Back
BackThermochemistry and Electronic Structure: Study Notes for General Chemistry
Study Guide - Smart Notes

Thermochemistry
Calorimetry: Measuring Heat Flow
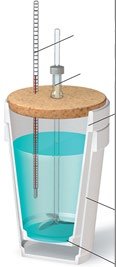
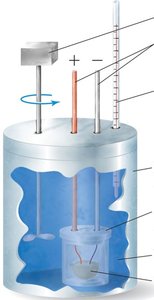
Calorimetry is the experimental technique used to measure the heat exchanged in chemical reactions or physical changes. Two common types of calorimeters are the coffee-cup calorimeter (constant pressure) and the bomb calorimeter (constant volume).
Coffee-Cup Calorimeter: Used for reactions in solution at constant atmospheric pressure. The heat measured corresponds to the enthalpy change, ΔH, of the reaction.
Bomb Calorimeter: Used for combustion reactions at constant volume. The heat measured corresponds to the change in internal energy, ΔE, of the reaction.
Bond Enthalpy and Reaction Energetics
Bond Enthalpy
Bond enthalpy (or bond dissociation energy) is the energy required to break one mole of a specific type of bond in a gaseous molecule. It is used to estimate the enthalpy change of reactions by considering the bonds broken and formed.
Breaking bonds: Requires energy (endothermic).
Forming bonds: Releases energy (exothermic).
Overall reaction enthalpy: ΔHrxn = Σ (bond enthalpies of bonds broken) - Σ (bond enthalpies of bonds formed)
Example: The combustion of ethane:
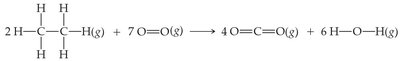
Electronic Structure of Atoms
Wave Nature of Light
Our understanding of atomic structure is based on the interaction of light (electromagnetic radiation) with matter. Light exhibits both wave-like and particle-like properties.
Wavelength (λ): The distance between two consecutive peaks of a wave.
Frequency (ν): The number of wave cycles passing a point per second (measured in Hz).
Speed of light (c): All electromagnetic radiation travels at the same speed in a vacuum, m/s.
Relationship:

The Electromagnetic Spectrum
The electromagnetic spectrum encompasses all types of electromagnetic radiation, from gamma rays to radio waves. Visible light is only a small portion of the spectrum.
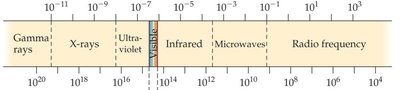
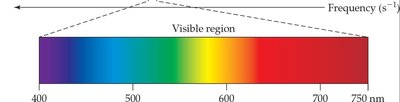
Units of Wavelength
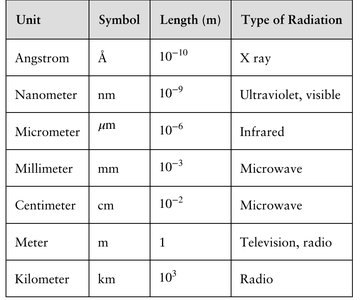
Different regions of the electromagnetic spectrum are described using different units of wavelength.
Unit | Symbol | Length (m) | Type of Radiation |
|---|---|---|---|
Angstrom | Å | X ray | |
Nanometer | nm | Ultraviolet, visible | |
Micrometer | μm | Infrared | |
Millimeter | mm | Microwave | |
Centimeter | cm | Microwave | |
Meter | m | 1 | Television, radio |
Kilometer | km | Radio |
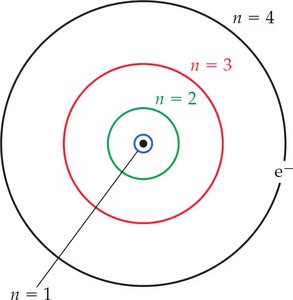
Bohr Model of the Atom
Quantized Orbits
Bohr proposed that electrons move in specific, quantized orbits around the nucleus, each with a fixed energy. The principal quantum number (n) designates these orbits.
As n increases, the orbit radius and energy increase.
The lowest energy state (n = 1) is called the ground state.
Higher n values correspond to excited states.
Quantum Mechanical Model
Electron Density and Orbitals
The quantum mechanical model describes electrons as wavefunctions, with the probability of finding an electron in a given region of space called electron density. Orbitals are regions where there is a high probability of finding an electron.
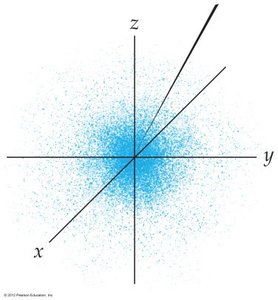
Nodes and Radial Probability
Nodes are regions where the probability of finding an electron is zero. The number of nodes increases with the principal quantum number (n).

Electron Spin and Quantum Numbers
Spin Magnetic Quantum Number
Electrons possess an intrinsic property called spin, which can have two possible values (+1/2 or -1/2). This property gives rise to magnetic behavior and is represented by the spin magnetic quantum number (ms).
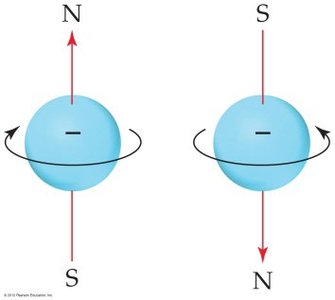
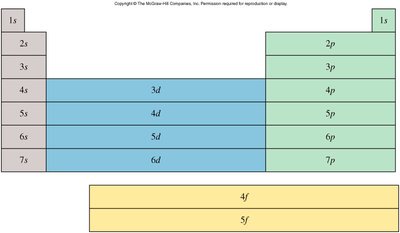
Electron Configurations and the Periodic Table
Orbital Filling and the Aufbau Principle
Electrons fill atomic orbitals in order of increasing energy, following the Aufbau principle. The arrangement of electrons in orbitals is called the electron configuration.
Periodic Table and Electron Configuration Blocks
The periodic table is organized into blocks (s, p, d, f) based on the type of atomic orbital being filled. This organization helps predict the electron configuration of elements.