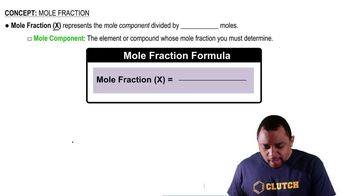
Weight Percent and Mole Fraction
Weight percent is a way of expressing the concentration of a component in a mixture, calculated as the mass of the component divided by the total mass of the mixture, multiplied by 100. To find the mole fraction, which is necessary for calculating partial pressures, the weight percent must be converted to moles using the molar mass of each gas.

 Verified step by step guidance
Verified step by step guidance