 Back
BackThermochemistry and Quantum-Mechanical Model of the Atom: Study Notes
Study Guide - Smart Notes

Thermochemistry
Law of Conservation of Energy
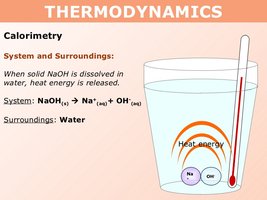
The law of conservation of energy states that energy cannot be created or destroyed, only transferred or transformed between the system and its surroundings. In chemical reactions, the system (the reaction itself) can either release energy to or absorb energy from the surroundings.
If the system loses energy (−ΔE), the surroundings gain that energy (+ΔE).
If the system gains energy (+ΔE), the surroundings lose that energy (−ΔE).
Change in Energy (ΔE)
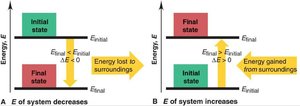
The change in energy of a system is calculated as the difference between the final and initial energy states:
ΔE = E_{final} – E_{initial}
If E_{final} < E_{initial}, ΔE is negative (energy is lost).
If E_{final} > E_{initial}, ΔE is positive (energy is gained).
For chemical reactions:
ΔE = E_{products} – E_{reactants}
Units of Energy
The SI unit of energy is the joule (J). Another common unit is the calorie (cal), where 1 cal = 4.184 J. In nutrition and chemistry, kilocalories (kcal) and kilojoules (kJ) are often used for convenience.

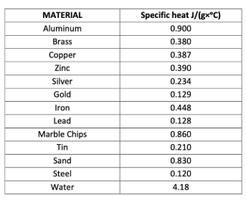
Specific Heat Capacity and Heat Calculations
Each substance has a unique specific heat capacity (C_s), which is the amount of heat required to raise the temperature of 1 gram of the substance by 1°C. The heat (q) absorbed or released is calculated by:
Where and m = mass in grams.
Different materials heat up at different rates due to their specific heat capacities.
MATERIAL | Specific heat J/(g×°C) |
|---|---|
Aluminum | 0.900 |
Brass | 0.380 |
Copper | 0.387 |
Zinc | 0.390 |
Silver | 0.234 |
Gold | 0.129 |
Iron | 0.448 |
Lead | 0.128 |
Marble Chips | 0.860 |
Tin | 0.210 |
Sand | 0.830 |
Steel | 0.120 |
Water | 4.18 |
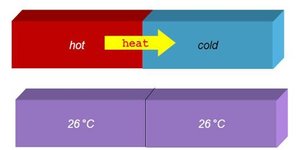
Thermal Equilibrium
Heat is the transfer of thermal energy from a hotter object to a colder one until both reach the same temperature, known as thermal equilibrium. Not all substances increase in temperature at the same rate when the same amount of heat is applied.
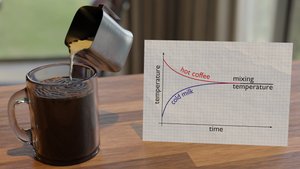
Calorimetry and Heat Exchange
In calorimetry, the heat lost by a hot object is equal to the heat gained by a cooler object at thermal equilibrium:
This principle is used to determine unknown masses, specific heats, or final equilibrium temperatures in calorimetry problems.
Enthalpy (ΔH): Heat at Constant Pressure
Enthalpy (H) is the heat evolved at constant pressure. For most reactions at constant pressure, the change in enthalpy (ΔH) is numerically equal to the change in energy (ΔE):
(heat at constant pressure)
Endothermic reaction: +ΔH (heat absorbed, feels cold)
Exothermic reaction: −ΔH (heat released, feels warm)
Thermochemical Equations and Stoichiometry
Thermochemical equations relate the enthalpy change to the stoichiometry of a reaction. For example:
C3H8(g) + 5O2(g) → 3CO2(g) + 4H2O(g) ΔHrxn = −2044 kJ
This means that 1 mole of propane releases 2044 kJ of heat when combusted completely.
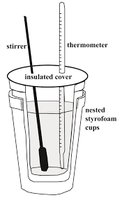
Constant Pressure Calorimetry (Coffee Cup Calorimeter)
A coffee cup calorimeter is a simple device used to measure the enthalpy change (ΔH) of reactions at constant pressure. It typically consists of nested Styrofoam cups, a thermometer, and a stirrer to minimize heat loss to the environment.
Hess’s Law
Hess’s Law states that the total enthalpy change for a reaction is the sum of the enthalpy changes for each step, regardless of the pathway taken. This allows calculation of ΔH for reactions that are difficult to measure directly by combining known reactions.
If a reaction is reversed, the sign of ΔH is reversed.
If a reaction is multiplied by a factor, ΔH is multiplied by the same factor.
For reactions occurring in steps, sum the ΔH values for each step.
Standard Enthalpies of Formation (ΔH°f)
The standard enthalpy of formation (ΔH°f) is the enthalpy change when one mole of a compound is formed from its elements in their standard states. For pure elements in their standard state, ΔH°f = 0.
ΔHrxn = Σ (mol) ΔH°f (products) − Σ (mol) ΔH°f (reactants)
The Quantum-Mechanical Model of the Atom
Quantum Mechanics and Atomic Structure
Quantum mechanics explains the arrangement of electrons in atoms and the periodic trends observed in the periodic table. Electrons exist in quantized energy levels and their behavior determines chemical bonding and properties.
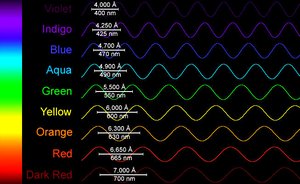
The Nature of Light: Waves and Particles
Light is a form of electromagnetic radiation, traveling as oscillating electric and magnetic fields at the speed of light (c = 3.00 × 108 m/s). Light exhibits both wave-like and particle-like properties.
Amplitude: Height of the wave, related to intensity/brightness.
Wavelength (λ): Distance between crests, determines color/energy.
Frequency (ν): Number of cycles per second (Hz).
c = λν
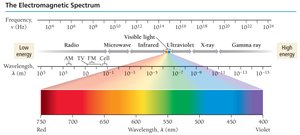
The Electromagnetic Spectrum
The electromagnetic spectrum includes all types of electromagnetic radiation, from radio waves to gamma rays. Visible light is only a small portion of the spectrum.
Energy of Photons and the Photoelectric Effect
Einstein proposed that light energy is quantized in packets called photons. The energy of a photon is proportional to its frequency and inversely proportional to its wavelength:
h = Planck’s constant = 6.626 × 10−34 J·s
The photoelectric effect demonstrates that electrons are ejected from a metal surface only when light of sufficient frequency (energy) strikes it.

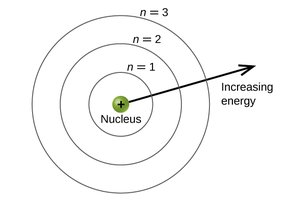
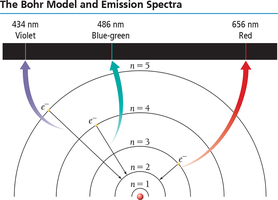
Atomic Spectroscopy and the Bohr Model
When atoms absorb energy, electrons are excited to higher energy levels. When they return to lower levels, they emit light at specific wavelengths, producing an emission spectrum unique to each element. The Bohr model describes electrons in quantized orbits around the nucleus.
Wave-Particle Duality and Electron Behavior
Electrons exhibit both wave-like and particle-like properties. The de Broglie wavelength of a particle is given by:
Electron diffraction experiments confirm the wave nature of electrons.

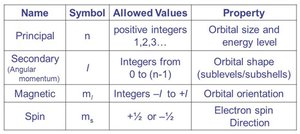
Quantum Numbers and Atomic Orbitals
The quantum-mechanical model uses four quantum numbers to describe the properties of atomic orbitals and electrons:
Name | Symbol | Allowed Values | Property |
|---|---|---|---|
Principal | n | 1, 2, 3, ... | Orbital size and energy level |
Angular momentum | l | 0 to n−1 | Orbital shape (s, p, d, f) |
Magnetic | m_l | −l to +l | Orbital orientation |
Spin | m_s | +1/2 or −1/2 | Electron spin direction |
Atomic Orbitals: Shapes and Types
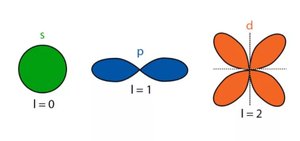
Atomic orbitals have characteristic shapes:
s orbitals: Spherical (l = 0)
p orbitals: Dumbbell-shaped (l = 1)
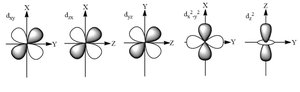
d orbitals: Cloverleaf-shaped (l = 2)
f orbitals: Complex shapes (l = 3)

Periodic Properties of the Elements
Mendeleev and the Periodic Law
Dmitri Mendeleev arranged elements by increasing atomic mass and observed repeating patterns of properties, leading to the periodic law. Modern periodic trends are explained by quantum mechanics and electron configurations.
Valence Electrons and Electron Configuration
Valence electrons are those in the outermost shell and determine chemical reactivity. Electron configurations describe the arrangement of electrons in shells and subshells, following the Aufbau principle, Pauli exclusion principle, and Hund’s rule.
Trends in Atomic Radius
Atomic radius decreases across a period (left to right) due to increasing effective nuclear charge, and increases down a group due to additional electron shells.
Ionization Energy and Electron Affinity
Ionization energy is the energy required to remove an electron from an atom. It increases across a period and decreases down a group. Electron affinity is the energy released when an atom gains an electron, with halogens having the highest values.
Properties of Metals and Nonmetals
Metals are malleable, ductile, shiny, and good conductors, while nonmetals are brittle, dull, and insulators. Metals tend to lose electrons (form cations), and nonmetals tend to gain electrons (form anions).
