively charged ion formed by the gain of electrons (typically nonmetals).
Electron Transfer: Electrons are transferred from the cation to the anion, stabilizing both ions.
Charge Balance: The overall charge of the ionic compound must be zero. This is achieved by balancing the charges of the ions.
Criss-Cross Method: Used to determine the formula of an ionic compound by swapping the charges of the ions to become subscripts in the formula.
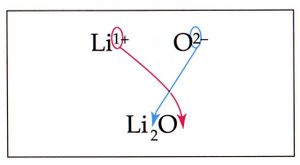
Example: Lithium oxide is formed by combining two lithium ions (Li+) with one oxide ion (O2-), resulting in the formula Li2O.
Naming and Writing Formulas for Ionic Compounds
It is important to be able to write both the names and formulas for ionic compounds, including those containing polyatomic ions.
Example 1: Aluminum fluoride = AlF3
Example 2: Calcium chlorate = Ca(ClO3)2
Example 3: Ammonium nitride = (NH4)3N
Example 4: Iron (VI) sulfate = Fe(SO4)6
Example 5: Lithium phosphate = Li3PO4
Additional info: Polyatomic ions are groups of atoms that carry a charge and act as a single unit in ionic compounds (e.g., sulfate SO42-, ammonium NH4+).
Electrons in Atoms and Ions
Valence Electrons and Light Emission
The valence electrons in atoms are responsible for the emission of colored light when energy is supplied, such as in flame tests. Different elements emit characteristic colors due to their unique electron configurations.
Flame Test Colors: Cu2+ = green, Li+ = red, Na+ = bright orange, etc.
Atomic Orbitals and Electron Arrangement
Electrons are found in specifically defined zones of probability called atomic orbitals. These orbitals are grouped into sublevels (s, p, d, f), each with a specific number of sublevels and maximum electrons.
Orbital | Sublevels | Max Electrons |
|---|---|---|
s | 1 | 2 |
p | 3 | 6 |
d | 5 | 10 |
f | 7 | 14 |
Valence Electrons: Electrons in the highest energy orbitals (e.g., 4p > 3p) are called valence electrons and are crucial for chemical bonding.
Electron Configuration Principles
Several rules govern the arrangement of electrons in atoms:
Aufbau Principle: Electrons fill the lowest energy orbitals first.
Hund’s Rule: Electrons occupy each sublevel singly before pairing up.
Pauli Exclusion Principle: No two electrons in the same orbital can have the same spin; they must have opposite spins.
Heisenberg Uncertainty Principle: The position and momentum of electrons cannot be simultaneously known.
Electron Configuration Notation
Electron configurations can be written in several ways:
Orbital Notation: Uses arrows to represent electrons in orbitals.
Electron Configuration: Uses superscripts to indicate the number of electrons in each sublevel. Example for oxygen:
Noble Gas Configuration: Uses the previous noble gas as a shorthand. Example for oxygen: [He]
Additional info: Orbital notation visually shows electron pairing and unpaired electrons, which is important for understanding chemical reactivity.
Practice Examples
Orbital notation for chlorine: 1s: ↑↓, 2s: ↑↓, 2p: ↑↓ ↑↓ ↑↓, 3s: ↑↓, 3p: ↑↓ ↑↓ ↑
Noble gas configuration for sodium: [Ne]
Electron configuration for Be2+:
Noble gas configuration for P3-: [Ne]
Orbital notation for Ne: 1s: ↑↓, 2s: ↑↓, 2p: ↑↓ ↑↓ ↑↓
Unpaired electrons in carbon: Carbon has 2 unpaired electrons. Orbital notation: 1s: ↑↓, 2s: ↑↓, 2p: ↑ ↑
Example: The electron configuration for oxygen is , and its noble gas configuration is [He] .
