 Back
BackThermochemistry: Energy, Heat, and Work in Chemical Systems
Study Guide - Smart Notes

Thermochemistry and the Nature of Energy
Introduction to Thermochemistry
Thermochemistry is the study of the relationships between chemistry and energy, focusing on how energy is transferred during chemical reactions and physical changes. Energy is defined as the capacity to do work, and it affects matter in various ways.
Energy: Anything with the capacity to do work.
Work: A force acting over a distance.
Heat: The flow of energy caused by a difference in temperature; always flows from hot to cold.
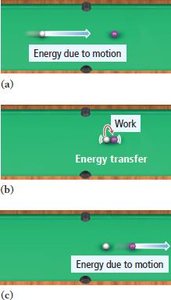

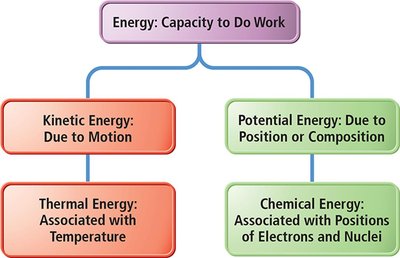
Classification of Energy
Energy can be classified into kinetic and potential forms, each with distinct characteristics and manifestations.
Kinetic Energy: Energy of motion or energy being transferred.
Potential Energy: Stored energy due to position or composition.
Kinetic and Potential Energy: Formulas
Kinetic and potential energy are quantified using specific mathematical relationships:
Kinetic Energy (KE):
Potential Energy (PE):
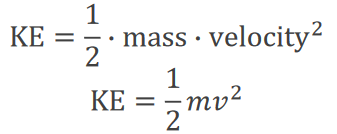
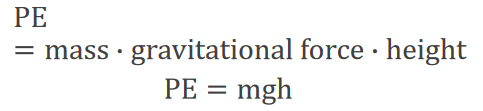
Work and Heat
Work and heat are two primary ways energy is exchanged between objects. Work is the result of a force acting through distance, while heat is the flow of energy caused by a temperature change.
Work: (force times distance)
Heat: Energy transfer due to temperature difference.
Energy, Heat, and Work in Chemical Systems
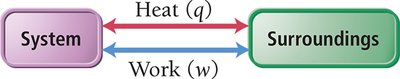
System and Surroundings
In thermochemistry, the system is the part of the universe under study, while the surroundings are everything else. Energy exchanges occur between the system and its surroundings.
Internal Energy (E): The sum of kinetic and potential energies of all particles in the system.
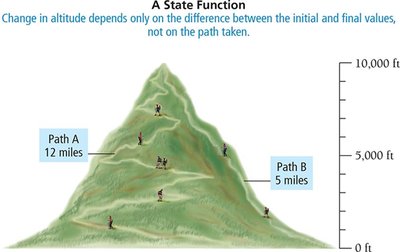
State Function: A property that depends only on the initial and final states, not the path taken.
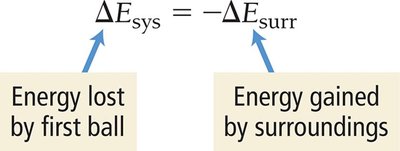
Energy Flow: First Law of Thermodynamics
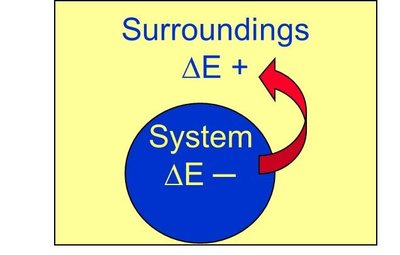
The first law of thermodynamics states that energy cannot be created or destroyed, only transferred. The total energy of the universe remains constant.
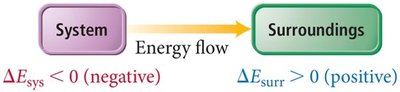
Energy flow out of system: (negative), (positive)
Energy flow into system: (positive), (negative)
Relationship:

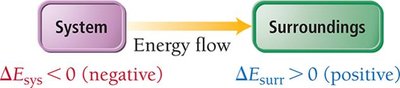
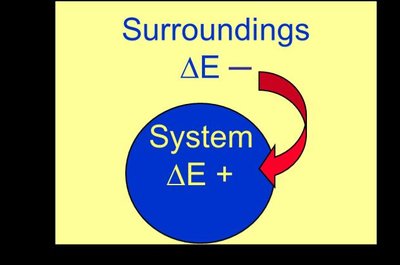

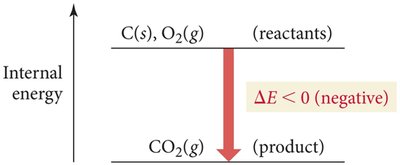
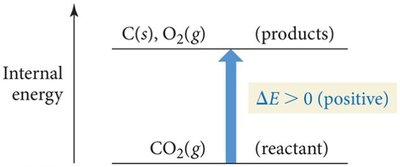
Energy Diagrams
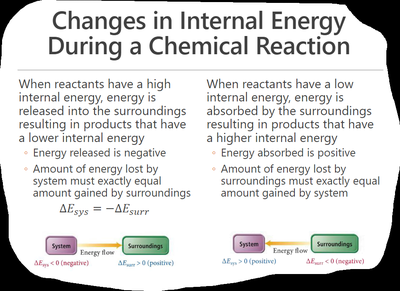
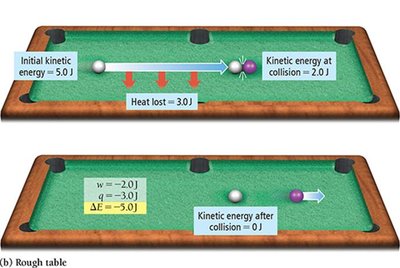
Energy diagrams visually represent the direction and magnitude of energy flow during a process. If reactants have higher internal energy than products, energy flows out of the system and is negative. If reactants have lower internal energy than products, energy flows into the system and $ \Delta E $ is positive.

Energy Exchange: Heat and Work
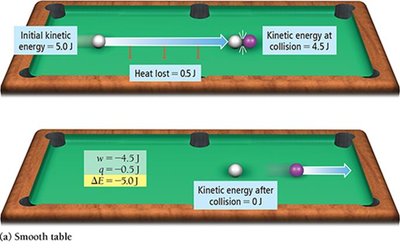
Energy is exchanged between the system and surroundings through heat (q) and work (w). The change in internal energy is given by:
Formula:
Sign conventions:
q > 0: System gains thermal energy
q < 0: System loses thermal energy
w > 0: Work done on the system
w < 0: Work done by the system
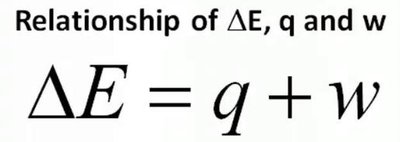
Heat Capacity and Specific Heat
Heat Capacity
When a system absorbs heat, its temperature increases. The proportionality constant between heat absorbed and temperature rise is called heat capacity (C).
Formula:
Units: J/°C or J/K
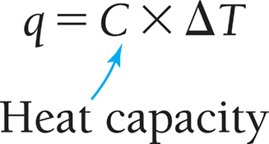
Specific Heat Capacity
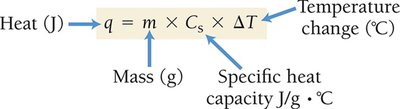
Specific heat capacity () is the amount of heat required to raise the temperature of one gram of a substance by one degree Celsius. Molar heat capacity is the heat required to raise the temperature of one mole of a substance.
Formula:
Units: J/g·°C
Thermal Energy Transfer
Heat Exchange Between Objects
When two objects at different temperatures are placed in contact, heat flows from the hotter to the colder object. The heat lost by the hot material equals the heat gained by the cold material.
Formula:

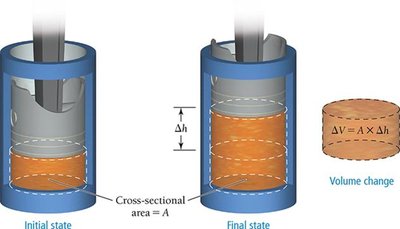
Pressure–Volume Work
PV Work
Pressure–volume (PV) work is caused by a volume change against an external pressure, commonly observed in gas reactions.
Formula:
To convert units:
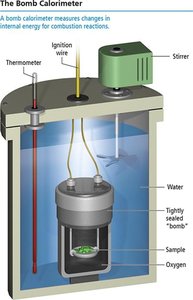
Calorimetry
Constant Volume Calorimetry
Calorimetry is used to measure thermal energy exchanged between a reaction and its surroundings. A bomb calorimeter operates at constant volume and is used for combustion reactions.
Formula: (at constant volume, w = 0)
Calorimeter constant: Amount of heat absorbed per degree rise in temperature.
Enthalpy and Chemical Reactions
Enthalpy (H)
Enthalpy is the sum of the internal energy and the product of pressure and volume. It is a state function, and the enthalpy change () of a reaction is the heat involved at constant pressure.
Formula:
Enthalpy change: (at constant pressure)
Endothermic and Exothermic Reactions
Reactions can be classified based on the sign of :
Endothermic: ; heat is absorbed, surroundings feel cold.
Exothermic: ; heat is released, surroundings feel warm.
Enthalpy of Reaction and Hess's Law
The enthalpy change for a reaction is an extensive property and can be calculated using Hess's Law, which states that the enthalpy change for an overall reaction is the sum of the enthalpy changes for each step.
Formula:
Standard Enthalpy of Formation
The standard enthalpy of formation () is the enthalpy change for forming one mole of a compound from its elements in their standard states. For a pure element in its standard state, kJ/mol.
Standard state: Pure gas at 1 atm, pure solid/liquid in most stable form at 1 atm and 25°C, solution at 1 M concentration.
Summary Table: Energy Types and Formulas
Type of Energy | Formula | Units |
|---|---|---|
Kinetic Energy | Joules (J) | |
Potential Energy | Joules (J) | |
Heat Capacity | Joules (J) | |
Specific Heat Capacity | Joules (J) | |
Internal Energy Change | Joules (J) | |
PV Work | Joules (J) | |
Enthalpy Change | kJ/mol |
