 Back
BackThermodynamics: Spontaneity, Entropy, and Free Energy
Study Guide - Smart Notes

Thermodynamics and Spontaneity
Introduction to Thermodynamics
Thermodynamics is the study of the relationships between energy, work, and heat in chemical and physical processes. It allows chemists to predict whether a process will occur under specified conditions, focusing on the concepts of spontaneity and equilibrium.
Spontaneous process: Occurs naturally under certain conditions without external intervention (e.g., water flowing downhill, iron rusting).
Nonspontaneous process: Requires continual input of energy from an external source (e.g., pumping water uphill).
Spontaneity is not related to the speed of a process; a process can be spontaneous but extremely slow.
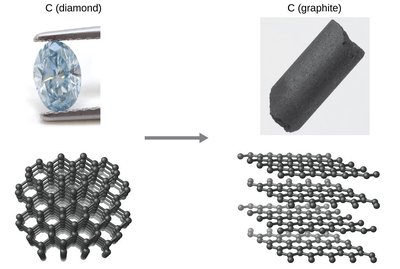
Example: The conversion of diamond to graphite is thermodynamically spontaneous at ambient pressure, but the process is so slow that diamonds persist for geological timescales. This illustrates the distinction between thermodynamic and kinetic stability.
Dispersal of Matter and Energy
Spontaneous processes often result in a more uniform distribution of matter or energy. For example, when a gas expands into a vacuum, it spreads out to fill the available space, increasing the dispersal of matter.
In an isolated system, the expansion of a gas into a vacuum is spontaneous and results in no change in internal energy, but a greater dispersal of matter.
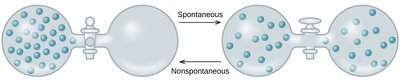
Example: When two objects at different temperatures are brought into contact, heat flows spontaneously from the hotter to the colder object, resulting in a more uniform distribution of thermal energy.

Entropy (S)
Definition and Statistical Interpretation
Entropy is a thermodynamic property that quantifies the dispersal of energy and matter in a system. It is a state function, meaning its value depends only on the current state of the system, not the path taken to reach that state.
Reversible process: Occurs infinitely slowly, always at equilibrium, and can be reversed by an infinitesimal change in conditions.
Irreversible process: Real processes that cannot be reversed without leaving a net change in the system and surroundings.
Clausius introduced entropy (S) to relate heat flow to temperature in thermodynamic processes.
Boltzmann provided a molecular interpretation of entropy, relating it to the number of possible microstates (W) of a system:
Microstate: A specific arrangement of the locations and energies of the particles in a system.
The number of microstates increases exponentially with the number of particles.

Example: Distributing 4 particles among 2 boxes results in 16 microstates. The most probable distribution is the one with the greatest entropy, where particles are evenly distributed.

Entropy and Spontaneous Processes
Spontaneous processes are characterized by an increase in the entropy of the universe. For example, the expansion of a gas or the flow of heat from hot to cold both result in increased entropy.

Predicting the Sign of Entropy Change (ΔS)
The sign of ΔS for a process can often be predicted by considering the change in the number of microstates or the dispersal of matter and energy:
ΔS > 0: Increase in entropy (e.g., melting, vaporization, dissolution, heating).
ΔS < 0: Decrease in entropy (e.g., freezing, condensation, precipitation).
For any substance: Sgas > Sliquid > Ssolid.
The Laws of Thermodynamics
Second Law of Thermodynamics
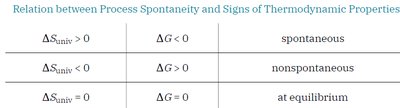
The second law states that all spontaneous changes increase the entropy of the universe:
If ΔSuniv > 0, the process is spontaneous.
If ΔSuniv < 0, the process is nonspontaneous (spontaneous in the reverse direction).
If ΔSuniv = 0, the system is at equilibrium.

Third Law of Thermodynamics
The third law states that the entropy of a pure, perfectly crystalline substance at absolute zero (0 K) is zero. This provides a reference point for measuring absolute entropies.
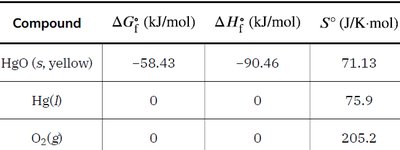
Standard Entropies and Calculations
Standard entropy values (S°) are tabulated for substances under standard conditions (1 bar, 298.15 K). The standard entropy change for a reaction is calculated as:


Gibbs Free Energy (G)
Definition and Significance
Gibbs free energy (G) is a thermodynamic function that combines enthalpy and entropy to predict the spontaneity of a process at constant temperature and pressure. The change in free energy (ΔG) is given by:
ΔG < 0: Process is spontaneous.
ΔG > 0: Process is nonspontaneous.
ΔG = 0: System is at equilibrium.

Calculating Free Energy Changes
ΔG can be calculated using standard enthalpy and entropy values, or from standard free energies of formation (ΔGf°):

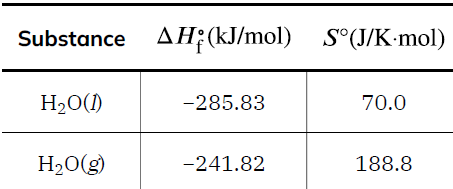

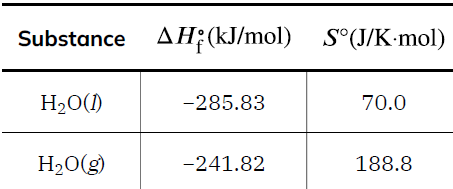
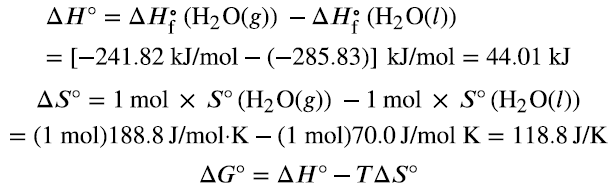

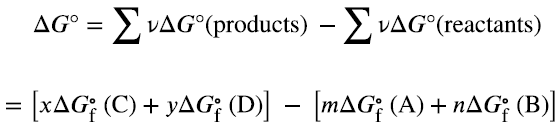
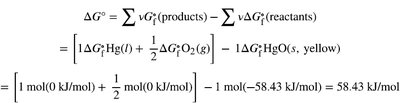
Temperature Dependence of Spontaneity
The spontaneity of a process can depend on temperature, as reflected in the signs of ΔH and ΔS:
ΔH > 0, ΔS > 0: Spontaneous at high T.
ΔH < 0, ΔS < 0: Spontaneous at low T.
ΔH > 0, ΔS < 0: Nonspontaneous at all T.
ΔH < 0, ΔS > 0: Spontaneous at all T.
Free Energy and Equilibrium
At equilibrium, ΔG = 0. The relationship between ΔG and the reaction quotient (Q) is:
At equilibrium, Q = K and ΔG = 0, so:
This equation links thermodynamics and equilibrium, allowing calculation of equilibrium constants from free energy changes.
Summary Table: Spontaneity and Thermodynamic Properties
ΔSuniv | ΔG | Process |
|---|---|---|
> 0 | < 0 | Spontaneous |
< 0 | > 0 | Nonspontaneous |
= 0 | = 0 | At equilibrium |
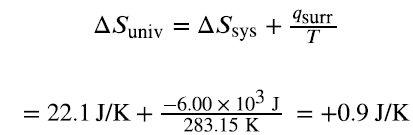
Key Equations:
Example Applications:
Predicting whether ice will melt or freeze at a given temperature using ΔSuniv.
Calculating the standard entropy change for a reaction using tabulated S° values.
Determining the spontaneity of a reaction at different temperatures using ΔG.
Relating free energy changes to equilibrium constants for chemical reactions.
