 Back
BackValence Bond and Molecular Orbital Theory: Chemical Bonding II
Study Guide - Smart Notes

Chemical Bonding II: Valence Bond and Molecular Orbital Theory
Introduction to Bonding Theories
Chemical bonding can be explained using several models, each with increasing complexity and accuracy. The Lewis Model, Valence Bond (VB) Theory, and Molecular Orbital (MO) Theory are foundational for understanding how atoms combine to form molecules and how molecular geometry arises from atomic orbitals.
Valence Bond Theory
Basic Principles of Valence Bond Theory
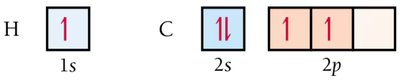
Valence electrons reside in quantum mechanical atomic orbitals (s, p, d, f, or hybrid orbitals).
A covalent bond forms when two half-filled atomic orbitals overlap, pairing their electrons.
The geometry of the overlapping orbitals determines the shape of the molecule.
Valence Bond Theory explains bonding as the overlap of atomic orbitals, but sometimes pure atomic orbitals do not predict observed molecular geometries, requiring the concept of hybridization.
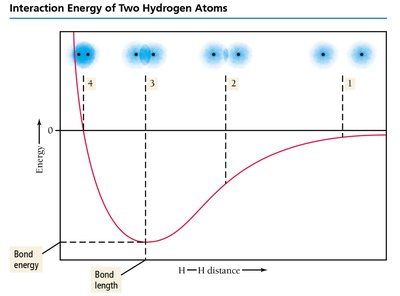
Interaction Energy and Bond Formation
As two atoms approach, their interaction energy changes due to the balance of attractive and repulsive forces. At the equilibrium bond distance, the energy is minimized, and a stable bond forms.
Atomic Orbitals and Molecular Geometry
Atomic orbitals can sometimes predict molecular geometry, but not always accurately. For example, in H2S, the predicted bond angle using pure p orbitals is 90°, but the observed angle is 92.1°.
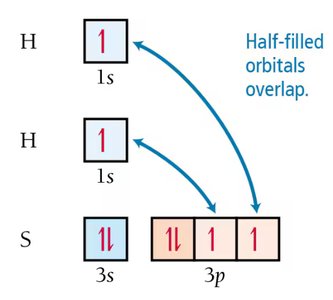
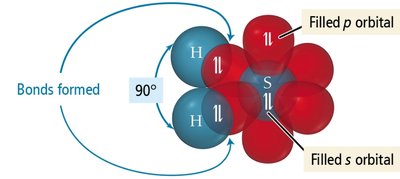
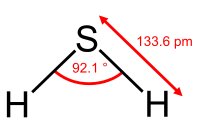
Hybridization of Atomic Orbitals
To explain observed geometries, atomic orbitals mix to form hybrid orbitals. The number of atomic orbitals mixed equals the number of hybrid orbitals formed.
sp3 hybridization: Mixing one s and three p orbitals forms four equivalent sp3 hybrid orbitals (e.g., CH4).
sp2 hybridization: Mixing one s and two p orbitals forms three sp2 hybrid orbitals (e.g., H2CO).
sp hybridization: Mixing one s and one p orbital forms two sp hybrid orbitals (e.g., C2H2).
sp3d and sp3d2 hybridizations explain expanded octets (e.g., PCl5, SF6).

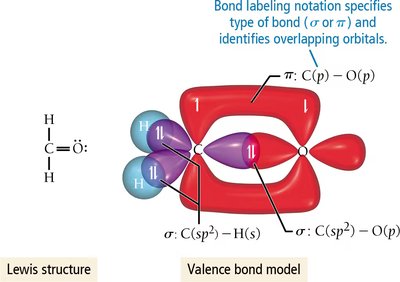
Single, Double, and Triple Bonds
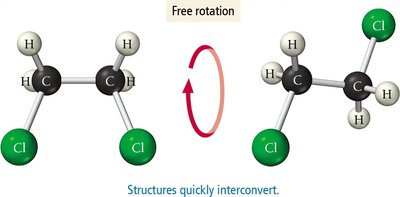
σ (sigma) bonds: Formed by end-to-end overlap of orbitals; allow free rotation.
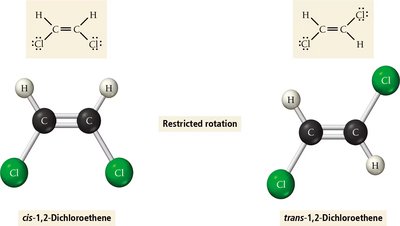
π (pi) bonds: Formed by side-to-side overlap of p orbitals; restrict rotation.
Double bonds: Consist of one σ and one π bond.
Triple bonds: Consist of one σ and two π bonds.
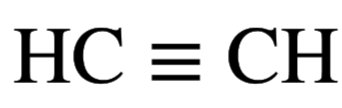
Summary Table: Types of Hybridization
Type | Atomic Orbitals Mixed | # Hybrid Orbitals | Example |
|---|---|---|---|
sp | 1 s + 1 p | 2 | BeCl2, C2H2 |
sp2 | 1 s + 2 p | 3 | BF3, H2CO |
sp3 | 1 s + 3 p | 4 | CH4, NH3 |
sp3d | 1 s + 3 p + 1 d | 5 | PCl5 |
sp3d2 | 1 s + 3 p + 2 d | 6 | SF6 |
Molecular Orbital Theory
Basic Principles of MO Theory
Molecular Orbital Theory describes bonding by combining atomic orbitals from all atoms in a molecule to form molecular orbitals that are spread out over the entire molecule. The Schrödinger equation is used to calculate these orbitals.
Bonding molecular orbitals are lower in energy than the original atomic orbitals.
Antibonding molecular orbitals are higher in energy.
Electrons fill molecular orbitals from lowest to highest energy.
Linear Combination of Atomic Orbitals (LCAO)
Atomic orbitals are mathematically combined (added or subtracted) to form molecular orbitals. Constructive interference leads to bonding orbitals, while destructive interference leads to antibonding orbitals.
Molecular Orbital Diagrams and Bond Order
MO diagrams show the relative energies of atomic and molecular orbitals. Bond order is calculated as:
If bond order > 0, a bond forms.
If bond order ≤ 0, no bond forms.
Examples
H2: Bond order = 1 (stable molecule)
He2: Bond order = 0 (no stable molecule)
He2+: Bond order = 0.5 (exists as a cation)
MO Theory for Polyatomic Molecules
MO theory can be extended to polyatomic molecules, providing a more accurate description of delocalized electrons and resonance structures. It also explains magnetic properties and molecules with odd numbers of electrons.
Comparison: Valence Bond vs. Molecular Orbital Theory
Valence Bond Theory | Molecular Orbital Theory |
|---|---|
Orbitals localized on atoms | Orbitals delocalized over molecule |
Explains molecular geometry well | Explains resonance and magnetism well |
Uses hybridization to explain shapes | Uses Schrödinger equation for orbitals |
Summary
Valence Bond Theory explains bonding through the overlap of half-filled orbitals and hybridization, predicting molecular geometry.
Molecular Orbital Theory combines all atomic orbitals to form molecular orbitals, explaining bond order, resonance, and magnetic properties.
Both theories are essential for a complete understanding of chemical bonding.
