 Back
BackThe Quantum-Mechanical Model of the Atom: Light, Electrons, and Atomic Structure
Study Guide - Smart Notes

The Quantum-Mechanical Model of the Atom
Introduction to Quantum Mechanics
The quantum-mechanical model of the atom explains the behavior of electrons, which in turn determines the chemical and physical properties of elements. This model is built upon the dual nature of light and electrons, which can exhibit both wave-like and particle-like properties.
The Nature of Light
Wave-Particle Duality
Light is a form of electromagnetic radiation, a type of energy composed of oscillating, perpendicular electric and magnetic fields. It exhibits both wave-like and particle-like behavior, a phenomenon known as wave-particle duality. Some properties of light are best explained by its wave nature, while others are explained by its particle nature.
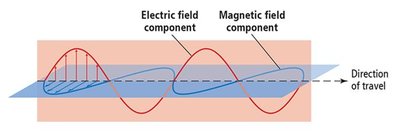
Wave Properties of Light
Amplitude: The height of the wave from the center line to the crest or trough. It determines the intensity (brightness) of light.
Wavelength (\( \lambda \)): The distance between successive crests (or troughs) of a wave. It determines the color of visible light.
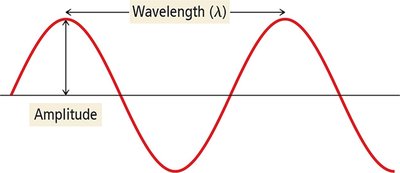
Frequency (\( \nu \)): The number of wave cycles that pass a point per second. Measured in hertz (Hz), where 1 Hz = 1 cycle/s.
Relationship: For electromagnetic waves, the speed of light (\( c \)) is constant, so wavelength and frequency are inversely proportional: where m/s (speed of light).
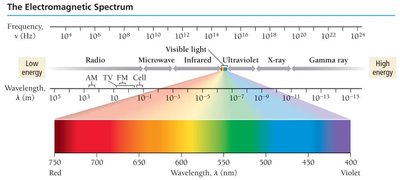
The Electromagnetic Spectrum
The electromagnetic spectrum encompasses all types of electromagnetic radiation, ranging from gamma rays (shortest wavelength, highest energy) to radio waves (longest wavelength, lowest energy). Visible light is only a small portion of this spectrum, with wavelength determining the perceived color.
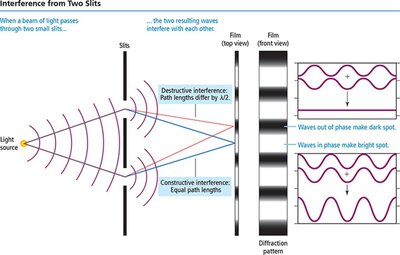
Wave Interactions: Interference and Diffraction
Interference: When two waves overlap, they can add together (constructive interference) or cancel each other out (destructive interference).
Diffraction: When waves encounter an obstacle or slit comparable in size to their wavelength, they bend around it. Particles do not diffract in this way.
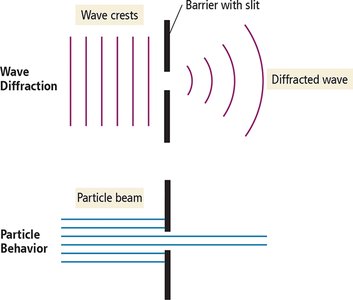
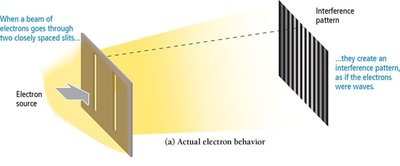
When light passes through two closely spaced slits, it produces an interference pattern characteristic of waves.
The Particle Nature of Light
Photons and the Photoelectric Effect
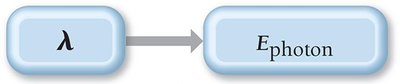
Einstein proposed that light energy is quantized in packets called photons. The energy of a photon is directly proportional to its frequency and inversely proportional to its wavelength:
Where J·s (Planck's constant)
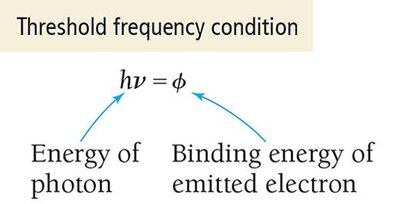
In the photoelectric effect, electrons are ejected from a metal surface when light of sufficient frequency shines on it. The minimum energy required to remove an electron is called the binding energy (\( \phi \)). The threshold condition is:
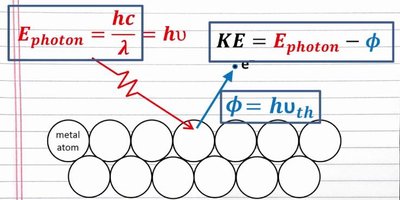
If the photon energy exceeds the binding energy, the excess energy becomes the kinetic energy of the ejected electron:
The Wave Nature of Matter
de Broglie Wavelength
Louis de Broglie proposed that particles such as electrons also have wave properties. The wavelength of a particle is given by:
Where is mass and is velocity.
Electron Diffraction
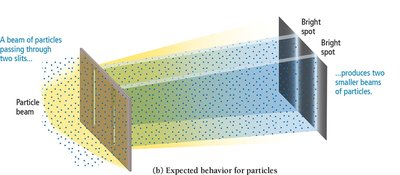
Experiments show that electrons can produce interference patterns, confirming their wave-like nature. If electrons behaved only as particles, no interference pattern would be observed.
Quantum Mechanics and Atomic Structure
Bohr Model of the Atom
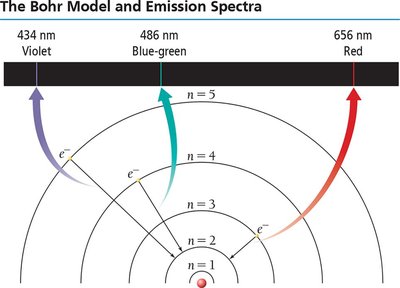
Niels Bohr proposed that electrons occupy quantized orbits (energy levels) around the nucleus. Electrons can move between these levels by absorbing or emitting photons with energy equal to the difference between the levels.
Energy levels are quantized: J, where
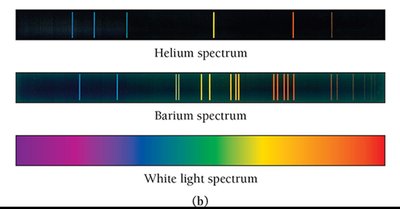
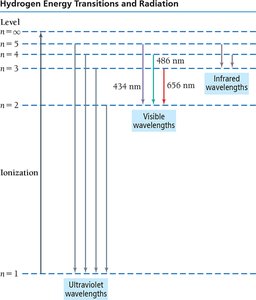
Atomic Spectroscopy
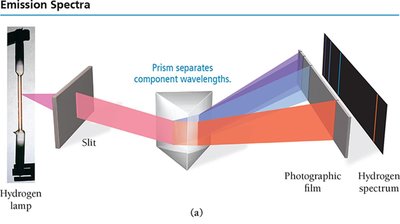
When atoms absorb energy, electrons are excited to higher energy levels. As they return to lower levels, they emit light at specific wavelengths, producing an emission spectrum unique to each element. This is the basis for flame tests and identifying elements.
Flame Tests
Different elements emit characteristic colors when heated in a flame, due to their unique emission spectra.




Quantum Numbers and Atomic Orbitals
Schrödinger Equation and Wave Functions
The Schrödinger equation describes the probability of finding an electron with a particular energy at a particular location in the atom. The solutions are wave functions (\( \psi \)), and the square of the wave function (\( \psi^2 \)) gives the probability density.
Quantum Numbers
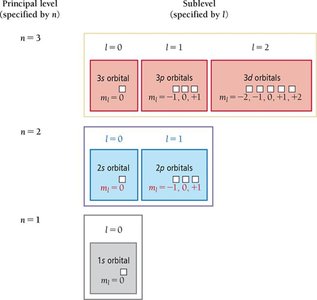
Principal quantum number (n): Specifies the energy level and size of the orbital (n = 1, 2, 3, ...).
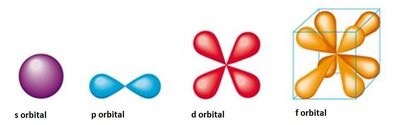
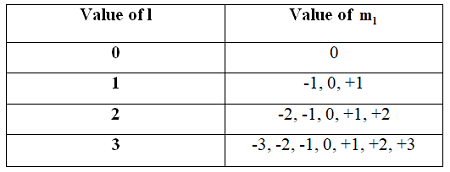
Angular momentum quantum number (l): Specifies the shape of the orbital (l = 0, 1, ..., n-1). - s (l = 0), p (l = 1), d (l = 2), f (l = 3)
Magnetic quantum number (ml): Specifies the orientation of the orbital (ml = -l to +l).
Spin quantum number (ms): Specifies the spin of the electron (ms = +1/2 or -1/2).
Energy Levels and Sublevels
Each principal energy level (n) contains n sublevels, and each sublevel contains a specific number of orbitals:
Number of sublevels in a level = n
Number of orbitals in a sublevel = 2l + 1
Number of orbitals in a level = n2
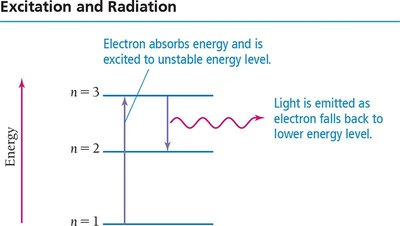
Atomic Spectroscopy Explained
Each line in an atom's emission spectrum corresponds to an electron transition between quantum-mechanical orbitals. When an electron relaxes from a higher to a lower energy level, a photon is emitted with energy equal to the difference between the two levels.
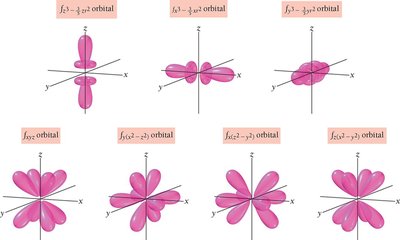
Shapes and Properties of Atomic Orbitals
s, p, d, and f Orbitals
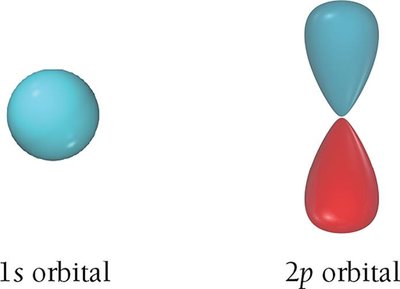
s orbitals (l = 0): Spherical shape, one per energy level.
p orbitals (l = 1): Dumbbell-shaped, three per energy level (n ≥ 2).
d orbitals (l = 2): Cloverleaf-shaped, five per energy level (n ≥ 3).
f orbitals (l = 3): Complex shapes, seven per energy level (n ≥ 4).
Probability Density and Radial Distribution
The probability density function (\( \psi^2 \)) gives the likelihood of finding an electron at a particular point in space. The radial distribution function shows the probability of finding an electron at a certain distance from the nucleus.
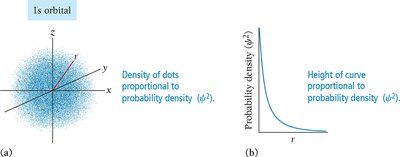
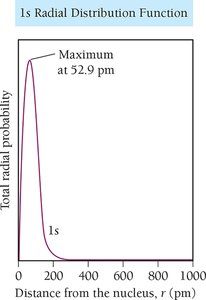
Nodes
Nodes are regions where the probability of finding an electron is zero. The number of nodes in an orbital is given by (n - 1).
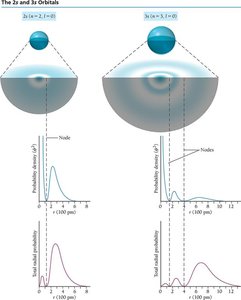
Phase of Orbitals
The sign of the wave function (phase) can be positive or negative, which is important in bonding and orbital interactions.

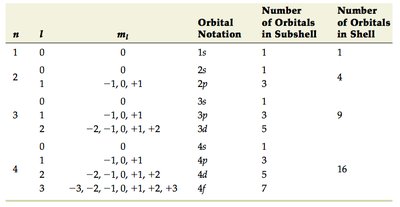
Summary Table: Quantum Numbers and Orbitals
n | l | ml | Orbital Notation | Number of Orbitals in Subshell | Number of Orbitals in Shell |
|---|---|---|---|---|---|
1 | 0 | 0 | 1s | 1 | 1 |
2 | 0 | 0 | 2s | 1 | 4 |
2 | 1 | -1, 0, +1 | 2p | 3 | 4 |
3 | 0 | 0 | 3s | 1 | 9 |
3 | 1 | -1, 0, +1 | 3p | 3 | 9 |
3 | 2 | -2, -1, 0, +1, +2 | 3d | 5 | 9 |
4 | 0 | 0 | 4s | 1 | 16 |
4 | 1 | -1, 0, +1 | 4p | 3 | 16 |
4 | 2 | -2, -1, 0, +1, +2 | 4d | 5 | 16 |
4 | 3 | -3, -2, -1, 0, +1, +2, +3 | 4f | 7 | 16 |
Additional info: This summary covers the quantum-mechanical model of the atom, including the nature of light, the photoelectric effect, atomic spectra, quantum numbers, and the shapes and properties of atomic orbitals. These concepts are foundational for understanding atomic structure and chemical behavior in general chemistry.
