 Back
BackThermochemistry: Energy, Heat, and Thermal Equilibrium
Study Guide - Smart Notes

Chapter 9: Thermochemistry
Introduction to Energy and Thermochemistry
Thermochemistry is the study of energy changes that occur during chemical reactions and physical transformations. Energy is a fundamental concept in chemistry, as it governs the behavior of atoms and molecules and their interactions.
Energy is the capacity to do work or produce heat.
Energy exists in various forms, including kinetic, potential, thermal, and chemical energy.
Thermochemistry focuses on the transfer of energy as heat and work between a system and its surroundings.
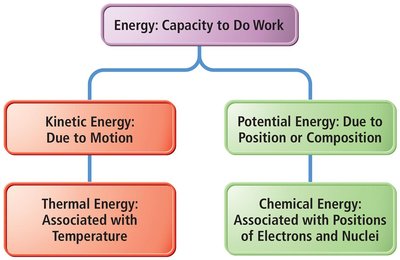
Types of Energy
Energy can be classified into several types, each relevant to chemical systems.
Kinetic Energy: Energy due to motion.
Thermal Energy: Associated with temperature; a form of kinetic energy.
Potential Energy: Energy due to position or composition.
Chemical Energy: Associated with the positions of electrons and nuclei in atoms and molecules.


Law of Conservation of Energy
The law of conservation of energy states that energy cannot be created or destroyed, only transferred or transformed.
The total energy of the universe is constant.
Energy transfer occurs between a system and its surroundings as heat (q) and work (w).
The change in internal energy (ΔE) of a system is the sum of heat and work:

Internal Energy and State Functions
Internal Energy (E): The sum of the kinetic and potential energies of all particles in a system.
Internal energy is a state function, meaning it depends only on the current state, not the path taken.
Change in internal energy:
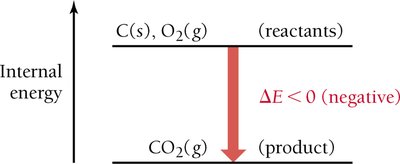
For chemical reactions:

Sign Conventions for q, w, and ΔE
The sign conventions for heat, work, and internal energy are important for interpreting energy changes.
Quantity | Positive (+) | Negative (-) |
|---|---|---|
q (heat) | System gains thermal energy | System loses thermal energy |
w (work) | Work done on the system | Work done by the system |
ΔE (internal energy) | Energy flows into the system | Energy flows out of the system |

First Law of Thermodynamics
The first law of thermodynamics is a restatement of the law of conservation of energy for thermodynamic systems.
The total energy of the universe is constant.
Energy transfer between system and surroundings:
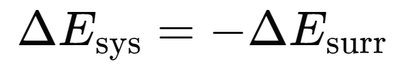
Endothermic and Exothermic Processes
Endothermic: System absorbs heat from surroundings (ΔE > 0).
Exothermic: System releases heat to surroundings (ΔE < 0).
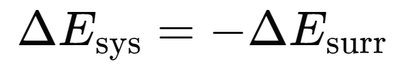
Specific Heat Capacity
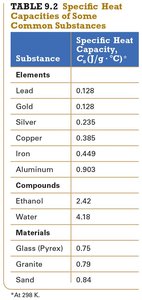
Specific heat capacity (Cs): The amount of heat required to raise the temperature of 1 gram of a substance by 1°C (units: J/g·°C).
Different substances have different specific heat capacities.
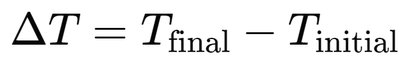
Calculating Heat Transfer
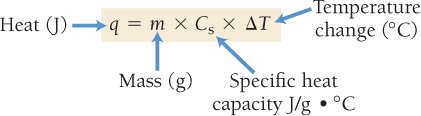
The heat transferred to or from a substance can be calculated using its mass, specific heat capacity, and temperature change.
Formula:
Where:
q: Heat (Joules)
m: Mass (grams)
Cs: Specific heat capacity (J/g·°C)
ΔT: Temperature change (°C)
Example: Cooling Aluminum
How much heat is released when 1.00 kg of aluminum cools from 35°C to 20°C?
Given: m = 1.00 × 10³ g, Cs = 0.903 J/g·°C, ΔT = 20.0°C – 35.0°C = -15.0°C
Calculation:

Heat Exchange and Thermal Equilibrium
When two substances at different temperatures are mixed or brought into contact, heat flows from the hotter to the cooler substance until thermal equilibrium is reached.
If thermally isolated, the heat lost by one substance equals the heat gained by the other:

Example: Aluminum and Water
A 32.5 g cube of aluminum at 45.8°C is submerged into 105.3 g of water at 15.4°C. What is the final temperature at thermal equilibrium?
Given:
Heat lost by aluminum equals heat gained by water:
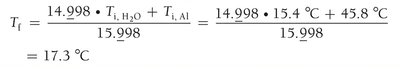
Substitute values:

Solve for final temperature:
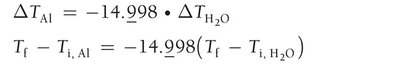
Summary Table: Specific Heat Capacities
Substance | Specific Heat Capacity, Cs (J/g·°C) |
|---|---|
Lead | 0.128 |
Gold | 0.128 |
Silver | 0.235 |
Copper | 0.385 |
Iron | 0.449 |
Aluminum | 0.903 |
Ethanol | 2.42 |
Water | 4.18 |
Glass (Pyrex) | 0.75 |
Granite | 0.79 |
Sand | 0.84 |

Key Equations in Thermochemistry
Change in internal energy:
Heat transfer:
Temperature change:
Heat exchange:
Practice and Application
Apply these concepts to solve problems involving heat transfer, specific heat, and thermal equilibrium.
Use the provided tables and equations to calculate energy changes in chemical and physical processes.

Additional info: The notes have been expanded to include full academic context, definitions, and step-by-step examples for clarity and completeness.
