 Back
BackWriting Equilibrium Constant Expressions for Chemical Equations
Study Guide - Smart Notes

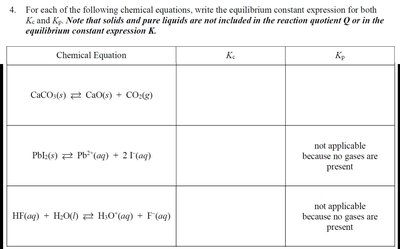
Q4. For each of the following chemical equations, write the equilibrium constant expression for both Kc and Kp. Note that solids and pure liquids are not included in the reaction quotient Q or in the equilibrium constant expression K.
Background
Topic: Chemical Equilibrium and Equilibrium Constants
This question tests your understanding of how to write equilibrium constant expressions (Kc for concentration and Kp for partial pressure) for different types of chemical reactions. It also checks your knowledge of which species are included in these expressions (only gases and aqueous species, not solids or pure liquids).
Key Terms and Formulas
Kc (Equilibrium Constant for Concentration): Uses molar concentrations of reactants and products.
Kp (Equilibrium Constant for Partial Pressure): Uses partial pressures of gaseous reactants and products.
Solids and pure liquids are omitted from equilibrium expressions.
General formula for a reaction:
Step-by-Step Guidance
For each equation, identify which species are included in the equilibrium expression (exclude solids and pure liquids).
Write the Kc expression using only the concentrations of aqueous and gaseous species.
For reactions involving gases, write the Kp expression using partial pressures. If no gases are present, note that Kp is not applicable.
Set up the equilibrium constant expressions for each reaction, but do not solve or simplify them yet.
Try solving on your own before revealing the answer!
