Ethyl propanoate, CH3CH2COOCH2CH3, gives a fruity pineapple-like smell. (e) What are the approximate bond angles around each carbon atom in the molecule?
Ch.9 - Molecular Geometry and Bonding Theories

Brown14th EditionChemistry: The Central ScienceISBN: 9780134414232Not the one you use?Change textbook
Chapter 9, Problem 91
From their Lewis structures, determine the number of σ and π bonds in each of the following molecules or ions: (a) hydrazine, N2H4 (b) hydrogen cyanide, HCN (c) sulphur trioxide, SO3
 Verified step by step guidance
Verified step by step guidance1
Step 1: Draw the Lewis structure for each molecule or ion. For hydrazine (N2H4), connect the two nitrogen atoms with a single bond and attach two hydrogen atoms to each nitrogen. For hydrogen cyanide (HCN), connect the hydrogen to carbon with a single bond and carbon to nitrogen with a triple bond. For sulfur trioxide (SO3), draw sulfur in the center with three double bonds to three oxygen atoms.
Step 2: Identify the σ (sigma) bonds in each molecule. Sigma bonds are the first bonds formed between two atoms. In N2H4, each N-H bond and the N-N bond are sigma bonds. In HCN, the H-C bond and one of the C-N bonds are sigma bonds. In SO3, each S-O bond is a sigma bond.
Step 3: Identify the π (pi) bonds in each molecule. Pi bonds are the second and third bonds formed between two atoms. In N2H4, there are no pi bonds as all bonds are single. In HCN, the remaining two bonds in the C-N triple bond are pi bonds. In SO3, each S-O double bond contains one pi bond.
Step 4: Count the total number of σ and π bonds in each molecule. For N2H4, count the total number of sigma bonds. For HCN, count the sigma and pi bonds separately. For SO3, count the sigma and pi bonds separately.
Step 5: Summarize the number of σ and π bonds for each molecule. Ensure that the total number of bonds matches the number of bonds in the Lewis structure for each molecule.
Key Concepts
Here are the essential concepts you must grasp in order to answer the question correctly.
Lewis Structures
Lewis structures are diagrams that represent the bonding between atoms in a molecule and the lone pairs of electrons that may exist. They help visualize the arrangement of electrons and the connectivity of atoms, allowing chemists to predict molecular geometry and reactivity. Understanding how to draw and interpret Lewis structures is essential for identifying the types of bonds present in a molecule.
Recommended video:
Guided course

Lewis Dot Structures: Ions
Sigma (σ) and Pi (π) Bonds
Sigma (σ) bonds are the first bonds formed between two atoms, created by the head-on overlap of atomic orbitals. They allow for free rotation around the bond axis. In contrast, pi (π) bonds are formed by the side-to-side overlap of p orbitals and exist in addition to a sigma bond in double and triple bonds, restricting rotation. Recognizing the types of bonds in a molecule is crucial for understanding its stability and reactivity.
Recommended video:
Guided course
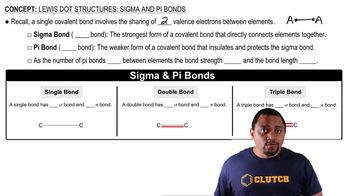
Lewis Dot Structures: Sigma & Pi Bonds
Molecular Geometry and Hybridization
Molecular geometry refers to the three-dimensional arrangement of atoms in a molecule, which is influenced by the types of bonds and lone pairs present. Hybridization is the concept of mixing atomic orbitals to form new hybrid orbitals that can accommodate bonding. Understanding these concepts helps predict the shape and bond angles in molecules, which are important for determining their physical and chemical properties.
Recommended video:
Guided course
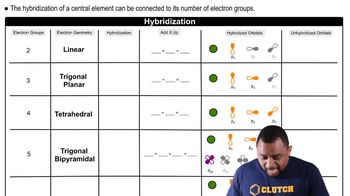
Hybridization and Electron Geometry
Related Practice
Textbook Question
2383
views
Textbook Question
Fill in the blank spaces in the following chart. If the molecule column is blank, find an example that fulfills the conditions of the rest of the row. Molecule Electron-Domain Hybridization Dipole Geometry of Central Atom Moment? Yes or No CO2 sp3 Yes sp3 No Trigonal planar No SF4 Octahedral No sp2 Yes Trigonal bipyramidal No XeF2
582
views
Textbook Question
Although I3- is a known ion, F3- is not. (d) Yet another classmate says F3- does not exist because F is too small to make bonds to more than one atom. Is this classmate possibly correct?
1325
views
1
rank
Textbook Question
Consider the molecule PF4Cl. (d) Would you expect the molecule to distort from its ideal electron-domain geometry? If so, how would it distort?
707
views
Textbook Question
An AB5 molecule adopts the geometry shown here. (c) Suppose the B atoms are halogen atoms. Of which group in the periodic table is atom A a member: (i) Group 15, (ii) Group 16, (iii) Group 17, (iv) Group 18, or (v) More information is needed?
618
views
